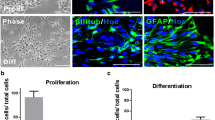
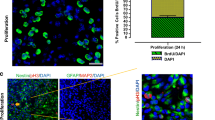
Abstract
A substantial lack of information is recognized on the features underlying the variable susceptibility to amyloid aggregate toxicity of cells with different phenotypes. Recently, we showed that different cell types are variously affected by early aggregates of a prokaryotic hydrogenase domain (HypF-N). In the present study we investigated whether differentiation affects cell susceptibility to amyloid injury using a human neurotypic SH-SY5Y cell differentiation model. We found that retinoic acid-differentiated cells were significantly more resistant against Aβ1-40, Aβ1-42 and HypF-N prefibrillar aggregate toxicity respect to undifferentiated cells treated similarly. Earlier and sharper increases in cytosolic Ca2+ and ROS with marked lipid peroxidation and mitochondrial dysfunction were also detected in exposed undifferentiated cells resulting in apoptosis activation. The reduced vulnerability of differentiated cells matched a more efficient Ca2+-ATPase equipment and a higher total antioxidant capacity. Finally, increasing the content of membrane cholesterol resulted in a remarkable reduction of vulnerability and ability to bind the aggregates in either undifferentiated and differentiated cells.








Similar content being viewed by others
Abbreviations
- BSA:
-
Bovine serum albumin
- CR:
-
Congo Red
- CM-H2,DCFDA:
-
2′,7′-Dichlorodihydrofluorescein diacetate, acetyl ester
- DCF:
-
Dichlorofluorescein
- DMEM:
-
Dulbecco’s Modified Eagle’s Medium
- DMSO:
-
Dimethylsulfoxide
- ECL:
-
Enhanced chemiluminescence
- FAD:
-
Familial Alzheimer disease
- FBS:
-
Fetal bovine serum
- GAP43:
-
Growth-associated-protein
- HBSS:
-
Hanks balanced salt solution
- HEPES:
-
N-(2-Hydroxyethyl)piperazine-N′-(2-ethanesulfonic acid)
- HRP:
-
Horseradish peroxidase
- HypF-N:
-
N-Terminal domain of the prokaryotic hydrogenase maturation factor
- LDH:
-
Lactate dehydrogenase
- MTP:
-
Mitochondrial permeability transition pore
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- NOS:
-
NO synthase
- PBS:
-
Phosphate buffered saline
- PEG-cholesterol:
-
Polyoxyetanyl-cholesteryl sebacate
- PDTC:
-
Pyrrolidinedithiocarbamate
- PI:
-
Propidium iodide
- PMSF:
-
Phenylmethylsulphonylfluoride
- PS:
-
Phosphatidylserine
- PVDF:
-
Polyvinylidene difluoride
- RA:
-
Retinoic acid
- RNS:
-
Reactive nitrogen species
- ROS:
-
Reactive oxygen species
- SDS-PAGE:
-
Sodium dodecylsulfate polyacrylamide gel electrophoresis
- TFE:
-
Trifluoroethanol
- TM-AFM:
-
Tapping mode atomic force microscopy
References
Hardy J, Selkoe DJ (2002) The amyloid hypothesis of Alzheimer’s disease: progress and problems on the road to therapeutics. Science 297:353–356
Masters CL, Simms G, Weinman NA et al (1985) Amyloid plaque core protein in Alzheimer disease and Down syndrome. Proc Natl Acad Sci USA 82:4245–4249
Lacor PN, Buniel MC, Chang L et al (2004) Synaptic targeting by Alzheimer’s-related amyloid β oligomers. J Neurosci 24:10191–10200
Lesne S, Koh MT, Kotilinek L et al (2006) A specific amyloid-beta protein assembly in the brain impairs memory. Nature 440:352–357
Bucciantini M, Giannoni E, Chiti F et al (2002) Inherent toxicity of aggregates implies a common mechanism for protein misfolding diseases. Nature 416:507–511
Walsh DM, Klyubin I, Fadeeva JV et al (2002) Naturally secreted oligomers of amyloid beta protein potently inhibit hippocampal long-term potentiation in vivo. Nature 416:535–539
Gharibyan AL, Zamotin V, Yamanandra K et al (2007) Lysozyme amyloid oligomers and fibrils induce cellular death via different apoptotic/necrotic pathways. J Mol Biol 365:1337–1349
Novitskaya V, Bocharova OV, Bronstein I et al (2006) Amyloid fibrils of mammalian prion protein are highly toxic to cultured cells and primary neurons. J Biol Chem 281:13828–13836
Bokvist M, Lindstrom F, Watts A et al (2004) Two types of Alzheimer’s beta-amyloid (1–40) peptide membrane interactions: aggregation preventing transmembrane anchoring versus accelerated surface fibril formation. J Mol Biol 335:1039–1049
Kayed R, Head E, Thompson JL et al (2003) Common structure of soluble amyloid oligomers implies common mechanism of pathogenesis. Science 300:486–489
Stefani M, Dobson CM (2003) Protein aggregation and aggregate toxicity: new insights into protein folding, misfolding diseases and biological evolution. J Mol Med 81:678–699
Kourie JI, Henry CL (2002) Ion channel formation and membrane-linked pathologies of misfolded hydrophobic proteins: the role of dangerous unchaperoned molecules. Clin Exp Pharmacol Physiol 29:741–753
Wakabayashi M, Okada T, Kozutsumi Y et al (2005) GM1 ganglioside-mediated accumulation of amyloid beta-protein on cell membranes. Biochim Biophys Res Com 328:1019–1023
Zhu M, Souillac PO, Ionescu-Zanetti C et al (2002) Surface-catalyzed amyloid fibril formation. J Biol Chem 277:50914–50922
Montine TJ, Neely MD, Quinn JF et al (2002) Lipid peroxidation in aging brain and Alzheimer’s disease. Free Radic Biol Med 33:620–626
Mattson MP, Guo Q, Furukawa K et al (1998) Presenilins, the endoplasmic reticulum, and neuronal apoptosis in Alzheimer’s disease. J Neurochem 70:1–14
Cecchi C, Fiorillo C, Baglioni S et al (2007) Increased susceptibility to amyloid toxicity in familial Alzheimer’s fibroblasts. Neurobiol Aging 28:863–876
Cecchi C, Fiorillo C, Sorbi S et al (2002) Oxidative stress and reduced antioxidant defenses in peripheral cells from familial Alzheimer’s patients. Free Rad Biol Med 33:1372–1379
Anderluh G, Gutierrez-Aguirre I, Rabzelj S et al (2005) Interaction of stefin B in the prefibrillar oligomeric form with membranes. Correlation with cellular toxicity. FEBS J 72:3042–3051
Bucciantini M, Calloni G, Chiti F et al (2004) Prefibrillar amyloid protein aggregates share common features of cytotoxicity. J Biol Chem 279:31374–31382
Cecchi C, Baglioni S, Fiorillo C et al (2005) Insights into the molecular basis of the differing susceptibility of varying cell types to the toxicity of amyloid aggregates. J Cell Sci 118:3459–3470
Dargusch R, Schubert D (2002) Specificity of resistance to oxidative stress. J Neurochem 81:1394–1400
Benvenuti S, Saccardi R, Luciani P et al (2006) A Neuronal differentiation of human mesenchymal stem cells: changes in the expression of the Alzheimer’s disease-related gene seladin-1. Exp Cell Res 312:2592–2604
Haughey NJ, Nath A, Chan SL et al (2002) Disruption of neurogenesis by amyloid beta-peptide, and perturbed neural progenitor cell homeostasis, in models of Alzheimer’s disease. J Neurochem 83:1509–1524
Chiti F, Bucciantini M, Capanni C et al (2001) Solution conditions can promote formation of either amyloid protofilaments or mature fibrils from the HypF N-terminal domain. Protein Sci 10:2541–2547
Relini A, Torrassa S, Rolandi R et al (2004) Monitoring the process of HypF fibrillization and liposome permeabilization by protofibrils. J Mol Biol 338:943–957
Datki Z, Papp R, Zadori D et al (2004) In vitro model of neurotoxicity of Abeta 1–42 and neuroprotection by a pentapeptide: irreversible events during the first hour. Neurobiol Dis 17:507–515
Negre-Salvayre A, Auge N, Duval C et al (2002) Detection of intracellular reactive oxygen species in cultured cells using fluorescent probes. Methods Enzymol 352:62–71
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Saborido A, Delgado J, Megias A (1999) Measurement of sarcoplasmic reticulum Ca2+-ATPase activity and E-type Mg2+-ATPase activity in rat heart homogenates. Anal Biochem 268:79–88
Petronilli V, Miotto G, Canton M et al (1999) Transient and long-lasting openings of the mitochondrial permeability transition pore can be monitored directly in intact cells by changes in mitochondrial calcein fluorescence. Biophys J 76:725–754
Datki Z, Juhasz A, Galfi M et al (2003) Method for measuring neurotoxicity of aggregating polypeptides with the MTT assay on differentiated neuroblastoma cells. Brain Res Bull 62:223–229
Arispe N, Doh M (2002) Plasma membrane cholesterol controls the cytotoxicity of Alzheimer’s disease AbetaP (1-40) and (1-42) peptides. FASEB J 16:1526–1536
Butterfield DA (2004) Proteomics: a new approach to investigate oxidative stress in Alzheimer’s disease brain. Brain Res 1000:1–7
Demuro A, Mina E, Kayed R et al (2005) Calcium dysregulation and membrane disruption as a ubiquitous neurotoxic mechanism of soluble amyloid oligomers. J Biol Chem 280:17294–17300
Loo DT, Copani A, Pike CJ et al (1993) Apoptosis is induced by beta-amyloid in cultured central nervous system neurons. Proc Natl Acad Sci USA 90:7951–7955
Hensley K, Carney JM, Mattson MP et al (1994) A model for beta-amyloid aggregation and neurotoxicity based on free radical generation by the peptide: relevance to Alzheimer disease. Proc Natl Acad Sci USA 91:3270–3274
Yip CM, Elton EA, Darabie AA et al (2001) Cholesterol, a modulator of membrane-associated Abeta-fibrillogenesis and neurotoxicity. J Mol Biol 311:723–734
Olesen OF, Dago L, Mikkelsen JD (1998) Amyloid beta neurotoxicity in the cholinergic but not in the serotonergic phenotype of RN46A cells. Brain Res Mol Brain Res 57:266–274
Subasinghe S, Unabia S, Barrow CJ et al (2003) Cholesterol is necessary both for the toxic effect of Abeta peptides on vascular smooth muscle cells and for Abeta binding to vascular smooth muscle cell membranes. J Neurochem 84:471–479
Cecchi C, Pensalfini A, Stefani M, Baglioni S, Fiorillo C, Cappadona S, Caporale R, Nosi D, Ruggiero M, Liguri G (2008) Replicating neuroblastoma cells in different cell-cycle phases display different vulnerability to amyloid toxicity. J Mol Med 86:197–209
Cecchi C, Rosati F, Pensalfini A, Formigli L, Nosi D, Liguri G, Dichiara F, Morello M, Danza G, Pieraccini G, Peri A, Serio M, Stefani M (2008) Seladin-1/DHCR24 protects neuroblastoma cells against Aβ toxicity by increasing membrane cholesterol content. J Cell Mol Med 12 [Epub ahead of print]
Ischiropoulos H, Gow A, Thom SR et al (1999) Detection of reactive nitrogen species using 2,7-dichlorodihydrofluorescein and dihydrorhodamine 123. Methods Enzymol 301:367–373
Ferreiro E, Resende R, Costa R et al (2006) An endoplasmic-reticulum-specific apoptotic pathway is involved in prion and amyloid-beta peptides neurotoxicity. Neurobiol Dis 23:669–678
Orrenius S, Zhivotovsky B, Nicotera P (2003) Regulation of cell death: the calcium-apoptosis link. Nat Rev Mol Cell Biol 4:552–565
Squier TC (2001) Oxidative stress and protein aggregation during biological aging. Exp Gerontol 36:1539–1550
Chan CS, Guzman JN, Ilijic E et al (2007) ‘Rejuvenation’ protects neurons in mouse models of Parkinson’s disease. Nature 447:1081–1086
Resende R, Pereira C, Agostinho P et al (2007) Susceptibility of hippocampal neurons to Abeta peptide toxicity is associated with perturbation of Ca2+ homeostasis. Brain Res 1143:11–21
Acknowledgements
We thank Roberto Caporale for technical advice. This study was supported by grants from the Italian MIUR (project number 2005054147_001 and 2005053998_001) and Compagnia di San Paolo, Torino, Italy (ref. n. 2004.0995).
Author information
Authors and Affiliations
Corresponding author
Additional information
Special issue article in honor of Dr. Anna Maria Giuffrida-Stella.
Rights and permissions
About this article
Cite this article
Cecchi, C., Pensalfini, A., Liguri, G. et al. Differentiation Increases the Resistance of Neuronal Cells to Amyloid Toxicity. Neurochem Res 33, 2516–2531 (2008). https://doi.org/10.1007/s11064-008-9627-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-008-9627-7