Abstract
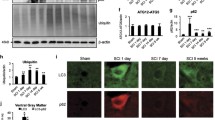
To demonstrate calpain involvement in neurodegeneration in rat spinal cord injury (SCI), we examined SCI segments for DNA fragmentation, neurons for calpain overexpression, neuronal death, and neuroprotection with calpain inhibitor (E-64-d). After the induction of SCI (40 g cm force) on T12, rats were treated within 15 min with vehicle (DMSO) or E-64-d. Sham animals underwent laminectomy only. Animals were sacrificed at 24 h, and five 1-cm long spinal cord segments were collected: two rostral (S1 and S2), one lesion (S3), and two caudal segments (S4 and S5). Agarose gel electrophoresis of DNA samples isolated from the SCI segments showed both random and internucleosomal DNA fragmentation indicating occurrence of necrosis as well as apoptosis mostly in the lesion, moderately in caudal, and slightly in rostral segments from SCI rats. Treatment of SCI rats with E-64-d (1 mg/kg) reduced DNA fragmentation in all segments. The lesion and adjacent caudal segments (S3 and S4) were further investigated by in situ double-immunofluorescent labelings that showed increase in calpain expression in neurons in SCI rats and decrease in calpain expression in SCI rats treated with E-64-d. In situ combined TUNEL and double-immunofluorescent labelings directly detected co-localization of neuronal death and calpain overexpressin in SCI rats treated with only vehicle while attenuation of neuronal death in SCI rats treated with E-64-d. Previous studies from our laboratory indirectly showed neuroprotective effect of E-64-d in SCI rats. Our current results provide direct in situ evidence for calpain involvement in neuronal death and neuroprotective efficacy of E-64-d in lesion and penumbra in SCI rats.




Similar content being viewed by others
References
National Spinal Cord Injury Statistical Center (2006) Spinal cord injury: facts and figures at a glance. National Spinal Cord Injury Statistical Center, Birmingham, AL
Dumont RJ, Okonkwo DO, Verma S et al (2001) Acute spinal cord injury, part I: pathophysiologic mechanisms. Clin Neuropharmacol 24:254–264
Ray SK, Matzelle DD, Wilford GG et al (2001) Cell death in spinal cord injury (SCI) requires de novo protein synthesis. Calpain inhibitor E-64-d provides neuroprotection in SCI lesion and penumbra. Ann NY Acad Sci 939:436–449
Beattie MS, Farooqui AA, Bresnahan JC (2000) Review of current evidence for apoptosis after spinal cord injury. J Neurotrauma 17:915–925
Dumont RJ, Verma S, Okonkwo DO et al (2001) Acute spinal cord injury, part II: contemporary pharmacotherapy. Clin Neuropharmacol 24:265–279
Li Y, Field PM, Raisman G (1997) Repair of adult rat corticospinal tract by transplants of olfactory ensheathing cells. Science 277:2000–2002
Nicholls J, Saunders N (1996) Regeneration of immature mammalian spinal cord after injury. Trends Neurosci 19:229–234
Liu XZ, Xu XM, Hu R et al (1997) Neuronal and glial apoptosis after traumatic spinal cord injury. J Neurosci 17:5395–5406
Springer JE, Azbill RD, Knapp PE (1999) Activation of the caspase-3 apoptotic cascade in traumatic spinal cord injury. Nat Med 5:943–946
Ray SK, Banik NL (2003) Calpain and its involvement in the pathophysiology of CNS injuries and diseases: therapeitic potential of calpain inhibitors for prevention of neurodegeneration. Curr Drug Targets – CNS Neurol Disord 2:173–189
Ray SK, Hogan EL, Banik NL (2003) Calpain in the pathophysiology of spinal cord injury: neuroprotection with calpain inhibitors. Brain Res Brain Res Rev 42:169–185
Wyllie AH, Morris RG, Smith AL et al (1984) Chromatin cleavage in apoptosis: association with condensed chromatin morphology and dependence on macromolecular synthesis. J Pathol 142:67–77
Ray SK, Schaecher KE, Shields DC et al (2000) Combined TUNEL and double immunofluorescent labeling for detection of apoptotic mononuclear phagocytes in autoimmune demyelinating disease. Brain Res Brain Res Protoc 5:305–311
Zhivotovsky B, Burgess DH, Vanags DM et al (1997) Involvement of cellular proteolytic machinery in apoptosis. Biochem Biophys Res Commun 230:481–488
Ray SK, Shields DC, Saido TC et al (1999) Calpain activity and translational expression increased in spinal cord injury. Brain Res 816:375–380
Banik NL, Chou CH, Deibler GE et al (1994) Peptide bond specificity of calpain: proteolysis of human myelin basic protein. J Neurosci Res 37:489–496
Jordan J, Galindo MF, Miller RJ (1997) Role of calpain- and interleukin-1β converting enzyme-like proteases in the β-amyloid-induced death of rat hippocampal neurons in culture. J Neurochem 68:1612–1621
Nath R, Raser KJ, Stafford D et al (1996) Non-erythroid α-spectrin breakdown by calpain and interleukin-1β-converting-enzyme-like protease(s) in apoptotic cells: contributory roles of both protease families in neuronal apoptosis. Biochem J 319(Pt 3):683–690
McGowan EB, Becker E, Detwiler TC (1989) Inhibition of calpain in intact platelets by the thiol protease inhibitor E-64-d. Biochem Biophys Res Commun 158:432–435
Perot PL Jr, Lee WA, Hsu CY et al (1987) Therapeutic model for experimental spinal cord injury in the rat: I. Mortality and motor deficit. J Neurotrauma 4:149–159
Ray SK, Matzelle DD, Wilford GG et al (2001) Inhibition of calpain-mediated apoptosis by E-64 d-reduced immediate early gene (IEG) expression and reactive astrogliosis in the lesion and penumbra following spinal cord injury in rats. Brain Res 916:115–126
Chakrabarti AK, Yoshida Y, Powers JM et al (1988) Calcium-activated neutral proteinase in rat brain myelin and subcellular fractions. J Neurosci Res 20:351–358
Pang Z, Bondada V, Sengoku T et al (2003) Calpain facilitates the neuron death induced by 3-nitropropionic acid and contributes to the necrotic morphology. J Neuropathol Exp Neurol 62:633–643
Kim MJ, Jo DG, Hong GS et al (2002) Calpain-dependent cleavage of cain/cabin1 activates calcineurin to mediate calcium-triggered cell death. Proc Natl Acad Sci USA 99:9870–9875
Wood DE, Thomas A, Devi LA et al (1998) Bax cleavage is mediated by calpain during drug-induced apoptosis. Oncogene 17:1069–1078
Blomgren K, Zhu C, Wang X et al (2001) Synergistic activation of caspase-3 by m-calpain after neonatal hypoxia-ischemia: a mechanism of “pathological apoptosis”? J Biol Chem 276:10191–10198
Nakagawa T, Yuan J (2000) Cross-talk between two cysteine protease families. Activation of caspase-12 by calpain in apoptosis. J Cell Biol 150:887–894
Ray SK, Wilford GG, Crosby CV et al (1999) Diverse stimuli induce calpain overexpression and apoptosis in C6 glioma cells. Brain Res 829:18–27
Varghese J, Radhika G, Sarin A (2001) The role of calpain in caspase activation during etoposide induced apoptosis in T cells. Eur J Immunol 31:2035–2041
Gores GJ, Miyoshi H, Botla R et al (1998) Induction of the mitochondrial permeability transition as a mechanism of liver injury during cholestasis: a potential role for mitochondrial proteases. Biochimica Biophys Acta 1366:167–175
Arataki S, Tomizawa K, Moriwaki A et al (2005) Calpain inhibitors prevent neuronal cell death and ameliorate motor disturbances after compression-induced spinal cord injury in rats. J Neurotrauma 22:398–406
Ray SK, Guyton MK, Sribnick EA et al (2007) Calpain as a target for prevention of neuronal death in injuries and diseases of the central nervous system. In: Lajtha A, Banik N (eds) Handbook of neurochemistry and molecular neurobiology: neural protein metabolism and function. Springer, New York, pp 445–467
Acknowledgments
This work was supported in part by R01 grants (NS-31622, NS-57811, and CA-91460) and a Medical Scientist Training Program (MSTP) grant (GM08716) from the National Institutes of Health, and also a Spinal Cord Injury Research Fund grants (SCIRF-0803 and SCIRF-1205) from the state of South Carolina.
Author information
Authors and Affiliations
Corresponding author
Additional information
Special issue in honor of Naren Banik.
Rights and permissions
About this article
Cite this article
Sribnick, E.A., Matzelle, D.D., Banik, N.L. et al. Direct Evidence for Calpain Involvement in Apoptotic Death of Neurons in Spinal Cord Injury in Rats and Neuroprotection with Calpain Inhibitor. Neurochem Res 32, 2210–2216 (2007). https://doi.org/10.1007/s11064-007-9433-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-007-9433-7