Abstract
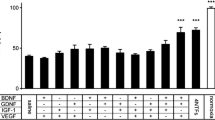
Pleiotrophin (PTN) is a heparin-binding growth factor involved in nerve regeneration after peripheral nerve injury. After crush injury, PTN is found in distal nerve segments in several non-neural cell types, including Schwann cells, macrophages, and endothelial cells, but not in axons. To further clarify the role for PTN in nerve regeneration, we investigated the effects of PTN applied to lesioned peripheral nerve in vivo. PTN in a dose of 1 mg/kg impaired muscle reinnervation. Thus, gastrocnemius muscle failed to recover its contractile properties as assessed by in situ maximal isometric tetanic force. PTN also decreased non-neural cell densities and delayed macrophage recruitment in the distal crushed nerve. These results are discussed in the light of recent evidence that PTN is a multifunctional polypeptide.





Similar content being viewed by others
References
Rauvala H (1989) An 18-kd heparin-binding protein of developing brain that is distinct from fibroblast growth factors. Embo J 8:2933–2941
Courty J, Dauchel MC, Caruelle D, Perderiset M, Barritault D (1991) Mitogenic properties of a new endothelial cell growth factor related to pleiotrophin. Biochem Biophys Res Commun 180:145–151
Li YS, Milner PG, Chauhan AK, Watson MA, Hoffman RM, Kodner CM, Milbrandt J, Deuel TF (1990) Cloning and expression of a developmentally regulated protein that induces mitogenic and neurite outgrowth activity. Science 250:1690–1694
Laaroubi K, Delbe J, Vacherot F, Desgranges P, Tardieu M, Jaye M, Barritault D, Courty J (1994) Mitogenic and in vitro angiogenic activity of human recombinant heparin affin regulatory peptide. Growth Factors 10:89–98
Rauvala H, Vanhala A, Castren E, Nolo R, Raulo E, Merenmies J, Panula P (1994) Expression of HB-GAM (heparin-binding growth-associated molecules) in the pathways of developing axonal processes in vivo and neurite outgrowth in vitro induced by HB-GAM. Brain Res Dev Brain Res 79:157–176
Silos-Santiago I, Yeh HJ, Gurrieri MA, Guillerman RP, Li YS, Wolf J, Snider W, Deuel TF (1996) Localization of pleiotrophin and its mRNA in subpopulations of neurons and their corresponding axonal tracts suggests important roles in neural-glial interactions during development and in maturity. J Neurobiol 31:283–296
Blondet B, Carpentier G, Lafdil F, Courty J (2005) Pleiotrophin cellular localization in nerve regeneration after peripheral nerve injury. J Histochem Cytochem 53:971–977
Seddon AP, Hulmes JD, Decker MM, Kovesdi I, Fairhurst JL, Backer J, Dougher-Vermazen M, Bohlen P (1994) Refolding and characterization of human recombinant heparin-binding neurite-promoting factor. Protein Expr Purif 5:14–21
Blanquaert F, Carpentier G, Morvan F, Caruelle JP, Barritault D, Tardieu M (2003) RGTA modulates the healing pattern of a defect in a monolayer of osteoblastic cells by acting on both proliferation and migration. J Biomed Mater Res 64A:525–532
Blondet B, Carpentier G, Ait-Ikhlef A, Murawsky M, Rieger F (2002) Motoneuron morphological alterations before and after the onset of the disease in the wobbler mouse. Brain Res 930:53–57
Kury P, Stoll G, Muller HW (2001) Molecular mechanisms of cellular interactions in peripheral nerve regeneration. Curr Opin Neurol 14:635–639
Nishio Y, Nishihira J, Ishibashi T, Kato H, Minami A (2002) Role of macrophage migration inhibitory factor (MIF) in peripheral nerve regeneration: anti-MIF antibody induces delay of nerve regeneration and the apoptosis of Schwann cells. Mol Med 8:509–520
Beuche W, Friede RL (1984) The role of non-resident cells in Wallerian degeneration. J Neurocytol 13:767–796
Liu HM, Yang LH, Yang YJ (1995) Schwann cell properties: 3. C-fos expression, bFGF production, phagocytosis and proliferation during Wallerian degeneration. J Neuropathol Exp Neurol 54:487–496
Tanaka K, Zhang QL, Webster HD (1992) Myelinated fiber regeneration after sciatic nerve crush: morphometric observations in young adult and aging mice and the effects of macrophage suppression and conditioning lesions. Exp Neurol 118:53–61
Ramon YCS (1928) Degeneration and regeneration of the nervous system. London, Oxford University press
Bunge RP (1980) Neurological mutants affecting myelination. Nature 286:106–107
Bovolenta P, Fernaud-Espinosa I (2000) Nervous system proteoglycans as modulators of neurite outgrowth. Prog Neurobiol 61:113–132
Hartmann U, Maurer P (2001) Proteoglycans in the nervous system––the quest for functional roles in vivo. Matrix Biol 20:23–35
Papadimitriou E, Polykratis A, Courty J, Koolwijk P, Heroult M, Katsoris P (2001) HARP induces angiogenesis in vivo and in vitro: implication of N or C terminal peptides. Biochem Biophys Res Commun 282:306–313
Vacherot F, Delbe J, Heroult M, Barritault D, Fernig DG, Courty J (1999) Glycosaminoglycans differentially bind HARP and modulate its biological activity. J Biol Chem 274:7741–7747
Heroult M, Bernard-Pierrot I, Delbe J, Hamma-Kourbali Y, Katsoris P, Barritault D, Papadimitriou E, Plouet J, Courty J (2004) Heparin affin regulatory peptide binds to vascular endothelial growth factor (VEGF) and inhibits VEGF-induced angiogenesis. Oncogene 23:1745–1753
Hienola A, Pekkanen M, Raulo E, Vanttola P, Rauvala H (2004) HB-GAM inhibits proliferation and enhances differentiation of neural stem cells. Mol Cell Neurosci 26:75–88
Chen S, Bisby MA (1993) Impaired motor axon regeneration in the C57BL/Ola mouseDelayed wallerian degeneration in sciatic nerves of C57BL/Ola mice is associated with impaired regeneration of sensory axons. J Comp Neurol 333:449–454
Hirata K, Mitoma H, Ueno N, He JW, Kawabuchi M (1999) Differential response of macrophage subpopulations to myelin degradation in the injured rat sciatic nerve. J Neurocytol 28:685–695
Hobson MI (2002) Increased vascularisation enhances axonal regeneration within an acellular nerve conduit. Ann R Coll Surg Engl 84:47–53
Hobson MI, Green CJ, Terenghi G (2000) VEGF enhances intraneural angiogenesis and improves nerve regeneration after axotomy. J Anat 197 (Pt 4):591–605
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Blondet, B., Carpentier, G., Ferry, A. et al. Exogenous Pleiotrophin Applied to Lesioned Nerve Impairs Muscle Reinnervation. Neurochem Res 31, 907–913 (2006). https://doi.org/10.1007/s11064-006-9095-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-006-9095-x