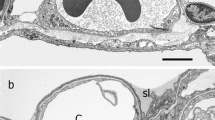
Electron microscopic study of the structural components of the frontal cerebral cortex, lumbar spinal ganglia, and sciatic nerve in rats with hypothyroidism induced by thyroidectomy was carried out. A 100-day-long period of deficiency of hormones produced by the thyroid gland evoked significant ultrastructural modifications in the examined regions of the nervous system; the pattern of these changes was mosaic and, to a certain extent, structure-specific. In the neocortex, many neurons were modified according to a light type; they contained a decreased number of the organelles and demonstrated manifestations of swelling. Other neurons were characterized by a high electronic density of the cytoplasm, and the integrity of the plasma membranes of such units was disturbed. Long-term hypothyroidism was accompanied by manifestations of swelling in cortical oligodendrocytes and astrocytes. Perivascularly localized astrocytes were found to be most sensitive to the negative influences of hypothyroidism; the respective shifts should inevitably influence the functioning of the blood-brain barrier. Nerve fibers in the cortex were considerably subjected to demyelination; swelling of the axons and synaptic terminals was observed. The number of vesicles in synaptic structures clearly decreased. Unidirectional changes were found in the somata of spinal ganglia neurons. Hypoplasia of all organelles, fragmentation of tubules of the endoplasmic reticulum, and generalized lysis of cristae and matrix in the mitochondria were observed. This was accompanied by an increase in the number of phagosomes, which was indicative of activation of the autolytic processes. Mantle gliocytes, unlike glial cells in the brain, demonstrated both dark- and light-type modifications. Processes of demyelination developed also in the structures of the peripheral nervous system, and the intensity of these changes was greater than in the brain. In both spinal ganglia and sciatic nerve, lamellae of the myelin sheaths separated from each other and became homogenized; axial cylinders were replaced by a degenerative thickened sheath. These changes were most clearly manifested in large-diameter myelinated fibers.
Similar content being viewed by others
References
G. A. Gerasimov and N. A. Petunina, Diseases of the Thyroid Gland [in Russian], Zdorov’ye, Moscow (1998).
V. V. Fadeyev and G. A. Melnichenko, Hypothyroidism (Manual for Physicians) [in Russian], RKI Severopress, Moscow (2002).
Yu. S. Voichulene, Epidemiological Study of Morbidity for Diseases of the Thyroid Gland in People Involved in Elimination of Consequences of the Chernobyl Disaster [in Ukrainian], Abstract of Canditate’s Thesis in Med. Sci., Chernovtsy (2009).
V. I. Kravtchenko and S. V. Postol, “Dynamics of morbidity for pathologies of the thyroid gland,” Éndokrinol. Sluzhba Ukr., 35, No. 3, 86-91 (2011).
T. O. Pertsova and O. M. Kulikova, “State of the reninangiotensin-aldosterone system in patients suffering from hypothyroidism and arterial hypertension,” Endokrinology, 9, No. 1, 97-100 (2004).
P. Kh. Dzhanashiya and G. B. Selivanova, “Hypothyroidism and arterial hypertension: Unresolved questions of pathogenesis, diagnostics, and pharmacotherapy,” Kardiovask. Ter. Profil., 2, No. 3, 125-132 (2004).
Kh. Aikhmali, Age-Related Peculiarities of Free-Radical Oxidation of Proteins and Lipids in the Brain of Rats with Hypothyroidism and Effects of Intense Physical Loading [in Ukrainian], Abstract of Canditate’s Thesis in Biol. Sci., Kharkiv (2008).
L. A. Shcheplyagina, “Problems of iodine deficiency,” Ross. Med. Zh., 11, 523-527 (1999).
L. A. Shcheplyagina, L. A. Nadezhdin, P. I. Khramtsov, and E. N. Sotnikova, “Iodine deficiency and mental development of children: Possibilities for correction,” Izd. Prakt. Vrachei, 4, No. 82, 143-147 (2011).
J. Bernal and J. Nunez, “Thyroid hormones and brain development,” Eur. J. Endocrinol., 133, No. 4, 390-398 (1995).
S. G. Amur, G. Shanker, and R. A. Pieringe, “Diagnostic and clinical significance of the MBP,” J. Neurochem., 43, 494-498 (1984).
A. P. Khokhlov and Yu. N. Savchenko, Myelinopathies and Demyelination Diseases [in Russian], Meditsina, Moscow (1990).
F. Barkhof, S. T. Frequin, and O. R. Hommes, “Physicochemical properties and biological role of myelin basic protein (MBP),” Neurology, 42, No. 1, 63-67 (1992).
V. P. Chekhonin, O. I. Gurina, T. B. Dmitriyeva, et al., “Basic protein of myelin. Structure, properties, function, and role in the diagnostics of demyelination diseases,” Vopr. Med. Khim., 5, No. 6, 95-101 (1992).
Patent for the invention No. 27821, State Patent of Ukraine, Method for Modeling of Hypothyroidism in Rats [in Russian], L. O. Stechenko, V. A. Petrenko, P. L. Byk, V. R. Kuzyan, and T. P. Kouftyreva, publ. Nov. 12, 2007, Byul. No. 2.
L. A. Stechenko, V. A. Petrenko, T. P. Kouftyreva, et al., The Heart in Hypothyroidism (an Experimental Study) [in Russian], Publ. House of the Ivano-Frank. Med. Univ., Kyiv (2008).
S. A. Mikhal’skii, T. Yu. Kvitnitskaya-Ryzhova, V. V. Beloshitskii, and S. P. Malysheva, “Apoptosis in the brain of rats of different ages after craniocerebral traumas; effects of gene therapy,” Probl. Star. Dolgolet., 20, No. 4, 113-117 (2011).
V. V. Beloshitskii, S. A. Mikhal’skii, and T. Yu. Kvitnitskaya-Ryzhova, “Effect of liposomal transfection of brain cells by the anoE3 gene on injuries of the axons resulting from experimental craniocerebral trauma,” Ukr. Neirokhir. Zh., 3, No. 3, 79-83 (2011).
L. M. Lapina, T. P. Kouftyreva, L. O. Stechenko, et al., “Ultrastructural changes in neurons of the brain cortex in the dynamics of hypothyroidism,” Morfol. Visnyk, 1, No. 3, 243-248 (2007).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rudyuk, T.Y., Chaikovskii, Y.B., Stechenko, L.O. et al. Hypothyroidism-Induced Ultrastructural Peculiarities of Neuronal and Glial Elements in the Neocortex and Peripheral Structures of the Nervous System. Neurophysiology 44, 115–122 (2012). https://doi.org/10.1007/s11062-012-9277-x
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11062-012-9277-x