Abstract
Introduction
The aim of the present study is to assess whether postoperative residual non-enhancing volume (PRNV) is correlated and predictive of overall survival (OS) in glioblastoma (GBM) patients.
Methods
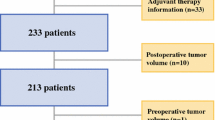
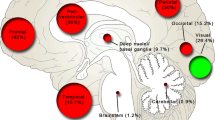
We retrospectively analyzed a total 134 GBM patients obtained from The University of Texas MD Anderson Cancer Center (training cohort, n = 97) and The Cancer Genome Atlas (validation cohort, n = 37). All patients had undergone postoperative magnetic resonance imaging immediately after surgery. We evaluated the survival outcomes with regard to PRNV. The role of possible prognostic factors that may affect survival after resection, including age, sex, preoperative Karnofsky performance status, postoperative nodular enhancement, surgically induced enhancement, and postoperative necrosis, was investigated using univariate and multivariate Cox proportional hazards regression analyses. Additionally, a recursive partitioning analysis (RPA) was used to identify prognostic groups.
Results
Our analyses revealed that a high PRNV (HR 1.051; p-corrected = 0.046) and old age (HR 1.031; p-corrected = 0.006) were independent predictors of overall survival. This trend was also observed in the validation cohort (higher PRNV: HR 1.127, p-corrected = 0.002; older age: HR 1.034, p-corrected = 0.022). RPA analysis identified two prognostic risk groups: low-risk group (PRNV < 70.2 cm3; n = 55) and high-risk group (PRNV ≥ 70.2 cm3; n = 42). GBM patients with low PRNV had a significant survival benefit (5.6 months; p = 0.0037).
Conclusion
Our results demonstrate that high PRNV is associated with poor OS. Such results could be of great importance in a clinical setting, particularly in the postoperative management and monitoring of therapy.


Similar content being viewed by others
References
Omuro A, DeAngelis LM (2013) Glioblastoma and other malignant gliomas: a clinical review. Jama 310:1842–1850
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114:97–109
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996
Rivera AL, Pelloski CE, Sulman E, Aldape K (2008) Prognostic and predictive markers in glioma and other neuroepithelial tumors. Curr Probl Cancer 32:97–123
Buckner JC (2003) Factors influencing survival in high-grade gliomas. Semin Oncol 30:10–14
Donato V, Papaleo A, Castrichino A, Banelli E, Giangaspero F, Salvati M, Delfini R (2007) Prognostic implication of clinical and pathologic features in patients with glioblastoma multiforme treated with concomitant radiation plus temozolomide. Tumori 93(3):248–256
Chaichana KL, Chaichana KK, Olivi A, Weingart JD, Bennett R, Brem H, Quinones-Hinojosa A (2011) Surgical outcomes for older patients with glioblastoma multiforme: preoperative factors associated with decreased survival. J Neurosurg 114:587–594
Lacroix M, Abi-Said D, Fourney DR, Gokaslan ZL, Shi W, DeMonte F, Lang FF, McCutcheon IE, Hassenbusch SJ, Holland E et al (2001) A multivariate analysis of 416 patients with glioblastoma multiforme: prognosis, extent of resection, and survival. J Neurosurg 95(2):190–198
Filippini G, Falcone C, Boiardi A, Broggi G, Bruzzone MG, Caldiroli D, Farina R, Farinotti M, Fariselli L, Finocchiaro G et al (2008) Prognostic factors for survival in 676 consecutive patients with newly diagnosed primary glioblastoma. Neuro-oncology 10:79–87
Tait MJ, Petrik V, Loosemore A, Bell BA, Papadopoulos MC (2007) Survival of patients with glioblastoma multiforme has not improved between 1993 and 2004: analysis of 625 cases. Br J Neurosurg 21:496–500
Pope WB, Sayre J, Perlina A, Villablanca JP, Mischel PS, Cloughesy TF (2005) MR imaging correlates of survival in patients with high-grade gliomas. AJNR Am J Neuroradiol 26(10):2466–2474
Al-Okaili RN, Krejza J, Wang S, Woo JH, Melhem ER (2006) Advanced MR imaging techniques in the diagnosis of intraaxial brain tumors in adults. Radiographics 26(Suppl 1):S173–S189
Wen PY, Macdonald DR, Reardon DA, Cloughesy TF, Sorensen AG, Galanis E, Degroot J, Wick W, Gilbert MR, Lassman AB et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28(11):1963–1972
Grabowski MM, Recinos PF, Nowacki AS, Schroeder JL, Angelov L, Barnett GH, Vogelbaum MA (2014) Residual tumor volume versus extent of resection: predictors of survival after surgery for glioblastoma. J Neurosurg 121:1–9
Hess KR (1999) Extent of resection as a prognostic variable in the treatment of gliomas. J Neurooncol 42:227–231
Iliadis G, Kotoula V, Chatzisotiriou A, Televantou D, Eleftheraki AG, Lambaki S, Misailidou D, Selviaridis P, Fountzilas G (2012) Volumetric and MGMT parameters in glioblastoma patients: survival analysis. BMC Cancer 12:3
Schoenegger K, Oberndorfer S, Wuschitz B, Struhal W, Hainfellner J, Prayer D, Heinzl H, Lahrmann H, Marosi C, Grisold W (2009) Peritumoral edema on MRI at initial diagnosis: an independent prognostic factor for glioblastoma? Eur J Neurol 16(7):874–878
Zinn PO, Mahajan B, Sathyan P, Singh SK, Majumder S, Jolesz FA, Colen RR (2011) Radiogenomic mapping of edema/cellular invasion MRI-phenotypes in glioblastoma multiforme. PLoS ONE 6:e25451
Zinn PO, Sathyan P, Mahajan B, Bruyere J, Hegi M, Majumder S, Colen RR (2012) A novel volume-age-KPS (VAK) glioblastoma classification identifies a prognostic cognate microRNA-gene signature. PLoS ONE 7:e41522
Stummer W, Reulen HJ, Meinel T, Pichlmeier U, Schumacher W, Tonn JC, Rohde V, Oppel F, Turowski B, Woiciechowsky C et al (2008) Extent of resection and survival in glioblastoma multiforme: identification of and adjustment for bias. Neurosurgery 62(3):564–576 (discussion 564–576)
Stummer W (2007) Mechanisms of tumor-related brain edema. Neurosurg Focus 22(5):E8
Lin ZX (2013) Glioma-related edema: new insight into molecular mechanisms and their clinical implications. Chin J Cancer 32(1):49–52
Brandes AA, Tosoni A, Franceschi E, Sotti G, Frezza G, Amistà P, Morandi L, Spagnolli F, Ermani M (2009) Recurrence pattern after temozolomide concomitant with and adjuvant to radiotherapy in newly diagnosed patients with glioblastoma: correlation with MGMT promoter methylation status. J Clin Oncol 27(8):1275–1279
Albert FK, Forsting M, Sartor K, Adams HP, Kunze S (1994) Early postoperative magnetic resonance imaging after resection of malignant glioma: objective evaluation of residual tumor and its influence on regrowth and prognosis. Neurosurgery 34(1):45–60 (discussion 60–41)
Fedorov A, Beichel R, Kalpathy-Cramer J, Finet J, Fillion-Robin JC, Pujol S, Bauer C, Jennings D, Fennessy F, Sonka M et al (2012) 3D Slicer as an image computing platform for the Quantitative Imaging Network. Magn Reson Imaging 30:1323–1341
Valeri G, Mazza FA, Maggi S, Aramini D, La Riccia L, Mazzoni G, Giovagnoni A (2014) Open source software in a practical approach for post processing of radiologic images. Radiol Med 120:309–323
Lescher S, Schniewindt S, Jurcoane A, Senft C, Hattingen E (2014) Time window for postoperative reactive enhancement after resection of brain tumors: less than 72 hours. Neurosurg Focus 37(6):E3
Oser AB, Moran CJ, Kaufman BA, Park TS (1997) Intracranial tumor in children: MR imaging findings within 24 hours of craniotomy. Radiology 205(3):807–812
Schumacher M, Binder H, Gerds T (2007) Assessment of survival prediction models based on microarray data. Bioinformatics 23(14):1768–1774
Clark TG, Bradburn MJ, Love SB, Altman DG (2003) Survival analysis part IV: further concepts and methods in survival analysis. Br J Cancer 89(5):781–786
Park C-K, Lee S-H, Han JH, Kim C-Y, Kim D-W, Paek SH, Kim DG, Heo DS, Kim IH, Jung H-W (2009) Recursive partitioning analysis of prognostic factors in WHO grade III glioma patients treated with radiotherapy or radiotherapy plus chemotherapy. BMC Cancer 9(1):450
Alkhasawneh MS, Ngah UK, Tay LT, Mat Isa NA, Al-Batah MS (2014) Modeling and testing landslide hazard using decision tree. J Appl Math 2014:9
Nestler U, Lutz K, Pichlmeier U, Stummer W, Franz K, Reulen HJ, Bink A (2014) Anatomic features of glioblastoma and their potential impact on survival. Acta Neurochir 157:179–186
Ogura K, Mizowaki T, Arakawa Y, Ogura M, Sakanaka K, Miyamoto S, Hiraoka M (2013) Initial and cumulative recurrence patterns of glioblastoma after temozolomide-based chemoradiotherapy and salvage treatment: a retrospective cohort study in a single institution. Radiat Oncol 8:97
Hou LC, Veeravagu A, Hsu AR, Tse VC (2006) Recurrent glioblastoma multiforme: a review of natural history and management options. Neurosurg Focus 20(4):E5
Li YM, Suki D, Hess K, Sawaya R (2015) The influence of maximum safe resection of glioblastoma on survival in 1229 patients: can we do better than gross-total resection?. J Neurosurg 124:1–12
Lin ZX, Yang LJ, Huang Q, Fu J (2010) Activated vascular endothelia regulate invasion of glioma cells through expression of fibronectin. Chin Med J 123(13):1754–1761
Hammoud MA, Sawaya R, Shi W, Thall PF, Leeds NE (1996) Prognostic significance of preoperative MRI scans in glioblastoma multiforme. J Neurooncol 27(1):65–73
Colen RR, Wang J, Singh SK, Gutman DA, Zinn PO (2015) Glioblastoma: imaging genomic mapping reveals sex-specific oncogenic associations of cell death. Radiology 275(1):215–227
Verhaak RG, Hoadley KA, Purdom E, Wang V, Qi Y, Wilkerson MD, Miller CR, Ding L, Golub T, Mesirov JP et al (2010) Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer cell 17(1):98–110
Brennan CW, Verhaak RG, McKenna A, Campos B, Noushmehr H, Salama SR, Zheng S, Chakravarty D, Sanborn JZ, Berman SH et al (2013) The somatic genomic landscape of glioblastoma. Cell 155(2):462–477
Acknowledgements
We thank The Cancer Genome Atlas and The Cancer Imaging Archive initiatives for making the clinical and imaging data publicly available. This research was partially funded by the John S. Dunn Sr. Distinguished Chair in Diagnostic Imaging Fund, MD Anderson Brain Tumor Center Program, MD Anderson Cancer Center startup funding, and the Cancer Prevention and Research Institute of Texas Individual Investigator Research Award (RP160150).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Kotrotsou, A., Elakkad, A., Sun, J. et al. Multi-center study finds postoperative residual non-enhancing component of glioblastoma as a new determinant of patient outcome. J Neurooncol 139, 125–133 (2018). https://doi.org/10.1007/s11060-018-2850-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-2850-4