Abstract
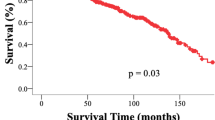
The World Health Organization (WHO) classification of tumors of the central nervous system (CNS) was recently updated, restructuring solitary fibrous tumor (SFT) and hemangiopericytoma (HPC) into one combined entity. This is the first population-based study to examine outcomes of SFT/HPC based on the new WHO guidelines. The Surveillance, Epidemiology, and End Results (SEER) database (1998–2013) was queried to examine age-adjusted incidence and prognostic factors associated with overall survival in 416 surgically resected cases. Age-adjusted incidence was calculated to be 3.77 per 10,000,000 and was rising. Median survival was 155 months, with 5- and 10-year survival rates of 78 and 61%, respectively. Younger age, Asian/Pacific Islander versus white race, benign histology, tumor location, gross-total resection (GTR), and GTR plus radiation (RT) versus subtotal resection were significantly associated with survival. In multivariable analysis, older age (HR = 1.038, p < 0.0001), infratentorial location (HR = 2.019, p = 0.038), GTR (HR = 0.313, p = 0.041), and GTR + RT (HR = 0.215, p = 0.008) were independent prognostic factors. In the HPC and borderline/malignant subgroups, GTR + RT was associated with significantly increased survival compared with GTR alone (HR = 0.537, p = 0.039 and HR = 0.525, p = 0.038). After eliminating patients that died within 3 months of diagnosis, GTR + RT was still associated with an incremental increase in survival (HR = 0.238, p = 0.031) over GTR alone (HR = 0.280, p = 0.054). GTR + RT may be optimal in the management CNS HPC and SFT/HPC tumors with borderline/malignant features. This study, in combination with existing literature, warrants further investigation of adjuvant radiation through a prospective clinical trial.


Similar content being viewed by others
References
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820. https://doi.org/10.1007/s00401-016-1545-1
Robinson DR, Wu Y-M, Kalyana-Sundaram S, Cao X, Lonigro RJ, Sung Y-S, Chen C-L, Zhang L, Wang R, Su F, Iyer MK, Roychowdhury S, Siddiqui J, Pienta KJ, Kunju LP, Talpaz M, Mosquera JM, Singer S, Schuetze SM, Antonescu CR, Chinnaiyan AM (2013) Identification of recurrent NAB2-STAT6 gene fusions in solitary fibrous tumor by integrative sequencing. Nat Genet 45: 180–185. http://www.nature.com/ng/journal/v45/n2/abs/ng.2509.html-supplementary-information
Chmielecki J, Crago AM, Rosenberg M, O’Connor R, Walker SR, Ambrogio L, Auclair D, McKenna A, Heinrich MC, Frank DA, Meyerson M (2013) Whole-exome sequencing identifies a recurrent NAB2-STAT6 fusion in solitary fibrous tumors. Nat Genet 45: 131–132. http://www.nature.com/ng/journal/v45/n2/abs/ng.2522.html-supplementary-information
Schweizer L, Koelsche C, Sahm F, Piro RM, Capper D, Reuss DE, Pusch S, Habel A, Meyer J, Göck T, Jones DTW, Mawrin C, Schittenhelm J, Becker A, Heim S, Simon M, Herold-Mende C, Mechtersheimer G, Paulus W, König R, Wiestler OD, Pfister SM, von Deimling A (2013) Meningeal hemangiopericytoma and solitary fibrous tumors carry the NAB2-STAT6 fusion and can be diagnosed by nuclear expression of STAT6 protein. Acta Neuropathol 125:651–658. https://doi.org/10.1007/s00401-013-1117-6
Bouvier C, Metellus P, de Paula AM, Vasiljevic A, Jouvet A, Guyotat J, Mokhtari K, Varlet P, Dufour H, Figarella-Branger D (2012) Solitary fibrous tumors and hemangiopericytomas of the meninges: overlapping pathological features and common prognostic factors suggest the same spectrum of tumors. Brain Pathol (Zurich) 22:511–521. https://doi.org/10.1111/j.1750-3639.2011.00552.x
Macagno N, Figarella-Branger D, Mokthari K, Metellus P, Jouvet A, Vasiljevic A, Loundou A, Bouvier C (2016) Differential diagnosis of meningeal SFT-HPC and meningioma: which immunohistochemical markers should be used? Am J Surg Pathol 40:270–278. https://doi.org/10.1097/pas.0000000000000526
Fritchie KJ, Jin L, Rubin BP, Burger PC, Jenkins SM, Barthelmess S, Moskalev EA, Haller F, Oliveira AM, Giannini C (2016) NAB2-STAT6 gene fusion in meningeal hemangiopericytoma and solitary fibrous tumor. J Neuropathol Exp Neurol 75:263–271. https://doi.org/10.1093/jnen/nlv026
Savary C, Rousselet MC, Michalak S, Fournier HD, Taris M, Loussouarn D, Rousseau A (2016) [Solitary fibrous tumors and hemangiopericytomas of the meninges: Immunophenotype and histoprognosis in a series of 17 cases]. Ann Pathol 36:258–267. https://doi.org/10.1016/j.annpat.2016.06.002
Fletcher CDM (2006) The evolving classification of soft tissue tumours: an update based on the new WHO classification. Histopathology 48:3–12. https://doi.org/10.1111/j.1365-2559.2005.02284.x
Yalcin CE, Tihan T (2016) Solitary fibrous tumor/hemangiopericytoma dichotomy revisited: a restless family of neoplasms in the CNS. Adv Anat Pathol 23:104–111. https://doi.org/10.1097/pap.0000000000000103
Bisceglia M, Galliani C, Giannatempo G, Lauriola W, Bianco M, D’Angelo V, Pizzolitto S, Vita G, Pasquinelli G, Magro G, Dor DB (2011) Solitary fibrous tumor of the central nervous system: a 15-year literature survey of 220 cases (August 1996–July 2011). Adv Anat Pathol 18:356–392. https://doi.org/10.1097/PAP.0b013e318229c004
Zeng L, Wang Y, Wang Y, Han L, Niu H, Zhang M, Ke C, Chen J, Lei T (2016) Analyses of prognosis-related factors of intracranial solitary fibrous tumors and hemangiopericytomas help understand the relationship between the two sorts of tumors. J Neuro-Oncol. https://doi.org/10.1007/s11060-016-2282-y
Tihan T, Viglione M, Rosenblum MK, Olivi A, Burger PC (2003) Solitary fibrous tumors in the central nervous system. A clinicopathologic review of 18 cases and comparison to meningeal hemangiopericytomas. Arch Pathol Lab Med 127:432–439. https://doi.org/10.1043/0003-9985(2003)127003C0432:sftitc003E2.0.co;2
Mena H, Ribas JL, Pezeshkpour GH, Cowan DN, Parisi JE (1991) Hemangiopericytoma of the central nervous system: a review of 94 cases. Hum Pathol 22:84–91
Kinslow CJ, Rajpara RS, Wu C-C, Bruce SS, Canoll PD, Wang S-H, Sonabend AM, Sheth SA, McKhann GM, Sisti MB, Bruce JN, Wang TJC (2017) Invasiveness is associated with metastasis and decreased survival in hemangiopericytoma of the central nervous system. J Neuro-Oncol. https://doi.org/10.1007/s11060-017-2450-8
Guthrie BL, Ebersold MJ, Scheithauer BW, Shaw EG (1989) Meningeal hemangiopericytoma: histopathological features, treatment, and long-term follow-up of 44 cases. Neurosurgery 25:514–522
Rutkowski MJ, Jian BJ, Bloch O, Chen C, Sughrue ME, Tihan T, Barani IJ, Berger MS, McDermott MW, Parsa AT (2012) Intracranial hemangiopericytoma: clinical experience and treatment considerations in a modern series of 40 adult patients. Cancer 118:1628–1636. https://doi.org/10.1002/cncr.26411
Rutkowski MJ, Bloch O, Jian BJ, Chen C, Sughrue ME, Tihan T, Barani IJ, Berger MS, McDermott MW, Parsa AT (2011) Management of recurrent intracranial hemangiopericytoma. J Clin Neurosci 18:1500–1504. https://doi.org/10.1016/j.jocn.2011.04.009
Ambrosini-Spaltro A, Eusebi V (2010) Meningeal hemangiopericytomas and hemangiopericytoma/solitary fibrous tumors of extracranial soft tissues: a comparison. Virchows Archiv Int J Pathol 456:343–354. https://doi.org/10.1007/s00428-010-0888-6
Mathieu D (2016) Why do hemangiopericytomas have such high recurrence rates? Expert Rev Anticancer Ther 16:1095–1096. https://doi.org/10.1080/14737140.2016.1237289
Bastin KT, Mehta MP (1992) Meningeal hemangiopericytoma: defining the role for radiation therapy. J Neurooncol 14:277–287
Schiariti M, Goetz P, El-Maghraby H, Tailor J, Kitchen N (2011) Hemangiopericytoma: long-term outcome revisited. J Neurosurg 114:747–755. https://doi.org/10.3171/2010.6.JNS091660
Ghose A, Guha G, Kundu R, Tew J, Chaudhary R (2014) CNS hemangiopericytoma: a systematic review of 523 patients. Am J Clin Oncol. https://doi.org/10.1097/coc.0000000000000146
Carneiro SS, Scheithauer BW, Nascimento AG, Hirose T, Davis DH (1996) Solitary fibrous tumor of the meninges: a lesion distinct from fibrous meningioma. A clinicopathologic and immunohistochemical study. Am J Clin Oncol 106:217–224
Stout AP, Murray MR (1942) Hemangiopericytoma: a vascular tumor featuring Zimmermann’s pericytes. Ann Surg 116:26–33
Kleihues P, Burger PC, Scheithauer BW (1993) The new WHO classification of brain tumours. Brain Pathol (Zurich) 3:255–268
Greenlee RT, Goodman MT, Lynch CF, Platz CE, Havener LA, Howe HL (2010) The occurrence of rare cancers in United States adults, 1995–2004. Public Health Rep 125: 28–43 (Washington, DC: 1974)
Bilimoria KY, Stewart AK, Winchester DP, Ko CY (2008) The national cancer data base: A powerful initiative to improve cancer care in the United States. Ann Surg Oncol 15:683–690. https://doi.org/10.1245/s10434-007-9747-3
Warren JL, Harlan LC (2003) Can cancer registry data be used to study cancer treatment? Med Care 41:1003–1005. https://doi.org/10.1097/01.mlr.0000086827.00805.b5
(released April 2016, based on the November 2015 submission) Surveillance, Epidemiology,and End Results (SEER) Program (http://www.seer.cancer.gov) SEER*Stat Database: Incidence- SEER 9 Regs Research Data, Nov 2015 Sub (1973–2013) < Katrina/Rita Population Adjustment>- Linked To County Attributes - Total U.S., 1969–2014 Counties.
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114:97–109. https://doi.org/10.1007/s00401-007-0243-4
Ghia AJ, Allen PK, Mahajan A, Penas-Prado M, McCutcheon IE, Brown PD (2013) Intracranial hemangiopericytoma and the role of radiation therapy: a population based analysis. Neurosurgery 72:203–209. https://doi.org/10.1227/NEU.0b013e31827b9e68
Sonabend AM, Zacharia BE, Goldstein H, Bruce SS, Hershman D, Neugut AI, Bruce JN (2014) The role for adjuvant radiotherapy in the treatment of hemangiopericytoma: a Surveillance, Epidemiology, and End Results analysis. J Neurosurg 120:300–308. https://doi.org/10.3171/2013.10.jns13113
Stessin AM, Sison C, Nieto J, Raifu M, Li B (2013) The role of postoperative radiation therapy in the treatment of meningeal hemangiopericytoma—Experience from the SEER database. Int J Radiat Oncol Biol Phys 85:784–790. https://doi.org/10.1016/j.ijrobp.2012.05.042
Noone AM, Lund JL, Mariotto A, Cronin K, McNeel T, Deapen D, Warren JL (2016) Comparison of SEER treatment data with medicare claims. Med Care 54:e55–e64. https://doi.org/10.1097/mlr.0000000000000073
Rutkowski MJ, Sughrue ME, Kane AJ, Aranda D, Mills SA, Barani IJ, Parsa AT (2010) Predictors of mortality following treatment of intracranial hemangiopericytoma. J Neurosurg 113:333–339. https://doi.org/10.3171/2010.3.jns091882
Soyuer S, Chang EL, Selek U, McCutcheon IE, Maor MH (2004) Intracranial meningeal hemangiopericytoma: the role of radiotherapy. Cancer 100:1491–1497. https://doi.org/10.1002/cncr.20109
Ramakrishna R, Rostomily R, Sekhar L, Rockhill J, Ferreira M (2014) Hemangiopericytoma: radical resection remains the cornerstone of therapy. J Clin Neurosci 21:612–615. https://doi.org/10.1016/j.jocn.2013.08.006
Kim JH, Jung HW, Kim YS, Kim CJ, Hwang SK, Paek SH, Kim DG, Kwun BD (2003) Meningeal hemangiopericytomas: long-term outcome and biological behavior. Surg Neurol 59:47–53 (discussion 53–44)
Lee EJ, Kim JH, Park ES, Khang SK, Cho YH, Hong SH, Kim CJ (2016) The impact of postoperative radiation therapy on patterns of failure and survival improvement in patients with intracranial hemangiopericytoma. J Neuro-Oncol 127:181–190. https://doi.org/10.1007/s11060-015-2030-8
Chen L-f, Yang Y, Yu X-g, Gui Q-p, Xu B-n, Zhou D-b (2015) Multimodal treatment and management strategies for intracranial hemangiopericytoma. J Clin Neurosci 22:718–725. https://doi.org/10.1016/j.jocn.2014.11.011
Choi J, Park S-H, Khang SK, Suh Y-L, Kim SP, Lee YS, Kwon HS, Kang S-G, Kim SH (2016) Hemangiopericytomas in the central nervous system: a Multicenter Study of Korean Cases with validation of the usage of STAT6 immunohistochemistry for diagnosis of disease. Ann Surg Oncol. https://doi.org/10.1245/s10434-016-5414-x
Zhang GJ, Wu Z, Zhang LW, Li D, Zhang JT (2017) Surgical management and adverse factors for recurrence and long-term survival in hemangiopericytoma patients. World Neurosurg. https://doi.org/10.1016/j.wneu.2017.05.010
Staples JJ, Robinson RA, Wen BC, Hussey DH (1990) Hemangiopericytoma—The role of radiotherapy. Int J Radiat Oncol Biol Phys 19:445–451. https://doi.org/10.1089/ten.2005.11.985
Ghia AJ, Chang EL, Allen PK, Mahajan A, Penas-Prado M, McCutcheon IE, Brown PD (2013) Intracranial hemangiopericytoma: patterns of failure and the role of radiation therapy. Neurosurgery 73:624–630. https://doi.org/10.1227/neu.0000000000000064 (discussion 630–621)
Zhu H, Duran D, Hua L, Tang H, Chen H, Zhong P, Zheng K, Wang Y, Che X, Bao W, Wang Y, Xie Q, Gong Y (2016) Prognostic factors in patients with primary hemangiopericytomas of the central nervous system: a series of 103 cases at a single institution. World Neurosurg 90:414–419. https://doi.org/10.1016/j.wneu.2016.02.103
Dufour H, Metellus P, Fuentes S, Murracciole X, Regis J, Figarella-Branger D, Grisoli F (2001) Meningeal hemangiopericytoma: a retrospective study of 21 patients with special review of postoperative external radiotherapy. Neurosurgery 48:756–762 (discussion 762–753)
Caldarella A, Crocetti E, Paci E (2011) Is the incidence of brain tumors really increasing? A population-based analysis from a cancer registry. J Neuro-Oncol 104:589–594. https://doi.org/10.1007/s11060-011-0533-5
Suissa S (2008) Immortal time bias in pharmacoepidemiology. Am J Epidemiol 167:492–499. https://doi.org/10.1093/aje/kwm324
Acknowledgements
We would like to thank the developers of the Surveillance, Epidemiology, and End Results program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Tony J. C. Wang reports personal fees and nonfinancial support from AbbVie, nonfinancial support from Merck, personal fees from AstraZeneca, personal fees from Doximity, nonfinancial support from Novocure, personal fees and nonfinancial support from Elekta and personal fees from Wolters Kluwer, outside the submitted work. All other authors report no financial conflicts of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kinslow, C.J., Bruce, S.S., Rae, A.I. et al. Solitary-fibrous tumor/hemangiopericytoma of the central nervous system: a population-based study. J Neurooncol 138, 173–182 (2018). https://doi.org/10.1007/s11060-018-2787-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-2787-7