Abstract
Purpose
This study aims to identify the neuropsychological tests commonly used for assessment in each neurocognitive domain, and quantify the post-operative changes in neurocognitive function in the immediate post-operation and follow-up.
Methods
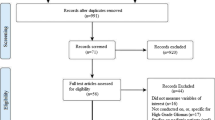
With the use of the PubMed, a comprehensive search of the English literature was performed following PRISMA (Preferred Reporting Items for Systematic reviews and Meta-Analyses) guidelines. There were 1021 publications identified for screening. Standardized mean differences (SMD) in neuropsychological task performance were calculated both for immediate post-operation (up to 1 week) and follow-up (up to 6 months).
Results
Out of 12 studies which met the inclusion criteria, 11 studies were analyzed in this meta-analysis, with a total of 313 patients (age range 18–82, 50% males) with intracranial gliomas (45% high-grade, 55% low-grade). Complex attention, language and executive function were the most frequently tested neurocognitive domains. Surgery had a positive impact in the domains of complex attention, language, learning and memory tasks in the immediate post-operative period and sustained improvement at follow-up. In contrast, surgery was found to negatively impact performance for executive function in the immediate post-operative period with sustained decline in performance in the long term.
Conclusions
This meta-analysis suggests that surgery for glioma confers a benefit for the domains of complex attention, language, learning and memory, while negatively affecting executive function, in the periods immediately after surgery and at 6 months follow-up. In addition, awake surgery seemed to confer a beneficial effect on neurocognitive functions. Future research should attempt to standardize a battery of neuropsychological tests for patients undergoing surgical resection for glioma, perhaps with a particular focus on executive function.





Similar content being viewed by others
References
Jakola AS, Skjulsvik AJ, Myrmel KS, Sjavik K, Unsgard G, Torp SH, Aaberg K, Berg T, Dai HY, Johnsen K, Kloster R, Solheim O (2017) Surgical resection versus watchful waiting in low-grade gliomas. Ann Oncol 28(8):1942–1948. https://doi.org/10.1093/annonc/mdx230
Brown TJ, Bota DA, Maher EA, Aregawi DG, Liau LM, Brown PD, Buckner JC, Weller M, Bent MJVD, Berger MS, Glantz MJ (2017) Association of aggressive resection with survival and progression-free survival in adult low-grade glioma: A systematic review and meta-analysis with numbers needed to treat. J Clin Oncol 35(15_suppl):2025–2025. https://doi.org/10.1200/JCO.2017.35.15_suppl.2025
Bunevicius A, Tamasauskas S, Deltuva V, Tamasauskas A, Radziunas A, Bunevicius R (2014) Predictors of health-related quality of life in neurosurgical brain tumor patients: focus on patient-centered perspective. Acta Neurochirurgica 156(2):367–374. https://doi.org/10.1007/s00701-013-1930-7
Zucchella C, Bartolo M, Di Lorenzo C, Villani V, Pace A (2013) Cognitive impairment in primary brain tumors outpatients: a prospective cross-sectional survey. Journal of Neuro Oncol 112(3):455–460. https://doi.org/10.1007/s11060-013-1076-8
Hentschel SJ, Lang FF (2005) Surgical resection of intrinsic insular tumors. Neurosurgery 57(1 Suppl):176–183; discussion 176–183
Lang FF, Olansen NE, DeMonte F, Gokaslan ZL, Holland EC, Kalhorn C, Sawaya R (2001) Surgical resection of intrinsic insular tumors: complication avoidance. J Neurosurg 95(4):638–650. https://doi.org/10.3171/jns.2001.95.4.0638
Neuloh G, Pechstein U, Schramm J (2007) Motor tract monitoring during insular glioma surgery. J Neurosurg 106(4):582–592. https://doi.org/10.3171/jns.2007.106.4.582
Sanai N, Polley MY, Berger MS (2010) Insular glioma resection: assessment of patient morbidity, survival, and tumor progression. J Neurosurg 112(1):1–9. https://doi.org/10.3171/2009.6.jns0952
Nasreddine ZS, Phillips NA, Bedirian V, Charbonneau S, Whitehead V, Collin I, Cummings JL, Chertkow H (2005) The montreal cognitive assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc 53(4):695–699. https://doi.org/10.1111/j.1532-5415.2005.53221.x
Olson RA, Iverson GL, Carolan H, Parkinson M, Brooks BL, McKenzie M (2011) Prospective comparison of two cognitive screening tests: diagnostic accuracy and correlation with community integration and quality of life. J Neuro Oncol 105(2):337–344. https://doi.org/10.1007/s11060-011-0595-4
Satoer D, Visch-Brink E, Dirven C, Vincent A (2016) Glioma surgery in eloquent areas: can we preserve cognition? Acta Neurochirurgica 158(1):35–50. https://doi.org/10.1007/s00701-015-2601-7
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Ann Intern Med 151(4):264–269, w264
Janulewicz PA, Krengel MH, Maule A, White RF, Cirillo J, Sisson E, Heeren T, Sullivan K (2017) Neuropsychological characteristics of Gulf War illness: a meta-analysis. PloS ONE 12(5):e0177121. https://doi.org/10.1371/journal.pone.0177121
Cohen J (1988) Statistical power analysis for the behavioral sciences. 2nd edn., New York University, New York
Abrams KR, Gillies CL, Lambert PC (2005) Meta-analysis of heterogeneously reported trials assessing change from baseline. Stat Med 24(24):3823–3844. https://doi.org/10.1002/sim.2423
Borenstein M, Hedges LV, Higgins JP, Rothstein HR (2011) Introduction to meta-analysis. Wiley, Hoboken
Higgins JP, Green S (2011) Cochrane handbook for systematic reviews of interventions, vol 4. Wiley, Hoboken
Higgins JP, Thompson SG, Deeks JJ, Altman DG (2003) Measuring inconsistency in meta-analyses. BMJ 327(7414):557–560. https://doi.org/10.1136/bmj.327.7414.557
Dickersin K, Berlin JA (1992) Meta-analysis: state-of-the-science. Epidemiol Rev 14:154–176
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21(11):1539–1558. https://doi.org/10.1002/sim.1186
Lu J, Wu J, Yao C, Zhuang D, Qiu T, Hu X, Zhang J, Gong X, Liang W, Mao Y, Zhou L (2013) Awake language mapping and 3-Tesla intraoperative MRI-guided volumetric resection for gliomas in language areas. J Clin Neurosci 20(9):1280–1287. https://doi.org/10.1016/j.jocn.2012.10.042
Bello L, Gallucci M, Fava M, Carrabba G, Giussani C, Acerbi F, Baratta P, Songa V, Conte V, Branca V, Stocchetti N, Papagno C, Gaini SM (2007) Intraoperative subcortical language tract mapping guides surgical removal of gliomas involving speech areas. Neurosurgery 60(1):67–80. https://doi.org/10.1227/01.NEU.0000249206.58601.DE discussion 80–62.
Braun V, Albrecht A, Kretschmer T, Richter HP, Wunderlich A (2006) Brain tumour surgery in the vicinity of short-term memory representation–results of neuronavigation using fMRI images. Acta Neurochirurgica 148(7):733–739. https://doi.org/10.1007/s00701-005-0668-2
Bryszewski B, Tybor K, Ormezowska EA, Jaskolski DJ, Majos A (2013) Rearrangement of motor centers and its relationship to the neurological status of low-grade glioma examined on pre- and postoperative fMRI. Clin Neurol Neurosurg 115(12):2464–2470. https://doi.org/10.1016/j.clineuro.2013.09.034
Charras P, Herbet G, Deverdun J, de Champfleur NM, Duffau H, Bartolomeo P, Bonnetblanc F (2015) Functional reorganization of the attentional networks in low-grade glioma patients: a longitudinal study. Cortex 63:27–41. https://doi.org/10.1016/j.cortex.2014.08.010
Herbet G, Lafargue G, Bonnetblanc F, Moritz-Gasser S, Duffau H (2013) Is the right frontal cortex really crucial in the mentalizing network? A longitudinal study in patients with a slow-growing lesion. Cortex 49(10):2711–2727. https://doi.org/10.1016/j.cortex.2013.08.003
Santini B, Talacchi A, Squintani G, Casagrande F, Capasso R, Miceli G (2012) Cognitive outcome after awake surgery for tumors in language areas. J Neuro Oncol 108(2):319–326. https://doi.org/10.1007/s11060-012-0817-4
Talacchi A, Santini B, Savazzi S, Gerosa M (2011) Cognitive effects of tumour and surgical treatment in glioma patients. J Neuro Oncol 103(3):541–549. https://doi.org/10.1007/s11060-010-0417-0
Teixidor P, Gatignol P, Leroy M, Masuet-Aumatell C, Capelle L, Duffau H (2007) Assessment of verbal working memory before and after surgery for low-grade glioma. J Neuro Oncol 81(3):305–313. https://doi.org/10.1007/s11060-006-9233-y
Whittle IR, Pringle A-M, Taylor R (1998) Effects of resective surgery for left-sided intracranial tumours on language function: a prospective study. Lancet 351(9108):1014–1018. https://doi.org/10.1016/s0140-6736(97)08295-0
Wolf J, Campos B, Bruckner T, Vogt L, Unterberg A, Ahmadi R (2016) Evaluation of neuropsychological outcome and “quality of life” after glioma surgery. Langenbecks Arch Surg 401(4):541–549. https://doi.org/10.1007/s00423-016-1403-6
Papagno C, Miracapillo C, Casarotti A, Romero Lauro LJ, Castellano A, Falini A, Casaceli G, Fava E, Bello L (2011) What is the role of the uncinate fasciculus? Surgical removal and proper name retrieval. Brain 134(Pt 2):405–414. https://doi.org/10.1093/brain/awq283
Barzilai O, Ben Moshe S, Sitt R, Sela G, Shofty B, Ram Z (2018) Improvement in cognitive function after surgery for low-grade glioma. J Neurosurg 23:1–9
Duffau H, Capelle L, Denvil D, Sichez N, Gatignol P, Taillandier L, Lopes M, Mitchell MC, Roche S, Muller JC, Bitar A (2003) Usefulness of intraoperative electrical subcortical mapping during surgery for low-grade gliomas located within eloquent brain regions: functional results in a consecutive series of 103 patients. J Neurosurg 98(4):764–778. https://doi.org/10.3171/jns.2003.98.4.0764
Duffau H, Gatignol P, Mandonnet E, Capelle L, Taillandier L (2008) Intraoperative subcortical stimulation mapping of language pathways in a consecutive series of 115 patients with Grade II glioma in the left dominant hemisphere. J Neurosurg 109(3):461–471. https://doi.org/10.3171/JNS/2008/109/9/0461
Scheibel RS, Meyers CA, Levin VA (1996) Cognitive dysfunction following surgery for intracerebral glioma: influence of histopathology, lesion location, and treatment. J Neurooncol 30(1):61–69
Campanella F, Fabbro F, Ius T, Shallice T, Skrap M (2015) Acute effects of surgery on emotion and personality of brain tumor patients: surgery impact, histological aspects, and recovery. Neuro Oncol 17(8):1121–1131
Schiffbauer H, Ferrari P, Rowley HA, Berger MS, Roberts TP (2001) Functional activity within brain tumors: a magnetic source imaging study. Neurosurgery 49(6):1313–1321
Atlas SW, Howard RSII, Maldjian J, Alsop D, Detre JA, Listerud J, D’Esposito M, Judy KD, Zager E, Stecker M (1996) Functional magnetic resonance imaging of regional brain activity in patients with intracerebral gliomas: findings and implications for clinical management. Neurosurgery 38(2):329–338
Desmurget M, Bonnetblanc F, Duffau H (2007) Contrasting acute and slow-growing lesions: a new door to brain plasticity. Brain 130(4):898–914. https://doi.org/10.1093/brain/awl300
Bosma I, Vos MJ, Heimans JJ, Taphoorn MJ, Aaronson NK, Postma TJ, van der Ploeg HM, Muller M, Vandertop WP, Slotman BJ, Klein M (2007) The course of neurocognitive functioning in high-grade glioma patients. Neuro Oncol 9(1):53–62. https://doi.org/10.1215/15228517-2006-012
Kayl AE, Meyers CA (2003) Does brain tumor histology influence cognitive function? Neuro Oncol 5(4):255–260. https://doi.org/10.1215/S1152851703000012
Satoer D, Vork J, Visch-Brink E, Smits M, Dirven C, Vincent A (2012) Cognitive functioning early after surgery of gliomas in eloquent areas. J Neurosurg 117(5):831–838. https://doi.org/10.3171/2012.7.JNS12263
De Witt Hamer PC1, Robles SG, Zwinderman AH, Duffau H, Berger MS (2012) Impact of intraoperative stimulation brain mapping on glioma surgery outcome: a meta-analysis. J Clin Oncol 30(20):2559–2565. https://doi.org/10.1200/JCO.2011.38.4818
Lu VM, Phan K, Rovin RA (2018) Comparison of operative outcomes of eloquent glioma resection performed under awake versus general anesthesia: a systematic review and meta-analysis. Clin Neurol Neurosurg 169:121–127. https://doi.org/10.1016/j.clineuro.2018.04.011
Talacchi A, Santini B, Casartelli M, Monti A, Capasso R, Miceli G (2013) Awake surgery between art and science. part II: language and cognitive mapping. Funct Neurol 28(3):223–239. https://doi.org/10.11138/FNeur/2013.28.3.223
Wager M, Du Boisgueheneuc F, Pluchon C, Bouyer C, Stal V, Bataille B, Guillevin CM, Gil R (2013) Intraoperative monitoring of an aspect of executive functions. Oper Neurosurg 72:ons169–ons180. https://doi.org/10.1227/NEU.0b013e31827bf1d6 (2 Suppl Operative)
Norman DA, Shallice T (1986) Attention to Action. In: Davidson RJ, Schwartz GE, Shapiro D (eds) Consciousness and self-regulation: advances in research and theory volume 4. Springer, Boston, pp 1–18. https://doi.org/10.1007/978-1-4757-0629-1_1
Kaleita TA, Wellisch DK, Cloughesy TF, Ford JM, Freeman D, Belin TR, Goldman J (2004) Prediction of neurocognitive outcome in adult brain tumor patients. J Neuro Oncol 67(1–2):245–253
Taphoorn MJ, Klein M (2004) Cognitive deficits in adult patients with brain tumours. Lancet Neurol 3(3):159–168. https://doi.org/10.1016/s1474-4422(04)00680-5
Sarubbo S, Latini F, Panajia A, Candela C, Quatrale R, Milani P, Fainardi E, Granieri E, Trapella G, Tugnoli V, Cavallo MA (2011) Awake surgery in low-grade gliomas harboring eloquent areas: 3-year mean follow-up. Neurol Sci 32(5):801–810. https://doi.org/10.1007/s10072-011-0587-3
Gehring K, Sitskoorn MM, Gundy CM, Sikkes SA, Klein M, Postma TJ, van den Bent MJ, Beute GN, Enting RH, Kappelle AC, Boogerd W (2009) Cognitive rehabilitation in patients with gliomas: a randomized, controlled trial. J Clin Oncol 27(22):3712–3722. https://doi.org/10.1200/JCO.2008.20.5765
Zucchella C, Capone A, Codella V, De Nunzio AM, Vecchione C, Sandrini G, Pace A, Pierelli F, Bartolo M (2013) Cognitive rehabilitation for early post-surgery inpatients affected by primary brain tumor: a randomized, controlled trial. J Neurooncol 114(1):93–100. https://doi.org/10.1007/s11060-013-1153-z
Funding
This research is supported by Singapore Ministry of Health’s National Medical Research Council under its Translational and Clinical Research Flagship Programme - Tier 1 (Project No: NMRC/TCR/016-NNI/2016).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors. For this type of study formal consent is not required.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ng, J.C.H., See, A.A.Q., Ang, T.Y. et al. Effects of surgery on neurocognitive function in patients with glioma: a meta-analysis of immediate post-operative and long-term follow-up neurocognitive outcomes. J Neurooncol 141, 167–182 (2019). https://doi.org/10.1007/s11060-018-03023-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-018-03023-9