Abstract
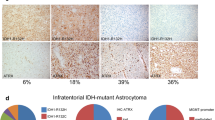
Glioneuronal tumor with neuropil-like islands (GTNI) is a rare, recently described neoplasm, whose pathogenesis has not been studied extensively. The role of ATRX mutations, a class-defining alteration in diffuse astrocytic neoplasms, has not been assessed in GTNIs previously. We therefore aimed to assess the status of ATRX, along with IDH1, 1p/19q and p53, in cases of GTNI in order to evaluate the molecular profile of these tumors. All cases of GTNI diagnosed at our Institute were retrieved and clinicopathological features were reviewed. Immunohistochemistry for ATRX, IDH1 and p53 was performed. We identified four cases of GTNI, majority of which occurred in young adults. Loss of ATRX immunoexpression, a surrogate marker for ATRX mutation, was seen in all four cases. All cases were immunopositive for p53, while IDH1 positivity was seen in all three cases assessed. 1p/19q codeletion was absent in the three cases analyzed. These results indicate that the molecular pathogenesis of GTNIs similar to that of diffuse astrocytic tumors. Further, the loss of ATRX expression is seen in both the glial as well as neuronal components, indicating that both arise from the same tumor stem/progenitor cell and that the latter may be a metaplastic change. Thus, loss of ATRX immunoexpression, shown for the first time in these tumors, along with immunopositivity for p53 and IDH1, indicates that these tumors are molecular astrocytomas, and their clinical behaviour is likely to recapitulate that of ATRX-mutant and IDH-mutant diffuse astrocytomas of the same grade.




Similar content being viewed by others
References
Agarwal S, Suri V, Rishi A, Shukla B, Garg A, Sharma MC, Sinha S, Sarkar C (2009) Glioneuronal tumor with neuropil-like islands: a new entity. Neuropathology 29:96–100
Prayson RA, Abramovich CM (2000) Glioneuronal tumor with neuropil-like islands. Hum Pathol 31:1435–1438
Schlamann A, von Bueren AO, Hagel C, Zwiener I, Seidel C, Kortmann RD, Müller K (2014) An individual patient data meta-analysis on characteristics and outcome of patients with papillary glioneuronal tumor, rosette glioneuronal tumor with neuropil-like islands and rosette forming glioneuronal tumor of the fourth ventricle. PLoS One 9:e101211
Barbashina V, Salazar P, Ladanyi M, Rosenblum MK, Edgar MA (2007) Glioneuronal tumor with neuropil-like islands (GTNI): a report of 8 cases with chromosome 1p/19q deletion analysis. Am J Surg Pathol 31:1196–1202
Huse JT, Nafa K, Shukla N, Kastenhuber ER, Lavi E, Hedvat CV, Ladanyi M, Rosenblum MK (2011) High frequency of IDH-1 mutation links glioneuronal tumors with neuropil-like islands to diffuse astrocytomas. Acta Neuropathol 122:367–369
Jha P, Sarkar C, Pathak P, Sharma MC, Kale SS, Gupta D, Chosdol K, Suri V (2011) Detection of allelic status of 1p and 19q by microsatellite-based PCR versus FISH: limitations and advantages in application to patient management. Diagn Mol Pathol 20(1):40–47
Jha P, Suri V, Sharma V, Singh G, Sharma MC, Pathak P, Chosdol K, Jha P, Suri A, Mahapatra AK, Kale SS, Sarkar C (2011) IDH1 mutations in gliomas: first series from a tertiary care centre in India with comprehensive review of literature. Exp Mol Pathol 91(1):385–393
Teo JG, Gultekin SH, Bilsky M, Gutin P, Rosenblum MK (1999) A distinctive glioneuronal tumor of the adult cerebrum with neuropil-like (including “rosetted”) islands: report of 4 cases. Am J Surg Pathol 23:502–510
Buccoliero AM, Castiglione F, Degl’innocenti DR, Moncini D, Paglierani M, Sardi I, Giunti L, Giordano F, Sanzo M, Mussa F, Aricò M, Genitori L, Taddei GL (2012) Glioneuronal tumor with neuropil-like islands: clinical, morphologic, immunohistochemical, and molecular features of three pediatric cases. Pediatr Dev Pathol 15:352–360
Chesser JD, Friedman NR, Prayson RA (2016) Congenital glioneuronal tumor with neuropil-like islands. J Clin Neurosci 24:156–157
Comunoglu N, Kilickesmez O, Oz B (2014) Spinal cord glioneuronal tumor with rosette neuropil-like islands in pediatric age group. Case Rep Pathol 2014:471645
Keyvani K, Rickert CH, von Wild K, Paulus W (2001) Rosetted glioneuronal tumor: a case with proliferating neuronal nodules. Acta Neuropathol 101:525–528
Vajtai I, Reinert MM (2007) Malignant glioneuronal tumor of the adult cerebrum with neuropil-like islands involving “proliferating nodules”: confirmatory report of an unusual variant. Acta Neuropathol 113:711–713
Ishizawa K, Hirose T, Sugiyama K, Kageji T, Nobusawa S, Homma T, Komori T, Sasaki A (2012) Pathologic diversity of glioneuronal tumor with neuropil-like islands: a histological and immunohistochemical study with a special reference to isocitrate dehydrogenase 1 (IDH1) in 5 cases. Clin Neuropathol 31:67–76
Serra SM, Dabdoub CB, da Cunha AH, Salazar B, Lima TP, Azevedo-Filho HC (2013) Disseminated glioneuronal tumor with neuropil-like islands of the spinal cord: a distinctive entity. World Neurosurg 80:655.e1–5
Barbashina V, Salazar P, Holland EC, Rosenblum MK, Ladanyi M (2005) Allelic losses at 1p36 and 19q13 in gliomas: correlation with histologic classification, definition of a 150-kb minimal deleted region on 1p36, and evaluation of CAMTA1 as a candidate tumor suppressor gene. Clin Cancer Res 11:1119–1128
Jiao Y, Killela PJ, Reitman ZJ, Rasheed AB, Heaphy CM, de Wilde RF, Rodriguez FJ, Rosemberg S, Oba-Shinjo SM, Nagahashi Marie SK, Bettegowda C, Agrawal N, Lipp E, Pirozzi C, Lopez G, He Y, Friedman H, Friedman AH, Riggins GJ, Holdhoff M, Burger P, McLendon R, Bigner DD, Vogelstein B, Meeker AK, Kinzler KW, Papadopoulos N, Diaz LA, Yan H (2012) Frequent ATRX, CIC, FUBP1 and IDH1 mutations refine the classification of malignant gliomas. Oncotarget 3:709–722
Wiestler B, Capper D, Holland-Letz T, Korshunov A, von Deimling A, Pfister SM, Platten M, Weller M, Wick W (2013) ATRX loss refines the classification of anaplastic gliomas and identifies a subgroup of IDH mutant astrocytic tumors with better prognosis. Acta Neuropathol 126:443–451
Acknowledgments
AK and MCS conceptualized the study. AK, AN, KK, and MCS evaluated the histopathology and immunohistochemistry. AK, MCS and CS reviewed all data and made the final diagnoses. SSK provided the clinical data and was the primary treating surgeon. SM treated the patients following surgery, and provided details of radiotherapy, chemotherapy and followed up the patients. AG provided the radiological data for the patients included. MCS, CS and VS supervised the study and provided material support and resources. AK drafted the manuscript. MCS critically reviewed the manuscript. All authors approved of the final manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kakkar, A., Nambirajan, A., Kaur, K. et al. ATRX loss in glioneuronal tumors with neuropil-like islands indicates similarity to diffuse astrocytic tumors. J Neurooncol 130, 63–68 (2016). https://doi.org/10.1007/s11060-016-2224-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-016-2224-8