Abstract
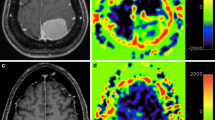
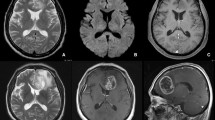
Malignant dural neoplasms are not reliably distinguished from benign dural neoplasms with contrast-enhanced magnetic resonance imaging (MRI). MRI enhancement in central nervous system (CNS) diseases imaged with ferumoxytol has been attributed to intracellular uptake in macrophages rather than vascular leakage. We compared imaging to histopathology and immunohistochemistry in meningiomas and dural metastases having ferumoxytol-enhanced MRI (FeMRI) and gadolinium-enhanced MRI (GdMRI) in order to correlate enhancement patterns to macrophage presence and vascular state. All patients having extraaxial CNS tumors were retrospectively selected from one of two ongoing FeMRI studies. Enhancement was compared between GdMRI and FeMRI. Diagnoses were confirmed histologically and/or by characteristic imaging. Tumor and vascular histology was reviewed. Immunohistochemical staining for CD68 (a macrophage marker), Connexin-43 (Cx43) (a marker of normal gap junctions), and smooth muscle actin (SMA) as a marker of vascularity, was performed in seven study cases with available tissue. Immunohistochemistry was performed on archival material from 33 subjects outside of the current study as controls: 20 WHO grade I cases of meningioma and 13 metastatic tumors. Metastases displayed marked delayed enhancement on FeMRI, similar to GdMRI. Four patients with dural metastases and one patient with meningioma showed similar enhancement on FeMRI and GdMRI. Five meningiomas with typical enhancement on GdMRI lacked enhancement on FeMRI. Enhancement on FeMRI was better associated with decreased Cx43 expression than intralesional macrophages. These pilot data suggest that FeMRI may better differentiate metastatic disease from meningiomas than GdMRI, and that differences in tumor vasculature rather than macrophage presence could underlie differences in contrast enhancement.







Similar content being viewed by others
References
Weinstein JS, Varallyay CG, Dosa E et al (2010) Superparamagnetic iron oxide nanoparticles: diagnostic magnetic resonance imaging and potential therapeutic applications in neurooncology and central nervous system inflammatory pathologies, a review. J Cereb Blood Flow Metab 30:15–35
Dosa E, Tuladhar S, Muldoon LL, Hamilton BE, Rooney WD, Neuwelt EA (2011) MRI using ferumoxytol improves the visualization of central nervous system vascular malformations. Stroke 42:1581–1588
Christ GJ, Spray DC, el-Sabban M, Moore LK, Brink PR (1996) Gap junctions in vascular tissues. Evaluating the role of intercellular communication in the modulation of vasomotor tone. Circ Res 79:631–646
Domingues PH, Teodosio C, Ortiz J et al (2012) Immunophenotypic identification and characterization of tumor cells and infiltrating cell populations in meningiomas. Am J Pathol 181:1749–1761
http://www.dailymed.nlm.nih.gov/dailymed/archives/fdaDrugInfo.cfm?archiveid=9818. Accessed 26 Feb 2016
Maiuri F, Iaconetta G, de Divitiis O, Cirillo S, Di Salle F, De Caro ML (1999) Intracranial meningiomas: correlations between MR imaging and histology. Eur J Radiol 31:69–75
Bo L, Mork SJ, Nyland H (1992) An immunohistochemical study of mononuclear cells in meningiomas. Neuropathol Appl Neurobiol 18:548–558
Morantz RA, Wood GW, Foster M, Clark M, Gollahon K (1979) Macrophages in experimental and human brain tumors. Part 2: studies of the macrophage content of human brain tumors. J Neurosurg 50:305–311
Dejana E (1996) Endothelial adherens junctions: implications in the control of vascular permeability and angiogenesis. J Clin Invest 98:1949–1953
Arishima H, Sato K, Kubota T (2002) Immunohistochemical and ultrastructural study of gap junction proteins connexin26 and 43 in human arachnoid villi and meningeal tumors. J Neuropathol Exp Neurol 61:1048–1055
Long DM (1970) Capillary ultrastructure and the blood–brain barrier in human malignant brain tumors. J Neurosurg 32:127–144
Long DM (1973) Vascular ultrastructure in human meningiomas and schwannomas. J Neurosurg 38:409–419
http://www.fda.gov/Drugs/DrugSafety/ucm440138.htm. Accessed 3 March 2016
Acknowledgments
This work was supported by a Veterans Administration merit review Grant and by National Institute of Health Grants NS44687, and CA137488, and by the Walter S. and Lucienne Driskill Foundation to EAN. The authors thank Samantha Rolph and Emily Hochhalter Youngers for their help with manuscript preparation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Disclosures
Dr. Neuwelt’s studies involving ferumoxytol were entirely funded by the Veterans Administration and NIH research Grants, and the Walter S. and Lucienne Driskill Foundation. None of the authors have financial interests in ferumoxytol, or in its developer AMAG.
Rights and permissions
About this article
Cite this article
Hamilton, B.E., Woltjer, R.L., Prola-Netto, J. et al. Ferumoxytol-enhanced MRI differentiation of meningioma from dural metastases: a pilot study with immunohistochemical observations. J Neurooncol 129, 301–309 (2016). https://doi.org/10.1007/s11060-016-2175-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-016-2175-0