Abstract
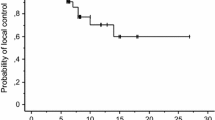
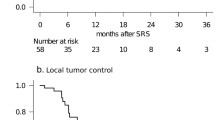
It is not uncommon for brain metastasis (BM) treated with stereotactic radiosurgery (SRS) to demonstrate radiographic enlargement, with the patient developing neurological deficits attributable to a lesion at the site of SRS. The management of both local recurrence and radiation-induced necrosis (RN) poses a significant therapeutic dilemma, if surgical resection is not feasible, and effective therapies have yet to be established. This preliminary study introduces our initial experience with salvage SRS using adjuvant bevacizumab for this refractory entity. We retrospectively reviewed five patients who had received salvage SRS using adjuvant bevacizumab for recurrent BM complicated by RN. The diagnosis was based on clinical features, serial imaging studies and/or histopathological findings. Patients underwent salvage SRS followed by the first cycle of bevacizumab (7.5–10 mg/kg intravenous). Bevacizumab was repeated every 3–4 weeks until tumor progression or significant toxic events. The number of bevacizumab doses ranged from 2 to 16 (median 4). Follow-up MR imaging demonstrated a clear radiographic response in all lesions. Neurological symptoms improved in three patients and stabilized in two. In two patients, bevacizumab treatment was discontinued due to anemia and gastrointestinal bleeding, respectively. At the time of data analysis, four patients had died and the other was still alive. The causes of death were neurological decline and systemic disease progression in two patients each. Salvage SRS with adjuvant bevacizumab use appeared to provide an adequate radiographic response as well as neurological palliation for selected patients with heavily treated recurrent BM complicated by RN.




Similar content being viewed by others
References
Yamamoto M, Serizawa T, Shuto T, Akabane A, Higuchi Y, Kawagishi J, Yamanaka K, Sato Y, Jokura H, Yomo S, Nagano O, Kenai H, Moriki A, Suzuki S, Kida Y, Iwai Y, Hayashi M, Onishi H, Gondo M, Sato M, Akimitsu T, Kubo K, Kikuchi Y, Shibasaki T, Goto T, Takanashi M, Mori Y, Takakura K, Saeki N, Kunieda E, Aoyama H, Momoshima S, Tsuchiya K (2014) Stereotactic radiosurgery for patients with multiple brain metastases (JLGK0901): a multi-institutional prospective observational study. Lancet Oncol 15:387–395. doi:10.1016/S1470-2045(14)70061-0
Sneed PK, Mendez J, Vemer-van den Hoek JG, Seymour ZA, Ma L, Molinaro AM, Fogh SE, Nakamura JL, McDermott MW (2015) Adverse radiation effect after stereotactic radiosurgery for brain metastases: incidence, time course, and risk factors. J Neurosurg. doi:10.3171/2014.10.JNS141610
Vecil GG, Suki D, Maldaun MV, Lang FF, Sawaya R (2005) Resection of brain metastases previously treated with stereotactic radiosurgery. J Neurosurg 102:209–215. doi:10.3171/jns.2005.102.2.0209
Truong MT, St Clair EG, Donahue BR, Rush SC, Miller DC, Formenti SC, Knopp EA, Han K, Golfinos JG (2006) Results of surgical resection for progression of brain metastases previously treated by gamma knife radiosurgery. Neurosurgery 59:86–97. doi:10.1227/01.NEU.0000219858.80351.38 (discussion 86–97)
Dequesada IM, Quisling RG, Yachnis A, Friedman WA (2008) Can standard magnetic resonance imaging reliably distinguish recurrent tumor from radiation necrosis after radiosurgery for brain metastases? A radiographic-pathological study. Neurosurgery 63:898–903. doi:10.1227/01.NEU.0000333263.31870.31 (discussion 904)
Kano H, Kondziolka D, Lobato-Polo J, Zorro O, Flickinger JC, Lunsford LD (2010) T1/T2 matching to differentiate tumor growth from radiation effects after stereotactic radiosurgery. Neurosurgery 66:486–491. doi:10.1227/01.NEU.0000360391.35749.A5 (discussion 491–482)
Stockham AL, Tievsky AL, Koyfman SA, Reddy CA, Suh JH, Vogelbaum MA, Barnett GH, Chao ST (2012) Conventional MRI does not reliably distinguish radiation necrosis from tumor recurrence after stereotactic radiosurgery. J Neurooncol 109:149–158. doi:10.1007/s11060-012-0881-9
Leeman JE, Clump DA, Flickinger JC, Mintz AH, Burton SA, Heron DE (2013) Extent of perilesional edema differentiates radionecrosis from tumor recurrence following stereotactic radiosurgery for brain metastases. Neuro Oncol 15:1732–1738. doi:10.1093/neuonc/not130
Nath SK, Sheridan AD, Rauch PJ, Yu JB, Minja FJ, Vortmeyer AO, Chiang VL (2014) Significance of histology in determining management of lesions regrowing after radiosurgery. J Neurooncol 117:303–310. doi:10.1007/s11060-014-1389-2
Mathews MS, Linskey ME, Hasso AN, Fruehauf JP (2008) The effect of bevacizumab (Avastin) on neuroimaging of brain metastases. Surg Neurol 70:649–652. doi:10.1016/j.surneu.2007.06.029 (discussion 653)
Iwamoto FM, Fine HA (2010) Bevacizumab for malignant gliomas. Arch Neurol 67:285–288. doi:10.1001/archneurol.2010.11
Kleinschmidt-DeMasters BK, Damek DM (2010) The imaging and neuropathological effects of Bevacizumab (Avastin) in patients with leptomeningeal carcinomatosis. J Neurooncol 96:375–384. doi:10.1007/s11060-009-9969-2
Yomo S, Hayashi M (2014) A minimally invasive treatment option for large metastatic brain tumors: long-term results of two-session Gamma Knife stereotactic radiosurgery. Radiat Oncol 9:132. doi:10.1186/1748-717X-9-132
Mitsuya K, Nakasu Y, Horiguchi S, Harada H, Nishimura T, Bando E, Okawa H, Furukawa Y, Hirai T, Endo M (2010) Perfusion weighted magnetic resonance imaging to distinguish the recurrence of metastatic brain tumors from radiation necrosis after stereotactic radiosurgery. J Neurooncol 99:81–88. doi:10.1007/s11060-009-0106-z
Chernov M, Hayashi M, Izawa M, Ochiai T, Usukura M, Abe K, Ono Y, Muragaki Y, Kubo O, Hori T, Takakura K (2005) Differentiation of the radiation-induced necrosis and tumor recurrence after gamma knife radiosurgery for brain metastases: importance of multi-voxel proton MRS. Minim Invasive Neurosurg 48:228–234. doi:10.1055/s-2005-870952
Tsuyuguchi N, Sunada I, Iwai Y, Yamanaka K, Tanaka K, Takami T, Otsuka Y, Sakamoto S, Ohata K, Goto T, Hara M (2003) Methionine positron emission tomography of recurrent metastatic brain tumor and radiation necrosis after stereotactic radiosurgery: is a differential diagnosis possible? J Neurosurg 98:1056–1064. doi:10.3171/jns.2003.98.5.1056
Terakawa Y, Tsuyuguchi N, Iwai Y, Yamanaka K, Higashiyama S, Takami T, Ohata K (2008) Diagnostic accuracy of 11C-methionine PET for differentiation of recurrent brain tumors from radiation necrosis after radiotherapy. J Nucl Med 49:694–699. doi:10.2967/jnumed.107.048082
Serizawa T, Saeki N, Higuchi Y, Ono J, Matsuda S, Sato M, Yanagisawa M, Iuchi T, Nagano O, Yamaura A (2005) Diagnostic value of thallium-201 chloride single-photon emission computerized tomography in differentiating tumor recurrence from radiation injury after gamma knife surgery for metastatic brain tumors. J Neurosurg 102(Suppl):266–271
Nonoguchi N, Miyatake S, Fukumoto M, Furuse M, Hiramatsu R, Kawabata S, Kuroiwa T, Tsuji M, Fukumoto M, Ono K (2011) The distribution of vascular endothelial growth factor-producing cells in clinical radiation necrosis of the brain: pathological consideration of their potential roles. J Neurooncol 105:423–431. doi:10.1007/s11060-011-0610-9
Ammirati M, Cobbs CS, Linskey ME, Paleologos NA, Ryken TC, Burri SH, Asher AL, Loeffler JS, Robinson PD, Andrews DW, Gaspar LE, Kondziolka D, McDermott M, Mehta MP, Mikkelsen T, Olson JJ, Patchell RA, Kalkanis SN (2010) The role of retreatment in the management of recurrent/progressive brain metastases: a systematic review and evidence-based clinical practice guideline. J Neurooncol 96:85–96. doi:10.1007/s11060-009-0055-6
Papavassiliou E, Gogate N, Proescholdt M, Heiss JD, Walbridge S, Edwards NA, Oldfield EH, Merrill MJ (1997) Vascular endothelial growth factor (vascular permeability factor) expression in injured rat brain. J Neurosci Res 49:451–460
Proescholdt MA, Heiss JD, Walbridge S, Muhlhauser J, Capogrossi MC, Oldfield EH, Merrill MJ (1999) Vascular endothelial growth factor (VEGF) modulates vascular permeability and inflammation in rat brain. J Neuropathol Exp Neurol 58:613–627
Gonzalez J, Kumar AJ, Conrad CA, Levin VA (2007) Effect of bevacizumab on radiation necrosis of the brain. Int J Radiat Oncol Biol Phys 67:323–326. doi:10.1016/j.ijrobp.2006.10.010
Torcuator R, Zuniga R, Mohan YS, Rock J, Doyle T, Anderson J, Gutierrez J, Ryu S, Jain R, Rosenblum M, Mikkelsen T (2009) Initial experience with bevacizumab treatment for biopsy confirmed cerebral radiation necrosis. J Neurooncol 94:63–68. doi:10.1007/s11060-009-9801-z
Schmid S, Aboul-Enein F, Pfisterer W, Birkner T, Stadek C, Knosp E (2010) Vascular endothelial growth factor: the major factor for tumor neovascularization and edema formation in meningioma patients. Neurosurgery 67:1703–1708. doi:10.1227/NEU.0b013e3181fb801b (discussion 1708)
Levin VA, Bidaut L, Hou P, Kumar AJ, Wefel JS, Bekele BN, Prabhu S, Loghin M, Gilbert MR, Jackson EF (2011) Randomized double-blind placebo-controlled trial of bevacizumab therapy for radiation necrosis of the central nervous system. Int J Radiat Oncol Biol Phys 79:1487–1495. doi:10.1016/j.ijrobp.2009.12.061
Williams BJ, Park DM, Sheehan JP (2012) Bevacizumab used for the treatment of severe, refractory perilesional edema due to an arteriovenous malformation treated with stereotactic radiosurgery. J Neurosurg 116:972–977. doi:10.3171/2012.1.JNS111627
Gutin PH, Iwamoto FM, Beal K, Mohile NA, Karimi S, Hou BL, Lymberis S, Yamada Y, Chang J, Abrey LE (2009) Safety and efficacy of bevacizumab with hypofractionated stereotactic irradiation for recurrent malignant gliomas. Int J Radiat Oncol Biol Phys 75:156–163. doi:10.1016/j.ijrobp.2008.10.043
Park KJ, Kano H, Iyer A, Liu X, Niranjan A, Flickinger JC, Lieberman FS, Lunsford LD, Kondziolka D (2012) Salvage gamma knife stereotactic radiosurgery followed by bevacizumab for recurrent glioblastoma multiforme: a case-control study. J Neurooncol 107:323–333. doi:10.1007/s11060-011-0744-9
Cuneo KC, Vredenburgh JJ, Sampson JH, Reardon DA, Desjardins A, Peters KB, Friedman HS, Willett CG, Kirkpatrick JP (2012) Safety and efficacy of stereotactic radiosurgery and adjuvant bevacizumab in patients with recurrent malignant gliomas. Int J Radiat Oncol Biol Phys 82:2018–2024. doi:10.1016/j.ijrobp.2010.12.074
Plotkin SR, Stemmer-Rachamimov AO, Barker FG 2nd, Halpin C, Padera TP, Tyrrell A, Sorensen AG, Jain RK, di Tomaso E (2009) Hearing improvement after bevacizumab in patients with neurofibromatosis type 2. N Engl J Med 361:358–367. doi:10.1056/NEJMoa0902579
Furuse M, Kawabata S, Kuroiwa T, Miyatake S (2011) Repeated treatments with bevacizumab for recurrent radiation necrosis in patients with malignant brain tumors: a report of 2 cases. J Neurooncol 102:471–475. doi:10.1007/s11060-010-0333-3
Boothe D, Young R, Yamada Y, Prager A, Chan T, Beal K (2013) Bevacizumab as a treatment for radiation necrosis of brain metastases post stereotactic radiosurgery. Neuro Oncol 15:1257–1263. doi:10.1093/neuonc/not085
Deibert CP, Ahluwalia MS, Sheehan JP, Link MJ, Hasegawa T, Yomo S, Feng WH, Li P, Flickinger JC, Lunsford LD, Kondziolka D (2013) Bevacizumab for refractory adverse radiation effects after stereotactic radiosurgery. J Neurooncol 115:217–223. doi:10.1007/s11060-013-1214-3
Iwamoto FM, Abrey LE, Beal K, Gutin PH, Rosenblum MK, Reuter VE, DeAngelis LM, Lassman AB (2009) Patterns of relapse and prognosis after bevacizumab failure in recurrent glioblastoma. Neurology 73:1200–1206. doi:10.1212/WNL.0b013e3181bc0184
Acknowledgements
The authors certify that no funding was received to conduct this study and/or for preparation of this manuscript. We are grateful to Bierta Barfod, M.D., M.P.H. for her help with the language editing of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Yomo receives speaking fees from Chugai Pharmaceutical Corporation.
Rights and permissions
About this article
Cite this article
Yomo, S., Hayashi, M. Salvage stereotactic radiosurgery with adjuvant use of bevacizumab for heavily treated recurrent brain metastases: a preliminary report. J Neurooncol 127, 119–126 (2016). https://doi.org/10.1007/s11060-015-2019-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-015-2019-3