Abstract
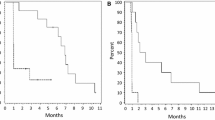
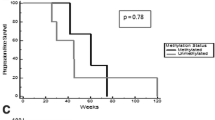
Bevacizumab is frequently used to treat patients with recurrent high-grade glioma (HGG), but responses are generally not durable. Panobinostat is a histone deacetylase inhibitor with anti-neoplastic and anti-angiogenic effects and may work synergistically with VEGF inhibitors. We performed a phase I study to evaluate the safety and tolerability of the combination of orally administered panobinostat with bevacizumab in patients with recurrent HGG. Patients with recurrent HGG were treated on a 3 + 3 trial design. Patients received bevacizumab 10 mg/kg every other week in combination with oral panobinostat. The starting dose of panobinostat was 20 mg three times per week, weekly (cohort 1). Due to concerns for thrombocytopenia with the weekly dosing regimen, the protocol was amended to examine an every other week regimen. Cohort 2 received panobinostat 20 mg three times per week, every other week, and cohort 3 received 30 mg three times per week, every other week. Dose-limiting toxicity during the first 30 days was used to determine the maximum-tolerated dose. Twelve patients (median age 50, median KPS 90) with recurrent HGG were enrolled. One dose-limiting toxicity (DLT) (Grade 3 thrombocytopenia) was observed in cohort 1. No DLTs were observed in cohorts 2 and 3. The following grade 3 toxicities were seen in one patient each: thrombocytopenia, hypophosphatemia, esophageal hemorrhage, and deep venous thrombosis. There were no grade 4 or 5 toxicities. There were three patients with partial responses and seven with stable disease. The recommended doses for further study are oral panobinostat 30 mg three times per week, every other week, in combination with bevacizumab 10 mg/kg every other week. A phase II clinical trial in recurrent HGG is underway.
Similar content being viewed by others
References
CBTRUS (2009) CBTRUS. Statistical report: primary brain tumors in the United States, 2000-2004. http://www.cbtrus.org/reports/2009-NPCR-04-05/CBTRUS-NPCR2004-2005-Report-.pdf. Accessed October 2009
Wen PY, Kesari S (2008) Malignant gliomas in adults. N Engl J Med 359(5):492–507
Friedman HS, Prados MD, Wen PY et al (2009) Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J Clin Oncol 27(28):4733–4740
Kreisl TN, Kim L, Moore K et al (2009) Phase II trial of single-agent bevacizumab followed by bevacizumab plus irinotecan at tumor progression in recurrent glioblastoma. J Clin Oncol 27(5):740–745
Desjardins A, Reardon DA et al (2008) Bevacizumab plus irinotecan in recurrent WHO grade 3 malignant gliomas. Clin Cancer Res 14(21):7068–7073
Vredenburgh JJ, Desjardins A, Herndon JE 2nd et al (2007) Phase II trial of bevacizumab and irinotecan in recurrent malignant glioma. Clin Cancer Res 13(4):1253–1259
Norden AD, Drappatz J, Wen PY (2008) Novel anti-angiogenic therapies for malignant gliomas. Lancet Neurol 7(12):1152–1160
Quant EC, Norden AD, Drappatz J et al (2009) Role of a second chemotherapy in recurrent malignant glioma patients who progress on bevacizumab. Neuro Oncol 11(5):550–555
Atadja P (2009) Development of the pan-DAC inhibitor panobinostat (LBH589): successes and challenges. Cancer Lett 280(2):233–241
Gensert JM, Baranova OV, Weinstein DE, Ratan RR (2007) CD81, a cell cycle regulator, is a novel target for histone deacetylase inhibition in glioma cells. Neurobiol Dis 26(3):671–680
Ugur HC, Ramakrishna N, Bello L et al (2007) Continuous intracranial administration of suberoylanilide hydroxamic acid (SAHA) inhibits tumor growth in an orthotopic glioma model. J Neurooncol 83(3):267–275
Yin D, Ong JM, Hu J et al (2007) Suberoylanilide hydroxamic acid, a histone deacetylase inhibitor: effects on gene expression and growth of glioma cells in vitro and in vivo. Clin Cancer Res 13(3):1045–1052
Sharma V, Koul N, Joseph C, Dixit D, Ghosh S, Sen E (2010) HDAC inhibitor, scriptaid, induces glioma cell apoptosis through JNK activation and inhibits telomerase activity. J Cell Mol Med 14(8):2151–2161
Eyupoglu IY, Hahnen E, Trankle C et al (2006) Experimental therapy of malignant gliomas using the inhibitor of histone deacetylase MS-275. Mol Cancer Ther 5(5):1248–1255
Wetzel M, Premkumar DR, Arnold B, Pollack IF (2005) Effect of trichostatin A, a histone deacetylase inhibitor, on glioma proliferation in vitro by inducing cell cycle arrest and apoptosis. J Neurosurg 103(6 Suppl):549–556
Eyupoglu IY, Hahnen E, Buslei R et al (2005) Suberoylanilide hydroxamic acid (SAHA) has potent anti-glioma properties in vitro, ex vivo and in vivo. J Neurochem 93(4):992–999
Sawa H, Murakami H, Kumagai M et al (2004) Histone deacetylase inhibitor, FK228, induces apoptosis and suppresses cell proliferation of human glioblastoma cells in vitro and in vivo. Acta Neuropathol 107(6):523–531
Galanis E, Jaeckle KA, Maurer MJ et al (2009) Phase II trial of vorinostat in recurrent glioblastoma multiforme: a north central cancer treatment group study. J Clin Oncol 27(12):2052–2058
Deroanne CF, Bonjean K, Servotte S et al (2002) Histone deacetylases inhibitors as anti-angiogenic agents altering vascular endothelial growth factor signaling. Oncogene 21(3):427–436
Qian DZ, Kato Y, Shabbeer S et al (2006) Targeting tumor angiogenesis with histone deacetylase inhibitors: the hydroxamic acid derivative LBH589. Clin Cancer Res 12(2):634–642
Qian DZ, Wang X, Kachhap SK et al (2004) The histone deacetylase inhibitor NVP-LAQ824 inhibits angiogenesis and has a greater antitumor effect in combination with the vascular endothelial growth factor receptor tyrosine kinase inhibitor PTK787/ZK222584. Cancer Res 64(18):6626–6634
Sawa H, Murakami H, Ohshima Y et al (2002) Histone deacetylase inhibitors such as sodium butyrate and trichostatin A inhibit vascular endothelial growth factor (VEGF) secretion from human glioblastoma cells. Brain Tumor Pathol 19(2):77–81
Batchelor TT, Sorensen AG, di Tomaso E et al (2007) AZD2171, a pan-VEGF receptor tyrosine kinase inhibitor, normalizes tumor vasculature and alleviates edema in glioblastoma patients. Cancer Cell 11(1):83–95
Mandawat A, Fiskus W, Buckley KM et al (2010) Pan-histone deacetylase inhibitor panobinostat depletes CXCR4 levels and signaling and exerts synergistic antimyeloid activity in combination with CXCR4 antagonists. Blood 116(24):5306–5315
Kioi M, Vogel H, Schultz G et al (2010) Inhibition of vasculogenesis, but not angiogenesis, prevents the recurrence of glioblastoma after irradiation in mice. J Clin Invest 120(3):694–705
Rubin JB, Kung AL, Klein RS et al (2003) A small-molecule antagonist of CXCR4 inhibits intracranial growth of primary brain tumors. Proc Natl Acad Sci USA 100(23):13513–13518
Lin R, Hu J, Paul S, et al. (2009) Characteristics of thrombocytopenia in patients treated with oral panobinostat (LBH589). Blood (ASH Annual Meeting Abstracts) 114: Abstract 2740
Wen PY, Macdonald DR, Reardon DA et al (2010) Updated response assessment criteria for high-grade gliomas: response assessment in neuro-oncology working group. J Clin Oncol 28(11):1963–1972
Novartis (2011) LBH 589 (Panobinostat) Investigator’s Brochure 7th edn, Brochure. Basel, Switzerland
Acknowledgments
E.Q. Lee has served on the advisory board of Novartis and Genentech/Roche. T. T. Batchelor has served on the advisory board for Genentech/Roche. R. Beroukhim has received research support from Novartis. S.M. Snodgrass is an employee of Novartis. J.J. Raizer has received research support from Novartis and Genentech/Roche, served on the advisory boards of both Novartis and Genentech/Roche and is on the speaker’s bureau of Genentech/Roche. P.Y. Wen has received research support from Novartis and Genentech/Roche and served on the advisory boards of both Novartis and Genentech/Roche.
Conflict of interest
All other authors report no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
First three authors contributed equally to this work
Rights and permissions
About this article
Cite this article
Drappatz, J., Lee, E.Q., Hammond, S. et al. Phase I study of panobinostat in combination with bevacizumab for recurrent high-grade glioma. J Neurooncol 107, 133–138 (2012). https://doi.org/10.1007/s11060-011-0717-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-011-0717-z