Abstract
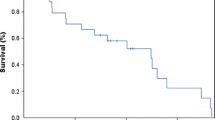
To assess the tolerability and efficacy of liposomal cytarabine (LC), an encapsulated, sustained-release, intrathecal (IT) formulation of cytosine arabinoside, in de novo and relapsed central nervous system (CNS) embryonal tumors in children and young adults. We studied retrospectively all patients less than age 30 at our institution treated consecutively with LC for medulloblastoma (MB), primitive neuroectodermal tumor (PNET), and atypical teratoid rhabdoid tumor (ATRT). Seventeen patients received LC (2 mg/kg up to 50 mg, every 2 weeks to monthly) at diagnosis of high-risk CNS embryonal tumor (2 PNET, 3 ATRT) or relapse of MB (12 MB; 9 had leptomeningeal metastases). Sixteen patients received concurrent systemic chemotherapy. A total of 108 doses were administered (IT 82, intraventricular 26) with a mean of six (range 1–16) treatments per patient. Only three administrations were associated with adverse effects of arachnoiditis or headache. None developed malignant cerebrospinal fluid (CSF) cytology while receiving LC. All the six evaluable patients with malignant CSF cytology and treated with at least two doses cleared their CSF (mean 3 doses, range 1–5). Median overall survival in relapse patients was 9.1 months. Five patients (4 de novo and 1 relapsed) remain alive in complete remission for a median 26.8 months from first LC. Liposomal cytarabine is an easily administered, well-tolerated, and active drug in patients with high-risk embryonal neoplasms. One-third of our cohort remains in remission from otherwise fatal diagnoses. Our findings warrant a phase II trial of LC in newly diagnosed or recurrent CNS embryonal tumors.

Similar content being viewed by others
References
Gurney JG, Smith MA, Bunin GR (1999) CNS and miscellaneous intracranial and intraspinal neoplasms. In: Ries LAG, Smith MA, Gurney JG et al (eds) Cancer Incidence and Survival Among children and Adolescents: United States SEER Program 1975–1995, National Cancer Institute, SEER Program. NIH Pub. No. 99–4649, Bethesda, MD
Blaney SM, Boyett J, Friedman H, Gajjar A, Geyer R, Horowtiz M et al (2005) Phase I clinical trial of mafosfamide in infants and children aged 3 years or younger with newly diagnosed embryonal tumors: a pediatric brain tumor consortium study (PBTC-001). J Clin Oncol 23(3):525–531
Shapiro WR, Young DF, Mehta BM (1975) Methotrexate: distribution in cerebrospinal fluid after intravenous, ventricular, and lumbar injections. N Engl J Med 293(4):161–166
Strong JM, Collins JM, Lester C, Poplack DG (1986) Pharmacokinetics of intraventricular and intravenous N,N’,N”-triethylenethiophosphoramide (thiotepa) in rhesus monkey and humans. Cancer Res 46(12 Pt 1):6101–6104
Zimm S, Collins JM, Miser J, Chatterji D, Poplack DG (1984) Cytosine arabinoside cerebrospinal fluid kinetics. Clin Pharmacol Ther 35(6):826–830
Kim S, Chatelut E, Kim JC, Howell SB, Cates C, Kormanik PA et al (1993) Extended CSF cytarabine exposure following intrathecal administration of DTC 101. J Clin Oncol 11(11):2186–2193
Glantz MJ, LaFollette S, Jaeckle KA, Shapiro W, Swinnen L, Rozental JR et al (1999) Randomized trial of slow-release versus a standard formulation of cytarabine for the intrathecal treatment of lymphomatous meningitis. J Clin Oncol 17(10):3110–3116
Glantz MJ, Jaeckle KA, Chamberlain MC, Phuphanich S, Recht L, Swinnen LJ et al (1999) A randomized controlled trial comparing intrathecal sustained-release cytarabine (Depocyt) to intrathecal methotrexate in patients with neoplastic meningitis from solid tumors. Clin Cancer Res 5(11):3394–3402
Cole BF, Glantz MJ, Jaeckle KA, Chamberlain MC, Mackowiak JI (2003) Quality-of-life-adjusted survival comparison of sustained-release cytosine arabinoside versus intrathecal methotrexate for treatment of solid tumor neoplastic meningitis. Cancer 97(12):3053–3060
Lassaletta A, Perez-Olleros P, Scaglione C, Sirvent S, De Prada I, Perez-Martinez A et al (2007) Successful treatment of intracranial ependymoma with leptomeningeal spread with systemic chemotherapy and intrathecal liposomal cytarabine in a two-year-old child. J Neurooncol 83(3):303–306
Chamberlain MC, Glantz MJ (2008) Sequential salvage chemotherapy for recurrent intracranial hemangiopericytoma. Neurosurgery 63(4):720–727
Bomgaars L, Geyer JR, Franklin J, Dahl G, Park J, Winick NJ et al (2004) Phase I trial of intrathecal liposomal cytarabine in children with neoplastic meningitis. J Clin Oncol 22(19):3916–3921
Benesch M, Siegler N, Hoff K, Lassay L, Kropshofer G, Müller H et al (2009) Safety and toxicity of intrathecal liposomal cytarabine (Depocyte) in children and adolescents with recurrent or refractory brain tumors: a multi-institutional retrospective study. Anticancer Drugs 20(9):794–799
Grossman SA, Finkelstein DM, Ruckdeschel JC, Trump DL, Moynihan T, Ettinger DS (1993) Randomized prospective comparison of intraventricular methotrexate and thiotepa in patients with previously untreated neoplastic meningitis. J Clin Oncol 11(3):561–569
Fisher PG, Kadan-Lottick NS, Korones DN (2007) Intrathecal thiotepa: reappraisal of an established therapy. J Pediatr Hematol Oncol 24(4):274–278
Blaney SM, Heideman R, Berg S, Adamson P, Gillespie A, Geyer JR et al (2003) Phase I clinical trial of intrathecal topotecan in patients with neoplastic meningitis. J Clin Oncol 21(1):143–147
Groves MD, Glantz MJ, Chamberlain MC, Baumgartner KE, Conrad CA, Hsu S et al (2008) A multicenter phase II trial of intrathecal topotecan in patients with meningeal malignancies. Neuro-Oncol 10(2):208–215
Bernardi RJ, Bomgaars L, Fox E, Balis FM, Egorin MJ, Lagattuta TF et al (2008) Phase I clinical trial of intrathecal gemcitabine in patients with neoplastic meningitis. Cancer Chemother Pharmacol 62(2):355–361
Blaney SM, Balis FM, Berg S, Arndt CA, Heideman R, Geyer JR et al (2005) Intrathecal mafosfamide: a preclinical pharmacology and phase I trial. J Clin Oncol 23(7):1555–1563
Gururangan S, Petros WP, Poussaint TY, Hancock ML, Phillips PC, Friedman HS et al (2006) Phase I trial of intrathecal spartaject busulfan in children with neoplastic meningitis: a Pediatric Brain Tumor Consortium Study (PBTC-004). Clin Cancer Res 12(5):1540–1546
Lassaletta A, Lopez-Ibor B, Mateos E, Gonzalez-Vicent M, Perez-Martinez A, Sevilla J et al (2009) Intrathecal liposomal cytarabine in children under 4 years with malignant brain tumors. J Neurooncol 95(1):65–69
Pui C (2007) Toward optimal use of intrathecal liposomal cytarabine. Leuk Lymphoma 48(9):1672–1673
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Partap, S., Murphy, P.A., Vogel, H. et al. Liposomal cytarabine for central nervous system embryonal tumors in children and young adults. J Neurooncol 103, 561–566 (2011). https://doi.org/10.1007/s11060-010-0419-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-010-0419-y