Abstract
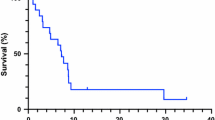
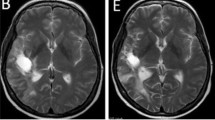
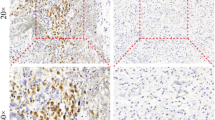
Twenty-two patients with recurrent glioblastoma (GBM) were prospectively treated with everolimus and gefitinib, designed to test the combined inhibition of mammalian target of rapamycin (mTOR) and epidermal growth factor receptor (EGFR) as part of a larger clinical trial. The primary endpoint was radiographic response rate. Secondary endpoints included progression-free survival and correlation of molecular profiles with treatment response. 36% of patients had stable disease and 14% a partial response; however, responses were not durable and only one patient was progression-free at six months. Radiographic changes were not well characterized by conventional response criteria, and implied differential effects of therapy within the tumor and/or antiangiogenic effects. EGFR and PTEN status did not clearly predict response to treatment.

Similar content being viewed by others
References
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–996. doi:10.1056/NEJMoa043330
Salomon DS, Brandt R, Ciardiello F, Normanno N (1995) Epidermal growth factor-related peptides and their receptors in human malignancies. Crit Rev Oncol Hematol 19:183–232. doi:10.1016/1040-8428(94)00144-I
Chakravarti A, Chakladar A, Delaney MA, Latham DE, Loeffler JS (2002) The epidermal growth factor receptor pathway mediates resistance to sequential administration of radiation and chemotherapy in primary human glioblastoma cells in a RAS-dependent manner. Cancer Res 62:4307–4315
Shinojima N, Tada K, Shiraishi S et al (2003) Prognostic value of epidermal growth factor receptor in patients with glioblastoma multiforme. Cancer Res 63:6962–6970
Li B, Yuan M, Kim IA, Chang CM, Bernhard EJ, Shu HK (2004) Mutant epidermal growth factor receptor displays increased signaling through the phosphatidylinositol-3 kinase/AKT pathway and promotes radioresistance in cells of astrocytic origin. Oncogene 23:4594–4602. doi:10.1038/sj.onc.1207602
Rich JN, Reardon DA, Peery T et al (2004) Phase II trial of gefitinib in recurrent glioblastoma. J Clin Oncol 22:133–142. doi:10.1200/JCO.2004.08.110
Prados MD, Lamborn KR, Chang S et al (2006) Phase 1 study of erlotinib HCl alone and combined with temozolomide in patients with stable or recurrent malignant glioma. Neuro-oncology 8:67–78. doi:10.1215/S1522851705000451
Halatsch ME, Schmidt U, Behnke-Mursch J, Unterberg A, Wirtz CR (2006) Epidermal growth factor receptor inhibition for the treatment of glioblastoma multiforme and other malignant brain tumours. Cancer Treat Rev 32:74–89. doi:10.1016/j.ctrv.2006.01.003
Mellinghoff IK, Wang MY, Vivanco I et al (2005) Molecular determinants of the response of glioblastomas to EGFR kinase inhibitors. N Engl J Med 353:2012–2024. doi:10.1056/NEJMoa051918
Haas-Kogan DA, Prados MD, Tihan T et al (2005) Epidermal growth factor receptor, protein kinase B/Akt, and glioma response to erlotinib. J Natl Cancer Inst 97:880–887
Bent MVD, Brandes A, Rampling R et al (2007) Randomized phase II trial of erlotinib (E) versus temozolomide (TMZ) or BCNU in recurrent glioblastoma multiforme (GBM): EORTC 26034. Paper presented at the Abstracts for the Twelfth Annual Meeting of the Society for Neuro-Oncology, 1 October 2007
Prados M, DeBoer R, Chang S et al (2007) Phase II study of Tarceva plus Temodar during and following radiotherapy in patients newly diagnosed with glioblastoma or gliosarcoma. Paper presented at the Abstracts for the Twelfth Annual Meeting of the Society for Neuro-Oncology, 1 October 2007
Hudson CC, Liu M, Chiang GG et al (2002) Regulation of hypoxia-inducible factor 1alpha expression and function by the mammalian target of rapamycin. Mol Cell Biol 22:7004–7014. doi:10.1128/MCB.22.20.7004-7014.2002
Humar R, Kiefer FN, Berns H, Resink TJ, Battegay EJ (2002) Hypoxia enhances vascular cell proliferation and angiogenesis in vitro via rapamycin (mTOR)-dependent signaling. FASEB J 16:771–780. doi:10.1096/fj.01-0658com
Galanis E, Buckner JC, Maurer MJ et al (2005) Phase II trial of temsirolimus (CCI-779) in recurrent glioblastoma multiforme: a North Central Cancer Treatment Group Study. J Clin Oncol 23:5294–5304. doi:10.1200/JCO.2005.23.622
Chang SM, Wen P, Cloughesy T et al (2005) Phase II study of CCI-779 in patients with recurrent glioblastoma multiforme. Invest New Drugs 23:357–361. doi:10.1007/s10637-005-1444-0
Goudar RK, Shi Q, Hjelmeland MD et al (2005) Combination therapy of inhibitors of epidermal growth factor receptor/vascular endothelial growth factor receptor 2 (AEE788) and the mammalian target of rapamycin (RAD001) offers improved glioblastoma tumor growth inhibition. Mol Cancer Ther 4:101–112
Reardon DA, Quinn JA, Vredenburgh JJ et al (2006) Phase 1 trial of gefitinib plus sirolimus in adults with recurrent malignant glioma. Clin Cancer Res 12:860–868. doi:10.1158/1078-0432.CCR-05-2215
Doherty L, Gigas DC, Kesari S et al (2006) Pilot study of the combination of EGFR and mTOR inhibitors in recurrent malignant gliomas. Neurology 67:156–158. doi:10.1212/01.wnl.0000223844.77636.29
Therasse P, Arbuck SG, Eisenhauer EA et al (2000) New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst 92:205–216. doi:10.1093/jnci/92.3.205
Choe G, Horvath S, Cloughesy TF et al (2003) Analysis of the phosphatidylinositol 3′-kinase signaling pathway in glioblastoma patients in vivo. Cancer Res 63:2742–2746
Shaffer DR, Abrey LE, Beekman K et al (2006) A phase I/II trial of RAD 001 with gefitinib in patients with castrate metastatic prostate cancer and glioblastoma multiforme. J Clin Oncol 24:638s
Hu X, Pandolfi PP, Li Y, Koutcher JA, Rosenblum M, Holland EC (2005) mTOR promotes survival and astrocytic characteristics induced by Pten/AKT signaling in glioblastoma. Neoplasia 7:356–368. doi:10.1593/neo.04595
Shih AH, Dai C, Hu X, Rosenblum MK, Koutcher JA, Holland EC (2004) Dose-dependent effects of platelet-derived growth factor-B on glial tumorigenesis. Cancer Res 64:4783–4789. doi:10.1158/0008-5472.CAN-03-3831
Cloughesy TF, Yoshimoto K, Nghiemphu P et al (2008) Antitumor activity of rapamycin in a Phase I trial for patients with recurrent PTEN-deficient glioblastoma. PLoS Med 5(1):e8
O’Reilly KE, Rojo F, She QB et al (2006) mTOR inhibition induces upstream receptor tyrosine kinase signaling and activates Akt. Cancer Res 66:1500–1508. doi:10.1158/0008-5472.CAN-05-2925
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kreisl, T.N., Lassman, A.B., Mischel, P.S. et al. A pilot study of everolimus and gefitinib in the treatment of recurrent glioblastoma (GBM). J Neurooncol 92, 99–105 (2009). https://doi.org/10.1007/s11060-008-9741-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-008-9741-z