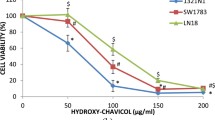
Abstract
Chalcones are considered the precursors of flavonoids and have been identified as interesting compounds with antitumor properties. Boronic-chalcone derivatives are more toxic to breast cancer cells compared to normal breast cells. Here, we studied the antitumor activities of trans-4-lodo,4′-boranyl-chalcone (TLBC), which is a boronic-chalcone derivative, in several glioma cell lines. TLBC showed a dose-dependent inhibition with inhibitory concentration 50% value in the μM range (5.5–25.5 μM) in various glioma cell lines. Flow cytometric and western blot assay demonstrated that TLBC induced apoptosis independent of changes to the tumor suppressor p53. This cytotoxic effect was the caspase-dependent manner. Also, TLBC lowered levels of anti-apoptotic Bcl-2 and/or Bcl-XL protein in several of the cell lines. To examine the antitumor effect of TLBC in vivo, we used a malignant glioma xenograft model. This result showed that in the mice treated with TLBC at 20 mg/kg, mean tumor volume was reduced by 43.9% (P < 0.01) in comparison with the control group. Immunohistochemical and western blot analysis showed that Bcl-2 protein levels were decreased and Bax protein levels were slightly increased in the tumors injected with 20 mg/kg TLBC compared with the control tumors. Therefore, we conclude that TLBC may be a potential chemotherapeutic agent for human glioma.







Similar content being viewed by others
Abbreviations
- Glioblastoma multiforme:
-
GBM
- Trans-4-lodo,4′-boranyl-chalcone:
-
TLBC
- Mouse double minute 2:
-
MDM2
- methylthiazol tetrazolium colorimetric assay:
-
MTT assay
- terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling:
-
TUNEL
- poly(ADP-ribose) polymerase:
-
PARP
- Benzyloxycarbonyl-Val-Ala-Asp (OMe) fluoromethylketone:
-
Z-VAD-fmk
- Half-maximal inhibition of cell proliferation:
-
IC50
References
Behin A, Hoang-Xuan K, Carpentier AF et al (2003) Primary brain tumours in adults. Lancet 361:323–331
Stoner GD, Mukhtar H (1995) Polyphenols as cancer chemopreventive agents. J Cell Biochem Suppl 22:169–180
Yang CS, Landau JM, Huang MT et al (2001) Inhibition of carcinogenesis by dietary polyphenolic compounds. Annu Rev Nutr 21:381–406
Hsu YL, Kuo PL, Tzeng WS et al (2006) Chalcone inhibits the proliferation of human breast cancer cell by blocking cell cycle progression and inducing apoptosis. Food Chem Toxicol 44:704–713
Fu Y, Hsieh TC, Guo J et al (2004) Licochalcone-A, a novel flavonoid isolated from licorice root (Glycyrrhiza glabra), causes G2 and late-G1 arrests in androgen-independent PC-3 prostate cancer cells. Biochem Biophys Res Commun 322:263–270
Zi X, Simoneau AR (2005) Flavokawain A, a novel chalcone from kava extract, induces apoptosis in bladder cancer cells by involvement of Bax protein-dependent and mitochondria-dependent apoptotic pathway and suppresses tumor growth in mice. Cancer Res 65:3479–3486
Kimura Y, Baba K (2003) Antitumor and antimetastatic activities of Angelica keiskei roots, part 1: isolation of an active substance, xanthoangelol. Int J Cancer 106:429–437
Tabata K, Motani K, Takayanagi N et al (2005) Xanthoangelol, a major chalcone constituent of Angelica keiskei, induces apoptosis in neuroblastoma and leukemia cells. Biol Pharm Bull 28:1404–1407
Pan L, Becker H, Gerhauser C (2005) Xanthohumol induces apoptosis in cultured 40–16 human colon cancer cells by activation of the death receptor- and mitochondrial pathway. Mol Nutr Food Res 49:837–843
Nakamura C, Kawasaki N, Miyataka H et al (2002) Synthesis and biological activities of fluorinated chalcone derivatives. Bioorg Med Chem 10:699–706
Go ML, Wu X, Liu XL (2005) Chalcones: an update on cytotoxic and chemoprotective properties. Curr Med Chem 12:481–499
Stoll R, Renner C, Hansen S et al (2001) Chalcone derivatives antagonize interactions between the human oncoprotein MDM2 and p53. Biochemistry 40:336–344
Kumar SK, Hager E, Pettit C et al (2003) Design, synthesis, and evaluation of novel boronic-chalcone derivatives as antitumor agents. J Med Chem 46:2813–2815
Ye CL, Liu JW, Wei DZ et al (2005) In vivo antitumor activity by 2′,4′-dihydroxy-6′-methoxy-3′,5′-dimethylchalcone in a solid human carcinoma xenograft model. Cancer Chemother Pharmacol 56:70–74
Bratton SB, MacFarlane M, Cain K et al (2000) Protein complexes activate distinct caspase cascades in death receptor and stress-induced apoptosis. Exp Cell Res 256:27–33
Cory S (1995) Regulation of lymphocyte survival by the bcl-2 gene family. Annu Rev Immunol 13:513–543
Korsmeyer SJ (1995) Regulators of cell death. Trends Genet 11:101–105
Reed CJ (2000) Apoptosis and cancer: strategies for integrating programmed cell death. Semin Hematol 37(4 Suppl 7):9–16
De Vincenzo R, Ferlini C, Distefano M et al (2000) In vitro evaluation of newly developed chalcone analogues in human cancer cells. Cancer Chemother Pharmacol 46:305–312
Gerhauser C, Alt A, Heiss E et al (2002) Cancer chemopreventive activity of Xanthohumol, a natural product derived from hop. Mol Cancer Ther 1:959–969
Ye CL, Qian F, Wei DZ et al (2005) Induction of apoptosis in K562 human leukemia cells by 2′,4′-dihydroxy-6′-methoxy-3′,5′-dimethylchalcone. Leuk Res 29:887–892
Moll UM, Petrenko O (2003) The MDM2-p53 interaction. Mol Cancer Res 1:1001–1008
Issaeva N, Bozko P, Enge M et al (2004) Small molecule RITA binds to p53, blocks p53-HDM-2 interaction and activates p53 function in tumors. Nat Med 10:1321–1328
Vassilev LT, Vu BT, Graves B et al (2004) In vivo activation of the p53 pathway by small-molecule antagonists of MDM2. Science 303:844–848
Young RL, Korsmeyer SJ (1993) A negative regulatory element in the bcl-2 5′-untranslated region inhibits expression from an upstream promoter. Mol Cell Biol 13:3686–3697
Schiavone N, Rosini P, Quattrone A et al (2000) A conserved AU-rich element in the 3′ untranslated region of bcl-2 mRNA is endowed with a destabilizing function that is involved in bcl-2 down-regulation during apoptosis. FASEB J 14:174–184
Acknowledgements
This work was supported in part by a Grant-in-Aid for Scientific Research to Eiji Kohmura (15659337), Atsufumi Kawamura (16591440), and Takashi Sasayama (17790968) from the Japanese Ministry of Education, Culture, Sports, Science and Technology. We are grateful to Saya H, M.D., Ph.D. (Kumamoto University, Kumamoto), for valuable suggestions on various analyses. We would also like to thank Katoh A and Takase M for technical assistance with the analysis in vitro and experiments in vivo.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sasayama, T., Tanaka, K., Mizukawa, K. et al. Trans-4-lodo,4′-boranyl-chalcone induces antitumor activity against malignant glioma cell lines in vitro and in vivo . J Neurooncol 85, 123–132 (2007). https://doi.org/10.1007/s11060-007-9395-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-007-9395-2