Abstract
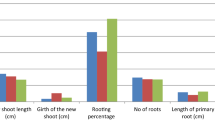
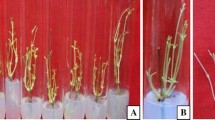
The present study aimed at examining the amenability of Stereospermum suaveolens DC. to vegetative propagation through rooting of leafy stem cuttings using low-cost polythene propagators. Rooting efficiencies of cuttings derived from two selected matured mother trees were studied using four different concentrations of Indole-3-butyric acid (IBA), viz. 0, 0.1, 0.2 and 0.4%. IBA applied at 40 μg per cutting (0.4% concentration) resulted in significantly higher rooting responses compared to the control (P < 0.05). Differences in the length of longest root for all the treatments were significant (P = 0.05) with cuttings treated with 0.2% IBA producing the longest root. The highest survival after 3 weeks of transference of the rooted cuttings into polythene bags was found for rooted cuttings treated with 0.2% IBA. Effects of three fertilizer treatments viz. T0 (no fertilizer), T1 (10 g of Urea + 20 g of TSP + 10 g of MOP dissolved in 1 l of water) and T2 (10 g of Urea + 20 g of TSP + 10 g of MOP dissolved in 2 l of water) on initial growth of established rooted cuttings (stecklings) of Stereospermum suaveolens in polythene bags were also measured for a period of 90 days. Stem length, collar diameter and leaf area of the stecklings increased throughout the observation period. Increments of both stem length and collar diameter were significantly higher for the stecklings treated with T2 in comparison with the control while increment of leaf area was found the highest for the stecklings treated with T1. The study clearly indicated that Stereospermum suaveolens is amenable to vegetative propagation technique using juvenile single-node leafy cuttings and the initial growth performance of the established rooted cuttings (stecklings) in polythene bags under different fertilizer treatments was satisfactory.





Similar content being viewed by others
References
Baul TK (2006) Propagation and growth performance of three important wild tree species of medicinal values. MSc thesis, Institute of Forestry and Environmental Sciences, University of Chittagong, Bangladesh
Berhe D, Negash L (1998) Asexual propagation of Juniperus procera from Ethiopia: a contribution to the conservation of African pencil cedar. For Ecol Manage 112(1–2):179–190
Binutu OA, Adesogan KE, Okogun JI (1996) Antibacterial and antifungal compounds from Kigelia pinnata. Planta Med 62(4):352–353. doi:10.1055/s-2006-957900
Güsewell S, Bollens U, Ryser P, Klötzli F (2003) Contrasting effects of nitrogen, phosphorus and water regime on first- and second-year growth of 16 wetland plant species. Funct Ecol 17(6):754–765. doi:10.1111/j.1365-2435.2003.00784.x
Hassan FU, Leitch MH (2000) Influence of seeding density on contents and uptake of N, P and K in Linseed (Linum usitatissimum L.). J Agron Crop Sci 185(3):193–199. doi:10.1046/j.1439-037x.2000.00427.x
Islam MS (2005) Study on propagation of five wild fruit trees and their domestication. MSc thesis, Institute of Forestry and Environmental Sciences, University of Chittagong, Bangladesh
Joshi KC, Bansal RK, Patni R (1977) Chemical examination of the roots of Stereospermum suaveolens DC. J Indian Chem Soc 54:648–649
Kamaluddin M (1996) Clonal propagation of Eucalyptus and Acacia hybrid by stem cuttings. Research report, Institute of Forestry and Environmental Sciences, University of Chittagong, Bangladesh
Leakey RRB (1987) Clonal forestry in the tropics—a review of developments, strategies and opportunities. Commonw For Rev 66:61–75
Leakey RRB (1990) Nauclea diderrichii: rooting of stem cuttings, clonal variation in shoot dominance, and branch plagiotropism. Trees (Berl) 4(3):164–169. doi:10.1007/BF00225781
Leakey RRB, Storento-West R (1992) The rooting ability of Triplochiton scleroxylon cuttings: the interactions between stock plant irradiance, light quality and nutrients. For Ecol Manage 49:133–150
Leakey RRB, Dick JMcP, Newton AC (1992) Stock plant-derived variation in rooting ability: the source of physiological youth. In: Proceedings of the symposium for mass production technology for genetically improved fast growing forest tree species, 14–18 September, Bordeaux, AFOCEL, Nangis, France, pp 171–178
Leakey RRB, Newton AC, Dick JMcP (1994) Capture of genetic variation by vegetative propagation: processes determining success. In: Leakey RRB, Newton AC (eds) Tropical trees: potential for domestication and the rebuilding of forest resources. HMSO, London, pp 72–83
Leakey RRB, Mesén JF, Tchoundjeu Z, Longman KA, Dick JMcP, Newton A, Matin A, Grace J, Munro RC, Muthoka PN (1990) Low-technology techniques for the vegetative propagation of tropical trees. Commonw For Rev 69(3):247–257
Mesén F, Newton AC, Leakey RRB (1997) Vegetative propagation of Cordia alliodora (Ruiz & Pavon) Oken: the effects of IBA concentration, propagation medium and cutting origin. For Ecol Manage 92(1–3):45–54
Mexal JG, Rangel RAC, Negreros-Castillo P, Lezama CP (2002) Nursery production practices affect survival and growth of tropical hardwoods in Quintana Roo, Mexico. For Ecol Manage 168(1–3):125–133
Minadawati N, Rostawati T (1989) The influence of container size and color on growth of Agathis loranthifolia seedlings. Bulletin Penelitian Hutun Pusat Penelitian dan Pengerbangan Hutun 505:9–10
Negash L (2002) Successful vegetative propagation techniques for the threatened African pencil cedar (Juniperus procera Hoechst. ex Endl.). For Ecol Manage 161(1–3):53–64
Ofori DA, Newton AC, Leakey RRB, Grace J (1996) Vegetative propagation of Milicia excelsa by leafy stem cuttings: effects of auxin concentration, leaf area and rooting medium. For Ecol Manage 84(1–3):39–48
Onegi B, Kraft C, Köhler I, Freund M, Jenett-Siems K, Siems K, Beyer G, Melzig MF, Bienzle U, Eich E (2002) Antiplasmodial activity of naphthoquinones and one anthraquinone from Stereospermum kunthianum. Phytochemistry 60(1):39–44. doi:10.1016/S0031-9422(02)00072-9
Paul SP, Hossain ATME (1996) Effect of cow dung and chemical fertilizers on the growth of Acacia mangium seedlings. Bangladesh J For Sci 25(1&2):49–52
Rieckermann H, Goldfarb B, Cunningham M, Kellison RC (1999) Influence of nitrogen, photoperiod, cutting type and clone on root and shoot development of rooted stem cuttings of sweetgum. New For 18(3):231–244. doi:10.1023/A:1006621330099
Sandermann W, Dietrichs HH (1957) Untersuchungen über termitenresistente Hölzer. Holz als Roh-und Werkstoff 15(7):281–297. doi:10.1007/BF02608522
Schulz KH, Garbe I, Hausen BM, Simatupang MH (1977) The sensitizing capacity of naturally occurring quinones. I. Naphthoquinones and related compounds. Arch Dermatol Forsch 258:41–52
Taiz L, Zeiger E (2002) Plant physiology, 3rd edn. Sinauer Associates, Inc., Sunderland
Tchoundjeu Z, Leakey RRB (1996) Vegetative propagation of African mahogany: effects of auxin, node position, leaf area and cutting length. New For 11(2):125–136. doi:10.1007/BF00033408
Tchoundjeu Z, Avana ML, Leakey RRB, Simons AJ, Asaah E, Duguma B, Bell JM (2002) Vegetative propagation of Prunus africana: effects of rooting medium, auxin concentrations and leaf area. Agrofor Syst 54(3):183–192. doi:10.1023/A:1016049004139
Tchoundjeu Z, Mpeck MLN, Asaah E, Amougou A (2004) The role of vegetative propagation in the domestication of Pausinystalia johimbe (K. Schum), a highly threatened medicinal species of West and Central Africa. For Ecol Manage 188(1–3):175–183
Troup RS (1986) Silviculture of Indian Trees Volume 2: Leguminosae (Caesalpinieae) to Verbenaceae. International Book Distributors, Dehradun, India
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Baul, T.K., Mezbahuddin, M. & Mohiuddin, M. Vegetative propagation and initial growth performance of Stereospermum suaveolens DC., a wild tropical tree species of medicinal value. New Forests 37, 275–283 (2009). https://doi.org/10.1007/s11056-008-9123-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11056-008-9123-6