Abstract
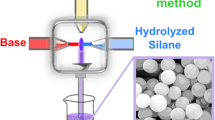
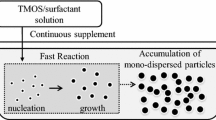
Monodisperse organically modified silica (ORMOSIL) particles, with an average diameter ranging from 550 nm to 4.2 μm, were prepared at low temperature at a scale of about 10 g/batch by a simple one-step self-emulsion process. The reaction mixture was composed only of water, phenyltrimethoxysilane (PTMS), and a base catalyst, without any surfactants. The size control of the particles and the monodispersity of resultant particles were achieved through the controlled supply of hydrolyzed PTMS monomer molecules, which was enabled by manipulating the reaction parameters, such as monomer concentration, type and amount of base catalyst, stirring rate, and reaction temperature. PTMS-based ORMOSIL particles were converted into silica particles by employing either a wet chemical reaction with an oleum-sulfuric acid mixture or thermal treatment above 650 °C. Complete removal of organic groups from the ORMOSIL particles was achieved by the thermal treatment while ~ 74% removal was done by the chemical process used.

ᅟ







Similar content being viewed by others
References
Argyo C, Weiss V, Bräuchle C, Bein T (2014) Multifunctional mesoporous silica nanoparticles as a universal platform for drug delivery. Chem Mater 26(1):435–451. https://doi.org/10.1021/cm402592t
Bogush GH, Tracy MA, Zukoski CF IV (1988) Preparation of monodisperse silica particles: control of size and mass fraction. J Non-Cryst Solids 104(1):95–106. https://doi.org/10.1016/0022-3093(88)90187-1
Chai GS, Shin IS, Yu J (2004) Synthesis of ordered, uniform, macroporous carbons with mesoporous walls templated by aggregates of polystyrene spheres and silica particles for us as catalyst supports in direct methanol fuel cells. Adv Mater 16(22):2057–2061. https://doi.org/10.1002/adma.200400283
Cheong WJ (2014) Porous silica particles as chromatographic separation media: a review. Bull Kor Chem Soc 35(12):3465–3474. https://doi.org/10.5012/bkcs.2014.35.12.3465
de Oliveira LF, Bouchmella K, Gonçalves KA, Bettini J, Kobarg J, Cardoso MB (2016) Functionalized silica nanoparticles as an alternative platform for targeted drug-delivery of water insoluble drugs. Langmuir 32(13):3217–3225. https://doi.org/10.1021/acs.langmuir.6b00214
Genorio B, Pirnat K, Cerc-Korosec R, Dominko R, Gaberscek M (2010) Electroactive organic molecules immobilized onto solid nanoparticles as a cathode material for lithium-ion batteries. Angew Chem Int Ed 49(40):7222–7224. https://doi.org/10.1002/anie.201001539
Ha JM, Jung GI, Kim JS, Kim JW, Jeon SJ, Koo SM (2015) Synthesis of hyperbranched poly(amidoamine) conjugated silica (HBPcS) particles and their application to colorimetric detection of cadmium ion. Colloid Polym Sci 293(8):2331–2339. https://doi.org/10.1007/s00396-015-3623-7
Jung CY, Kim JS, Kim HY, Ha JM, Kim YH, Koo SM (2012) One-pot synthesis and surface modifications of organically modified silica (ORMOSIL) particles having multiple functional groups. J Colloid Interface Sci 367(1):67–73. https://doi.org/10.1016/j.jcis.2011.09.016
Kim P, Kim H, Joo JB, Kim W, Song IK, Yi J (2005) Preparation and application of nanoporous carbon templated by silica particle for use as a catalyst support for direct methanol fuel cell. J Power Sources 145(2):139–146. https://doi.org/10.1016/j.jpowsour.2005.01.070
Kondoh S, Kawada S, Takahashi R, Tajima E (2011) 2.7 inch QVGA type FLC-device for SLM. Ferroelectrics 246(1):121–129. https://doi.org/10.1080/00150190008230059
Lei X, Yu B, Cong H, Tian C, Wang Y, Wang Q, Liu C (2014) Synthesis of monodisperse silica microspheres by a modified Stöber method. Integr Ferroelectr 154(1):142–146. https://doi.org/10.1080/10584587.2014.904651
Nagao D, Osuzu H, Yamada A, Mine E, Kobayashi Y, Konno M (2004) Particle formation in the hydrolysis of tetraethyl orthosilicate in pH buffer solution. J Colloid Interface Sci 279(1):143–149. https://doi.org/10.1016/j.jcis.2004.06.041
Quan B, Lee C, Yoo JS, Piao Y (2017) Facile scalable synthesis of highly monodisperse small silica nanoparticles using alkaline buffer solution and their application for efficient sentinel lymph node mapping. J Mater Chem B 5(3):586–594. https://doi.org/10.1039/C6TB02304E
Roy I, Kumar P, Kumar R, Ohulchanskyy TY, Yong K, Prasad PN (2014) ORMOSIL nanoparticles as a sustained-release drug delivery vehicle. RSC Adv 4(96):53498–53504. https://doi.org/10.1039/C4RA10293B
Santra S, Bagwe RP, Dutta D, Stanley JT, Walter GA, Tan W, Moudgil BM, Mericle RA (2005) Synthesis and characterization of fluorescent, radio-opaque, and paramagnetic silica nanoparticles for multimodal bioimaging applications. Adv Mater 17(18):2165–2169. https://doi.org/10.1002/adma.200500018
Stöber W, Fink A, Bohn E (1968) Controlled growth of monodisperse silica spheres in the micron size range. J Colloid Interface Sci 26(1):62–69. https://doi.org/10.1016/0021-9797(68)90272-5
Swihart MT (2003) Vapor-phase synthesis of nanoparticles. Curr Opin Colloid Interface Sci 8(1):127–133. https://doi.org/10.1016/S1359-0294(03)00007-4
Tan CG, Bowen BD, Epstein N (1987) Production of monodisperse colloidal silica spheres: effect of temperature. J Colloid Interface Sci 118(1):290–293. https://doi.org/10.1016/0021-9797(87)90458-9
Wang X, Shen Z, Sang T, Cheng X, Li M, Chen L, Wang Z (2010) Preparation of spherical silica particles by Stöber process with high concentration of tetra-ethyl-orthosilicate. J Colloid Interface Sci 341(1):23–29. https://doi.org/10.1016/j.jcis.2009.09.018
Wang R, Zhang M, Liu F, Bao S, Wu T, Jiang X, Zhang Q, Zhu M (2015) Investigation on the physical-mechanical properties of dental resin composites reinforced with novel bimodal silica nanostructures. Mater Sci Eng C 50:266–273. https://doi.org/10.1016/j.msec.2015.01.090
Yan F, Jiang J, Chen X, Tian S, Li K (2014) Synthesis and characterization of silica nanoparticles preparing by low-temperature vapor-phase hydrolysis of SiCl4. Ind Eng Chem Res 53(30):11884–11890. https://doi.org/10.1021/ie501759w
Yuan J, Zhou S, You B, Wu L (2005) Organic pigment particles coated with colloidal nano-silica particles via layer-by-layer assembly. Chem Mater 17(14):3587–3594. https://doi.org/10.1021/cm048000b
Zhang J, Raphael AP, Yang Y, Popat A, Prow TW, Yu C (2014) Nanodispersed UV blockers in skin-friendly silica vesicles with superior UV-attenuating efficiency. J Mater Chem B 2(44):7673–7678. https://doi.org/10.1039/C4TB01332H
Zhao B, Tian C, Zhang Y, Tang T, Wang F (2011) Size control of monodisperse nonporous silica particles by seed particle growth. Particuology 9(3):314–317. https://doi.org/10.1016/j.partic.2010.07.028
Funding
This work was supported by the Technology Innovation Program (10052730, Rutile TiO2 Powder Manufacturing Technology by TiCl4 Oxidation) funded by the Ministry of Trade, Industry and Energy (MI, Korea).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest
Electronic supplementary material
ESM 1
(DOCX 2246 kb)
Rights and permissions
About this article
Cite this article
Kim, J.S., Jung, G.I., Kim, S.J. et al. Ten-gram-scale preparation of PTMS-based monodisperse ORMOSIL nano- and microparticles and conversion to silica particles. J Nanopart Res 20, 73 (2018). https://doi.org/10.1007/s11051-018-4186-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-018-4186-6