Abstract
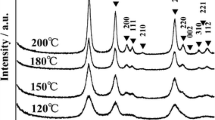
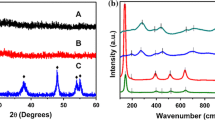
High-temperature phase-stable rice-like anatase TiO2 nanocrystals were synthesized by one-pot solvothermal method using soluble titania xerogel and isopropyl alcohol (IPA) as the precursor and the solvent, respectively. Sample characterization was carried out by powder X-ray diffraction, high-resolution transmission electron microscopy, field emission scanning electron microscope, X-ray photoelectron spectroscopy, and N2 adsorption–desorption isotherms. The results showed that TiO2 nanocrystals had rice-like shapes with an average size of 5 nm in width and 35 nm in length. The BET surface area was 153 m2/g. Unexpectedly, the rice-like TiO2 nanocrystals exhibited high-temperature phase stability, which could remain as pure anatase phase after calcinations at 1000 °C. Growth mechanism investigation revealed that the IPA solvent played a key role in nucleation and growth of rice-like anatase TiO2 nanocrystals. The photodegradation of rhodamine B demonstrated that rice-like anatase TiO2 nanocrystals exhibited enhanced photocatalytic activity under visible light irradiation.









Similar content being viewed by others
References
Bai Y, Mora-Seró I, De Angelis F, Bisquert J, Wang P (2014) Titanium dioxide nanomaterials for photovoltaic applications. Chem Rev. doi:10.1021/cr400606n
Chen X, Mao SS (2007) Titanium dioxide nanomaterials: synthesis, properties, modifications, and applications. Chem Rev 107:2891–2959. doi:10.1021/Cr0500535
D’Arienzo M et al (2011) Photogenerated defects in shape-controlled TiO2 anatase nanocrystals: a probe to evaluate the role of crystal facets in photocatalytic processes. J Am Chem Soc 133:17652–17661. doi:10.1021/ja204838s
Fujishima A, Rao TN, Tryk DA (2000) Titanium dioxide photocatalysis. J Photochem Photobiol C Photochem Rev 1:1–21
Grover IS, Singh S, Pal B (2014) Stable anatase TiO2 formed by calcination of rice-like titania nanorod at 800 °C exhibits high photocatalytic activity. RSC Adv 4:24704–24709. doi:10.1039/c4ra01850h
Li K, Xu J, Shi W, Wang Y, Peng T (2014) Synthesis of size controllable and thermally stable rice-like brookite titania and its application as a scattering layer for nano-sized titania film-based dye-sensitized solar cells. J Mater Chem A 2:1886–1896. doi:10.1039/c3ta13597g
Liao J, Shi L, Yuan S, Zhao Y, Fang J (2009) Solvothermal synthesis of TiO2 nanocrystal colloids from peroxotitanate complex solution and their photocatalytic activities. J Phys Chem C 113:18778–18783. doi:10.1021/jp905720g
Meng X et al (2012) Facile synthesis of direct sunlight-driven anatase TiO2 nanoparticles by in situ modification with trifluoroacetic acid. J Nanopart Res 14:1–7. doi:10.1007/s11051-012-1176-y
Nisar J, Topalian Z, De Sarkar A, Österlund L, Ahuja R (2013) TiO2-based gas sensor: a possible application to SO2. ACS Appl Mater Interfaces 5:8516–8522. doi:10.1021/am4018835
Parmar KPS, Ramasamy E, Lee J, Lee JS (2011) Rapid ([similar]10 min) synthesis of single-crystalline, nanorice TiO2 mesoparticles with a high photovoltaic efficiency of above 8%. Chem Commun 47:8572–8574. doi:10.1039/c1cc12150b
Peng H, Wang X, Li G, Pang H, Chen X (2010) Facile synthesis of rice-like anatase TiO2 nanocrystals. Mater Lett 64:1898–1901. doi:10.1016/j.matlet.2010.05.053
Sun F et al (2012) Fabrication of rice-like porous anatase TiO2 with high thermal stability and enhanced photocatalytic performance. ChemCatChem 4:844–850. doi:10.1002/cctc.201100448
Wang C, Deng Z-X, Zhang G, Fan S, Li Y (2002) Synthesis of nanocrystalline TiO2 in alcohols. Powder Technol 125:39–44. doi:10.1016/S0032-5910(01)00523-X
Wu Y-C, Tai Y-C (2013) Effects of alcohol solvents on anatase TiO2 nanocrystals prepared by microwave-assisted solvothermal method. J Nanopart Res 15:1–11. doi:10.1007/s11051-013-1686-2
Zhou ZY, Tian N, Li JT, Broadwell I, Sun SG (2011) Nanomaterials of high surface energy with exceptional properties in catalysis and energy storage. Chem Soc Rev 40:4167–4185. doi:10.1039/C0cs00176g
Acknowledgments
This work is supported by the National Natural Science Foundation of China (No. 51203094), the Joint program of Beijing Natural Science Foundation and Beijing Academy of Science and Technology (No. L140005), Beijing NOVA Program (Z131101000413038), and Beijing Local College Innovation Team Improve Plan (IDHT20140512).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lv, L., Chen, Q., Liu, X. et al. Facile synthesis of high-temperature (1000 °C) phase-stable rice-like anatase TiO2 nanocrystals. J Nanopart Res 17, 222 (2015). https://doi.org/10.1007/s11051-015-3028-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-015-3028-z