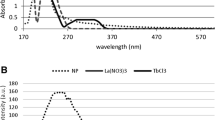
Abstract
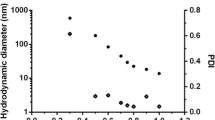
Indium (III) phthalocyanine (InPc) was encapsulated into nanoparticles of PEGylated poly(d,l-lactide-co-glycolide) (PLGA-PEG) to improve the photobiological activity of the photosensitizer. The efficacy of nanoparticles loaded with InPc and their cellular uptake was investigated with MCF-7 breast tumor cells, and compared with the free InPc. The influence of photosensitizer (PS) concentration (1.8–7.5 μmol/L), incubation time (1–2 h), and laser power (10–100 mW) were studied on the photodynamic effect caused by the encapsulated and the free InPc. Nanoparticles with a size distribution ranging from 61 to 243 nm and with InPc entrapment efficiency of 72 ± 6 % were used in the experiments. Only the photodynamic effect of encapsulated InPc was dependent on PS concentration and laser power. The InPc-loaded nanoparticles were more efficient in reducing MCF-7 cell viability than the free PS. For a light dose of 7.5 J/cm2 and laser power of 100 mW, the effectiveness of encapsulated InPc to reduce the viability was 34 ± 3 % while for free InPc was 60 ± 7 %. Confocal microscopy showed that InPc-loaded nanoparticles, as well as free InPc, were found throughout the cytosol. However, the nanoparticle aggregates and the aggregates of free PS were found in the cell periphery and outside of the cell. The nanoparticles aggregates were generated due to the particles concentration used in the experiment because of the small loading of the InPc while the low solubility of InPc caused the formation of aggregates of free PS in the culture medium. The participation of singlet oxygen in the photocytotoxic effect of InPc-loaded nanoparticles was corroborated by electron paramagnetic resonance experiments, and the encapsulation of photosensitizers reduced the photobleaching of InPc.











Similar content being viewed by others
References
Acharya S, Sahoo SK (2011) PLGA nanoparticles containing various anticancer agents and tumor delivery by EPR effect. Adv Drug Deliv Rev 63:170–183. doi:10.1016/j.addr.2010.10.008
Allison RR, Mota HC, Bagnato VS, Sibata CH (2008) Bio-nanotechnology and photodynamic therapy. State of the art review. Photodiagn Photodyn Ther 5:19–28. doi:10.1016/j.pdpdt.2008.02.001
Bechet D, Couleaud P, Frochot C, Viriot ML, Guillemin F, Barberi-Heyob M (2008) Nanoparticles as vehicles for delivery of photodynamic therapy agents. Trends Biotechnol 26:612–621. doi:10.1016/j.tibtech.2008.07.007
Berg K, Western A, Bommer JC, Moan J (1990) Intracellular localization of sulfonated mesotetraphenylporphines in a human carcinoma cell line. Photochem Photobiol 52:481–487. doi:10.1111/j.1751-1097.1990.tb01789.x
Blander JM, Medzhitov R (2006) Toll-dependent selection of microbial antigens for presentation by dendritic cells. Nature 440:808–812. doi:10.1038/nature04596
Bonnett R, Martinez G (2001) Photobleaching of sensitizers used in photodynamic therapy. Tetrahedron 57:9513–9547. doi:10.1016/S0040-4020(01)00952-8
Bourdon O, Mosqueira V, Legrand P, Blais J (2000) A comparative study of the cellular uptake, localization and phototoxicity of meta-tetra(hydroxyphenyl) chlorin encapsulated in surface-modified submicronic oil/water carriers in HT29 tumor cells. J Photochem Photobiol B 55:164–171. doi:10.1016/S1011-1344(00)00043-9
Bozzini G, Collin P, Betrouni N, Nevoux P, Ouzanne A, Puech P, Villers A, Mordon S (2012) Photodynamic therapy in urology: what can we do now and where are we heading? Photodiagn Photodyn Ther 9:261–273. doi:10.1016/j.pdpdt.2012.01.005
Calzavara-Pinton PG, Venturini M, Sala R (2005) A comprehensive overview of photodynamic therapy in the treatment of superficial fungal infections of the skin. J Photochem Photobiol B 78:1–6. doi:10.1016/j.jphotobiol.2004.06.006
Chen Y, Zheng X, Dobhal MP, Gryshuk A, Morgan J, Dougherty TJ, Oseroff A, Pandey RK (2005) Methyl pyropheophorbide-a analogs: potential fluorescent probes for the peripheral-type benzodiazepine receptor. Effect of central metal in photosensitizing efficacy. J Med Chem 48:3692–3695. doi:10.1021/jm050039k
Cruz LJ, Tacken PJ, Fokkink R, Figdor CG (2011) The influence of PEG chain length and targeting moiety on antibody-mediated delivery of nanoparticle vaccines to human dendritic cells. Biomaterials 32:6791–6803. doi:10.1016/j.biomaterials.2011.04.082
Garcia AM, Alarcon E, Munoz M, Scaiano JC, Edwards AM, Lissi E (2011) Photophysical behavior and photodynamic activity of zinc phthalocyanines associated to liposomes. Photochem Photobiol Sci 10:507–514. doi:10.1039/c0pp00289e
Gref R, Minamitake Y, Peracchia MT, Trubetskoy V, Torchilin V, Langer R (1994) Biodegradable long-circulating polymeric nanospheres. Science 263:1600–1603. doi:10.1126/science.8128245
Hans ML, Lowman AM (2002) Biodegradable nanoparticles for drug delivery and targeting. Curr Opin Solid State Mater Sci 6:319–327. doi:10.1016/S1359-0286(02)00117-1
Jeffery H, Davis SS, O’Hagan DT (1991) The preparation and characterization of poly(lactide-co-glycolide) microparticles, I. Oil-in-water emulsion solvent evaporation Int J Pharm 77:169–175. doi:10.1016/0378-5173(91)90314-E
Jokerst JV, Lobovkina T, Zare RN, Gambhir SS (2011) Nanoparticle PEGylation for imaging and therapy. Nanomed 6:715–728. doi:10.2217/nnm.11.19
Juzeniene A, Nielsen KP, Moan J (2006) Biophysical aspects of photodynamic therapy. J Environ Pathol Toxicol Oncol 25:7–28. doi:10.1615/JEnvironPatholToxicolOncol.v25.i1-2.20
Kamaly N, Xiao Z, Valencia PM, Radovic-Moreno AF, Farokhzad OC (2012) Targeted polymeric therapeutic nanoparticles: design, development and clinical translation. Chem Soc Rev 41:2971–3010. doi:10.1039/c2cs15344k
Knop K, Hoogenboom R, Fischer D, Schubert US (2010) Poly(ethylene glycol) in drug delivery: pros and cons as well as potential alternatives. Angew Chem Int Ed 49:6288–6308. doi:10.1002/anie.200902672
Konan YN, Gurny R, Allemann E (2002) State of the art in the delivery of photosensitizers for photodynamic therapy. J Photochem Photobiol B 66:89–106. doi:10.1016/S1011-1344(01)00267-6
Konan YN, Berton M, Gurny R, Allemann E (2003a) Enhanced photodynamic activity of meso-tetra(4-hydroxyphenyl)porphyrin by incorporation into sub-200 nm nanoparticles. Eur J Pharm Sci 18:241–249. doi:10.1016/S0928-0987(03)00017-4
Konan YN, Chevallier J, Gurny R, Allemann E (2003b) Encapsulation of p-THPP into nanoparticles: cellular uptake, subcellular localization and effect of serum on photodynamic therapy. Photochem Photobiol 77:638–644. doi:10.1562/0031-8655(2003)0770638EOPINC2.0.CO2
Korbelik M, Madiyalakan R, Woo T, Haddadi A (2012) Antitumor efficacy of photodynamic therapy using novel nanoformulations of hypocrellin photosensitizer SL052. Photochem Photobiol 88:188–193. doi:10.1111/j.1751-1097.2011.01035.x
Li K, Ma CQ, Liu Y, Zhao FL, Tong SY (2000) Rayleigh light scattering and its applications to biochemical analysis. Chin Sci Bull 45:386–394. doi:10.1007/10.1007/BF02884935
Lion Y, Delmelle M, van de Vorst A (1976) New method of detecting singlet oxygen production. Nature 263:442–443. doi:10.1038/263442a0
Lochmann A, Nitzsche H, von Einem S, Schwarz E, Mader K (2010) The influence of covalently linked and free polyethylene glycol on the structural and release properties of rhBMP-2 loaded microspheres. J Control Release 147:92–100. doi:10.1016/j.jconrel.2010.06.021
Maeda H, Wu J, Sawa T, Matsumura Y, Hori K (2000) Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J Control Release 65:271–284. doi:10.1016/S0168-3659(99)00248-5
Moor ACE (2000) Signaling pathways in cell death and survival after photodynamic therapy. J Photochem Photobiol B 57:1–13. doi:10.1016/S1011-1344(00)00065-8
Mundargi RC, Babu VR, Rangaswamy V, Patel P, Aminabhavi TM (2008) Nano/micro technologies for delivering macromolecular therapeutics using poly(D, L-lactide-co-glycolide) and its derivatives. J Control Release 125:193–209. doi:10.1016/j.jconrel.2007.09.013
Nishiyama N, Nakagishi Y, Morimoto Y, Lai P, Miyazaki K, Urano K, Horie S, Kumagai M, Fukushima S, Cheng Y, Jang W, Kikuchi M, Kataoka K (2009) Enhanced photodynamic cancer treatment by supramolecular nanocarriers charged with dendrimer phthalocyanine. J Control Release 133:245–251. doi:10.1016/j.jconrel.2008.10.010
O’Connor AE, Gallagher WM, Byrne AT (2009) Porphyrin and nonporphyrin photosensitizers in oncology: preclinical and clinical advances in photodynamic therapy. Photochem Photobiol 85:1053–1074. doi:10.1111/j.1751-1097.2009.00585.x
Ochsner M (1997) Photophysical and photobiological processes in the photodynamic therapy of tumours. J Photochem Photobiol B 39:1–18. doi:10.1016/S1011-1344(96)07428-3
Qiang YG, Zhang XP, Li J, Huang Z (2006) Photodynamic therapy for malignant and non-malignant diseases: clinical investigation and application. Chin Med J 119:845–857
Reddi E, Jori G (1988) Steady-state and time-resolved spectroscopic studies of photodynamic sensitizers: porphyrins and phthalocyanines. Rev Chem Intermed 10:241–268. doi:10.1007/BF03155995
Rosenfeld A, Morgan J, Goswami LN, Ohulchankyy T, Zheng X, Prasad PN, Oseroff A, Pandey RK (2006) Photosensitizers derived from 132-oxo-methyl pyropheophorbide-a: enhanced effect of indium(III) as a central metal in vitro and in vivo photosensitizing efficacy. Photochem Photobiol 82:626–634. doi:10.1562/2005-09-29-RA-704
Rosenthal I (1991) Phthalocyanines as photodynamic sensitizers. Photochem Photobiol 53:859–870
Sekkat N, van den Bergh H, Nyokong T, Lange N (2012) Like a bolt from the blue: phthalocyanines in biomedical optics. Molecules 17:98–144. doi:10.3390/molecules17010098
Sharma SK, Mroz P, Daí T, Huang Y, Denis TGS, Hamblin MR (2012) Photodynamic therapy for cancer and for infections: what is the difference? Israel J Chem 52:691–705. doi:10.1002/ijch.201100062
Shutova T, Kriska T, Nemeth A, Agabekov V, Gal D (2000) Physicochemical modeling of the role of free radicals in photodynamic therapy. Biochem Biophys Res Commun 270:125–130. doi:10.1006/bbrc.2000.2385
Silva AR, Inada NM, Rettori D, Baratti MO, Vercesi AE, Jorge RA (2009) In vitro photodynamic activity of chloro(5,10,15,20-tetraphenylporphyrinato)indium(III) loaded- poly(lactide-co-glycolide) nanoparticles in LNCaP prostate tumour cells. J Photochem Photobiol B 94:101–112. doi:10.1016/j.jphotobiol.2008.10.010
Silva AR, de Oliveira AM, Augusto F, Jorge RA (2011) Effects of preparation conditions on the characteristics of PLGA nanospheres loaded with chloro(5,10,15,20-tetraphenylporphyrinato)indium(III). J Nanosci Nanotechnol 11:5234–5246. doi:10.1166/jnn.2011.4136
Soares MV, Oliveira MR, dos Santos EP, Gitirana LB, Barbosa GM, Quaresma CH, Ricci-junior E (2011) Nanostructured delivery system for zinc phthalocyanine: preparation, characterization, and phototoxicity study against human lung adenocarcinoma A549 cells. Int J Nanomed 6:227–238. doi:10.2147/IJN.S15860
Stillman MJ, Nyokong T (1989) Chemical fixation and photoreduction of carbon dioxide catalyzed by metal phthalocyanine derivatives. In: Leznoff CC, Lever ABP (eds) Phthalocyanines: properties and applications, vol 1. chapter 3. Wiley, New York
Triesscheijn M, Baas P, Schellens JHM, Stewart FA (2006) Photodynamic therapy in oncology. Oncol 11:1034–1044. doi:10.1634/theoncologist.11-9-1034
Win KY, Feng SS (2005) Effects of particle size and surface coating on cellular uptake of polymeric nanoparticles for oral delivery of anticancer drugs. Biomaterials 26:2713–2722. doi:10.1016/j.biomaterials.2004.07.050
Xu S, Chen S, Zhang M, Shen T (2003) Synthesis, characterization and photodynamic activity of phenmethylamino-demethoxy-hypocrellin B. J Photochem Photobiol B 72:61–67. doi:10.1016/j.jphotobiol.2003.09.001
Zeisser-Labouebe M, Lange N, Gurny R, Delie F (2006) Hypericin-loaded nanoparticles for the photodynamic treatment of ovarian cancer. Int J Pharm 326:174–181. doi:10.1016/j.ijpharm.2006.07.012
Acknowledgments
We thank the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq) and the Federal Institute of Espírito Santo for financial support, the Instituto Nacional de Ciência e Tecnologia de Fotônica Aplicada à Biologia Celular (INFABIC) for the confocal microscopy analysis and Prof. Geovane Lopes de Sena from Federal University of Espírito Santo for fluorescence analysis.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Souto, C.A.Z., Madeira, K.P., Rettori, D. et al. Improved photodynamic action of nanoparticles loaded with indium (III) phthalocyanine on MCF-7 breast cancer cells. J Nanopart Res 15, 1879 (2013). https://doi.org/10.1007/s11051-013-1879-8
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-013-1879-8