Abstract
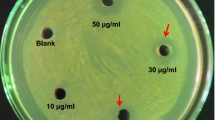
Xanthomonas oryzae pv. oryzae (Xoo) is one representative phytopathogenic bacterium causing bacteria infections in rice. The antibacterial activity of graphene suspended in different dispersants against Xoo was first investigated. Bacteriological test data, fluorescence microscope and transmission electron microscopy images are provided, which yield insight into the antibacterial action of the nanoscale materials. Surprisingly, the results showed graphene oxide (GO) exhibits superior bactericidal effect even at extremely low dose in water (250 μg/mL), almost killing 94.48 % cells, in comparison to common bactericide bismerthiazol with only 13.3 % mortality. The high efficiency in inactivating the bacteria on account of considerable changes in the cell membranes caused by the extremely sharp edges of graphene oxide and generation of reactive oxygen species, which may be the fatal factor for bacterial inactivation. Given the superior antibacterial effect of GO and the fact that GO can be mass-produced with low cost, we expect a new application could be developed as bactericide for controlling plant disease, which may be a matter of great importance for agricultural development.










Similar content being viewed by others
References
Akhavan O, Ghaderi E (2010) Toxicity of graphene and graphene oxide nanowalls against bacteria. ACS Nano 4:5731–5736
Arias LR, Yang LJ (2009) Inactivation of bacterial pathogens by carbon nanotubes in suspensions. Langmuir 25:3003–3012
Bai S, Shen XP (2012) Graphene-inorganic nanocomposites. RSC Adv 2:64–98
Baker B, Zambryski P, Staskawicz B, Dinesh-Kumar SP (1997) Signaling in plant-microbe interactions. Science 276:726–733
Balandin AA, Ghosh S, Bao WZ, Calizo I, Teweldebrhan D, Miao F, Lau CN (2008) Superior thermal conductivity of single-layer graphene. Nano Lett 8:902–907
Begum P, Ikhtiari R et al (2011) Graphene phytotoxicity in the seedling stage of cabbage, tomato, red spinach, and lettuce. Carbon 49:3907–3919
Bolotin KI, Sikes KJ, Jiang Z, Klima M, Fudenberg G, Hone J, Kim P, Stormer HL (2008) Ultrahigh electron mobility in suspended graphene. Solid State Commun 146:351–355
Chang YL, Yang ST, Liu JH, Dong E, Wang Y, Cao A, Liu YF, Wang HF (2011) In vitro toxicity evaluation of graphene oxide on A549 cells. Toxicol Lett 3:201–210
Chen CZS, Cooper SL (2002) Interactions between dendrimer biocides and bacterial membranes. Biomaterials 23:3359–3368
Dang TC, Fujii M, Rose AL, Bligh M, Waitea TD (2012) Characteristics of the freshwater cyanobacterium microcystis aeruginosa grown in iron-limited continuous culture. Appl Environ Microbiol 78:1574–1583
Dong L, Joseph KL, Witkowski CM, Craig MM (2008) Cytotoxicity of single-walled carbon nanotubes suspended in various surfactants. Nanotechnology 19(25):255702. doi:10.1088/0957-4484/19/25/255702
Gu K et al (2005) R gene expression induced by a type-III effector triggers disease resistance in rice. Nature 435:1122–1125
Hotze EM, Labille J, Alvarez P, Wiesner MR (2008) Mechanisms of photochemistry and reactive oxygen production by fullerene suspensions in water. Environ Sci Technol 42:4175–4180
Hu WB, Peng C, Luo WJ, Lv M, Li XM, Li D, Huang Q, Fan CH (2010) Graphene-based antibacterial paper. ACS Nano 4:4317–4323
Hummers WS, Offeman RE (1958) Preparation of graphitic oxide. J Am Chem Soc 80:1339
Hussain SM, Braydich-Stolle LK, Schrand AM, Murdock RC, Yu KO, Mattie DM, Schlager JJ, Terrones M (2009) Toxicity evaluation for safe use of nanomaterials: recent achievements and technical challenges. Adv Mater 21:1549–1559
Imfelda G, Vuilleumierb S (2012) Measuring the effects of pesticides on bacterial communities in soil: a critical review. Eur J Soil Biol 49:22–30
Jin Z, Nackashi D, Lu W, Kittrell C, Tour JM (2010) Decoration, migration, and aggregation of palladium nanoparticles on graphene sheets. Chem Mater 22:5695–5699
Kang S, Herzberg M, Rodrigues DF, Elimelech M (2008) Antibacterial effects of carbon nanotubes: size does matter! Langmuir 24:6409–6413
Khodakovskaya MV, de Silvaa K, Nedosekinb DA, Dervishic E, Birisa AS, Shashkovb EV et al (2011) Complex genetic, photothermal, and photoacoustic analysis of nanoparticle-plant interactions. Proc Natl Acad Sci USA 108:1028–1033
Leach JE, Leung H, Nelson HL, Mew TW (1995) Population biology of Xanthomonas oryzae pv. oryzae and approaches to its control. Curr Opin Biotechnol 6:298–304
Liao HH, Qi RL, Shen MW, Cao XY, Guo R, Zhang YZ, Shi XY (2011) Improved cellular response on multiwalled carbon nanotube-incorporated electrospun polyvinyl alcohol/chitosan nanofibrous scaffolds. Colloids Surf. B: Biointerfaces 84:528–535
Liu Z, Robinson JT, Sun XM, Dai HG (2008) PEGylated nanographene oxide for delivery of water-insoluble cancer drugs. J Am Chem Soc 130:10876–10877
Liu SB, Wei L, Hao L, Fang N, Chang MW, Xu R, Yang YH, Chen Y (2009) Sharper and faster “nano darts” kill more bacteria: a study of antibacterial activity of individually dispersed pristine single-walled carbon nanotube. ACS Nano 3:3891–3902
Liu SB, Zeng TH, Hofmann M, Burcombe E, Wei J, Jiang RR, Kong J, Chen Y (2011) Antibacterial activity of graphite, graphite oxide, graphene oxide, and reduced graphene oxide: membrane and oxidative stress. ACS Nano 5:6971–6980
Liu SB, Hu M, Zeng TH, Wu R, Jiang RR, Wei J, Wang L, Kong J, Chen Y (2012) Lateral dimension dependent antibacterial activity of graphene oxide sheets. Langmuir 28:12364–12372
Lyon DY, Alvarez PJJ (2008) Fullerene water suspension (nC60) exerts antibacterial effects via ROS-independent protein oxidation. Environ Sci Technol 42:8127–8132
Mew TW (1987) Current status and future prospects of research on bacterial blight of rice. Ann. Rev. Phytopathol 25:359–382
Monroc S, Badosa E, Besalú E, Planas M, Bardajía E, Montesinosb E, Feliua L (2006) Improvement of cyclic decapeptides against plant pathogenic bacteria using a combinatorial chemistry approach. Peptides 27:2575–2584
Moore VC, Strano MS, Haroz EH, Hauge RH, Smalley RE (2003) Individually suspended single-walled carbon nanotubes in various surfactants. Nano Lett 3:1379–1382
Ryba-White M, Notteghem JL, Leach JE (1995) Comparison of Xanthomonas oryzae pv. strains from Africa, north America and Asia by RFLP analysis. Intl Rice Res Notes 20:25–26
Stankovich S, Dikin DA, Piner RD, Kohlhaas KA, Kleinhammmes A, Jia Y, Wu Y, Nguyen ST, Ruoff RS (2007) Synthesis of graphene-based nanosheets via chemical reduction of exfoliated graphite oxide. Carbon 45:1558–1563
Tan XM, Lin C, Fugetsu B (2009) Studies on toxicity of multi-walled carbon nanotubes on suspension rice cells. Carbon 47:3479–3487
Vidaver AK (2002) Uses of antimicrobials in plant agriculture. Clin Infect Dis 34:107–110
Vila M et al (2012) Cell uptake survey of pegylated nanographene oxide. Nanotechnology 13(46):465103. doi:10.1088/0957-4484/23/46/465103
Wang XP, Liu XQ, Han HY (2013) Evaluation of antibacterial effects of carbon nanomaterials against copper-resistant Ralstonia solanacearum. Colliods Surf. B Biointerfaces 103:136–142
Xu YX, Bai H, Lu GW, Li C, Shi GQ (2008) Flexible graphene films via the filtration of water-soluble noncovalent functionalized graphene sheets. J Am Chem Soc 130:5856–5857
Yang XY, Zhang XY, Liu ZF, Ma YF, Huang Y, Chen YS (2008) High-efficiency loading and controlled release of doxorubicin hydrochloride on graphene oxide. J Phys Chem C 112:17554–17558
Zhang YB, Ali SF, Dervishi E, Xu Y, Li ZR, Casciano D, Biris AS (2010) Cytotoxicity effects of graphene and single-wall carbon nanotubes in neural phaeochromocytoma-derived PC12 cells. ACS Nano 4:3181–3186
Zhou Y, Bao QL, Tang LAL, Zhong YL, Loh KP (2009) Hydrothermal dehydration for the “green” reduction of exfoliated graphene oxide to graphene and demonstration of tunable optical limiting properties. Chem Mater 21:2950–2956
Acknowledgments
The authors gratefully acknowledge the financial support for this research from National Natural Science Foundation of China (21175051), the Fundamental Research Funds for the Central Universities (2011PY139), and the Natural Science Foundation of Hubei Province Innovation Team (2011CDA115).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Chen, J., Wang, X. & Han, H. A new function of graphene oxide emerges: inactivating phytopathogenic bacterium Xanthomonas oryzae pv. Oryzae . J Nanopart Res 15, 1658 (2013). https://doi.org/10.1007/s11051-013-1658-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-013-1658-6