Abstract
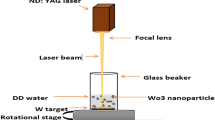
With the use of a modified plasma arc gas condensation technique and control of the processing parameters, namely, plasma current and chamber pressure, we synthesized tungsten oxide nanomaterials with aspect ratios ranging from 1.1 (for equiaxed particles with the length and width of 48 nm and 44 nm, respectively) to 12.7 (for rods with the length and width of 266 nm and 21 nm, respectively). The plasma current and chamber pressure, respectively, ranged from 70 to 90 A and from 200 to 600 Torr. We then characterized the tungsten oxide nanomaterials by means of X-ray diffraction, high-resolution transmission electron microscope, UV–visible spectroscope, and photoluminescence (PL) spectroscope. Experimental results show that equiaxed tungsten oxide nanoparticles were produced at a relatively low plasma current of 70 A, whereas nanorods were produced when plasma currents or chamber pressures were increased. All of the as-prepared tungsten oxide nanomaterials exhibited a WO2.8 phase. Compared to the nanoparticles, the nanorods exhibited unique properties, such as a redshift in the UV–visible spectrum, a blue emission in PL spectrum, and a good performance in field emission. With respect to the field emission, the turn-on voltage for WO2.8 nanorods was found to be as low as 1.7 V/μm.







Similar content being viewed by others
References
Ağiral A, Gardeniers JGE (2008) Synthesis and atmospheric pressure field emission operation of W18O49 nanorods. J Phys Chem C 112:15183–15189. doi:10.1021/jp809458j
Balázsi C, Wang L, Zayim EO, Szilágyi IM, Sedlacková K, Pfeifer J, Tóth AL, Gouma PI (2008) Nanosize hexagonal tungsten oxide for gas sensing applications. J Eur Ceram Soc 28:913–917. doi:10.1016/j.jeurceramsoc.2007.09.001
Biswas S, Kar S, Chaudhuri S (2006) Synthesis and characterization of zinc sulfide nanostructures. Synth React Inorg Met Org Nano Met Chem 36:33–36. doi:10.1080/15533170500471417
Chen CH, Wang SJ, Ko RM, Kuo YC, Uang KM, Chen TM, Liou BW, Tsai HY (2006) The influence of oxygen content in the sputtering gas on the self-synthesis of tungsten oxide nanowires on sputter-deposited tungsten films. Nanotechnology 17:217–223. doi:10.1088/0957-4484/17/1/036
Choi HG, Jung YH, Kim DK (2005) Solvothermal synthesis of tungsten oxide nanorod/nanowire/nanosheet. J Am Ceram Soc 88:1684–1686. doi:10.1111/j.1551-2916.2005.00341.x
Cross WB, Parkin IP (2003) Aerosol assisted chemical vapour deposition of tungsten oxide films from polyoxotungstate precursors: active photocatalysts. Chem Commun 9:1696–1697. doi:10.1039/b303800a
Feng M, Pan AL, Zhang HR, Li ZA, Liu F, Liu W, Shi DX, Zou BS, Gao HJ (2005) Strong photoluminescence of nanostructured crystalline tungsten oxide thin films. Appl Phys Lett 86:141901–141901-3. doi:10.1063/1.1898434
Fowler RH, Nordheim LW (1928) Electron emission in intense electric fields. Proc R Soc Lond A 119:173–181. doi:10.1098/rspa.1928.0091
Guo Y, Murata N, Ono K, Okazaki T (2005) Production of ultrafine particles of high-temperature tetragonal WO3 by dc arc discharge in Ar-O2 gases. J Nanopart Res 7:101–106. doi:10.1007/s11051-004-7900-5
He T, Yao J (2007) Photochromic materials based on tungsten oxide. J Mater Chem 17:4547–4557. doi:10.1039/b709380b
He T, Ma Y, Cao Y, Hu X, Liu H, Zhang G, Yang W, Yao J (2002) Photochromism of WO3 colloids combined with TiO2 nanoparticles. J Phys Chem B 106:12670–12676. doi:10.1021/jp026031t
Hong K, Xie M, Wu H (2006) Tungsten oxide nanowires synthesized by a catalyst-free method at low temperature. Nanotechnology 17:4830–4833. doi:10.1088/0957-4484/17/19/008
Hu R, Wu H, Hong K (2007) Growth of uniform tungsten oxide nanowires with small diameter via a two-step heating process. J Cryst Growth 306:395–399. doi:10.1016/j.jcrysgro.2007.05.007
Huang K, Pan Q, Yang F, Ni S, He D (2008) The catalyst-free synthesis of large-area tungsten oxide nanowire arrays on ITO substrate and field emission properties. Mater Res Bull 43:919–925. doi:10.1016/j.materresbull.2007.04.036
Jeon S, Yong K (2007) Direct synthesis of W18O49 nanorods from W2N film by thermal annealing. Nanotechnology 18:245602. doi:10.1088/0957-4484/18/24/245602
Kichambare P, Hi KF, Sadanadan RRB, Ro AM, Javed K, Menguc MP (2006) Growth of tungsten oxide nanorods with carbon caps. J Nanosci Nanotechnol 6:536–540. doi:10.1166/jnn.2006.095
Kojima Y, Kasuya K, Ooi T, Nagato K, Takayama K, Nakao M (2007) Effects of oxidation during synthesis on structure and field-emission property of tungsten oxide nanowires. Jpn J Appl Phys 46:6250–6253. doi:10.1143/JJAP.46.6250
Lee K, Seo WS, Park JT (2003) Synthesis and optical properties of colloidal tungsten oxide nanorods. J Am Chem Soc 125:3408–3409. doi:10.1021/ja034011e
Li YB, Bando Y, Golberg D, Kurashima K (2003) WO3 nanorods/nanobelts synthesized via physical vapor deposition process. Chem Phys Lett 367:214–218. doi:10.1016/S0009-2614(02)01702-5
Liu Z, Bando Y, Tang C (2003) Synthesis of tungsten oxide nanowires. Chem Phys Lett 372:179–182. doi:10.1016/S0009-2614(03)00397-X
Lou J, Ye BJ, Weng HM, Du HJ, Wang ZB, Wang XP (2008) The influence of filament temperature and oxygen concentration on tungsten oxide nanostructures by hot filament metal oxide deposition. J Phys D 41:155410. doi:10.1088/0022-3727/41/15/155410
Lu DY, Chen J, Zhou J, Deng SZ, Xu NS, Xu JB (2007) Raman spectroscopic study of oxidation and phase transition in W18O49 nanowires. J Raman Spectrosc 38:176–180. doi:10.1002/jrs.1620
Nilasson GA, Granqvist CG (2007) Electrochromics for smart windows: thin films of tungsten oxide and nickel oxide, and devices based on these. J Mater Chem 17:127–156. doi:10.1039/b612174h
Polleux J, Gurlo A, Barsan N, Weimar U, Antonietti M, Niederberger M (2005) Template-free synthesis and assembly of single-crystalline tungsten oxide nanowires and their gas-sensing properties. Angew Chem Int Ed Engl 45:261–265. doi:10.1002/anie.200502823
Seelaboyina R, Huang J, Park J, Kang DH, Choi WB (2006) Multistage field enhancement of tungsten oxide nanowires and its field emission in various vacuum conditions. Nanotechnology 17:4840–4844. doi:10.1088/0957-4484/17/19/010
Su CY, Lin HC (2009) Direct route to tungsten oxide nanorod bundles: microstructures and electro-optical properties. J Phys Chem C 113:4042–4046. doi:10.1021/jp809458j
Su CY, Lin CK, Cheng CW (2005) A modified plasma arc gas condensation technique to synthesize nanocrystalline tungsten oxide powders. Mater Trans 46:1016–1020. doi:10.2320/matertrans.46.1016
Su CY, Lin CK, Yang TK, Lin HC, Pan CT (2008) Oxygen partial pressure effect on the preparation of nanocrystalline tungsten oxide powders by a plasma arc gas condensation technique. Int J Refract Met Hard Mater 26:423–428. doi:10.1016/j.ijrmhm.2007.09.006
Subrahmanyam A, Karuppasamy A (2007) Optical and electrochromic properties of oxygen sputtered tungsten oxide (WO3) thin films. Sol Energy Mater Sol Cells 91:266–274. doi:10.1016/j.solmat.2006.09.005
Thangala J, Vaddiraju S, Bogale R, Thurman R, Powers T, Deb B, Sunkara MK (2007) Large-scale, hot-filament-assisted synthesis of tungsten oxide and related transition metal oxide nanowires. Small 3:890–896. doi:10.1002/smll.200600689
Zhang Y, Chen Y, Liu H, Zhou Y, Li R, Cai M, Sun X (2009) Three-dimensional hierarchical structure of single crystalline tungsten oxide nanowires: construction, phase transition, and voltammetric behavior. J Phys Chem C 113:1746–1750. doi:10.1021/jp808774m
Zhao YM, Li YH, Ahmad I, McCartney DG, Zhu YQ, Hu WB (2006) Two-dimensional tungsten oxide nanowire networks. Appl Phys Lett 89:133116. doi:10.1063/1.2357609
Zheng Y, Chen C, Zha Y, Lin X, Zheng Q, Wei K, Zhu J, Zhu Y (2007) Luminescence and photocatalytic activity of ZnO nanocrystals: correlation between structure and property. Inorg Chem 46:6675–6682. doi:10.1021/ic062394m
Zhou J, Gong L, Deng SZ, Chen J, She JC, Xu NS, Yang R, Wang ZL (2005) Growth and field-emission property of tungsten oxide nanotip arrays. Appl Phys Lett 87:223108. doi:10.1063/1.2136006
Acknowledgments
This study was supported by the National Science Council of Taiwan under contract no. NSC94-2216-E-027-008 and NSC95-2216-E-027-009. The authors would like to thank Ms. Ying-Mei Chang and Ms. Liang-Chu Wang for their technical assistance on transmission electron microscope.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Su, CY., Lin, HC., Yang, TK. et al. Structure and optical properties of tungsten oxide nanomaterials prepared by a modified plasma arc gas condensation technique. J Nanopart Res 12, 1755–1763 (2010). https://doi.org/10.1007/s11051-009-9730-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-009-9730-y