Abstract
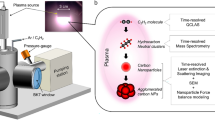
The microwave plasma process inherently produces nanoparticulate powders with very narrow particle size distribution. During synthesis, the particles carry electric charges of equal sign. Therefore, by electrostatic repulsion, particle growth is reduced and agglomeration thwarted. This is shown by gas kinetic considerations and experimental results. Furthermore, this process allows coating of the particles with organic or inorganic phases, reducing interaction of different particles. This makes it possible to technically exploit properties, characteristic for isolated particles. Additionally, the coating process allows the combination of different properties such as superparamagnetism and luminescence, as it is demonstrated in different examples.
Similar content being viewed by others
References
Albe V., Jouanin C. and Bertho D., (1998). Phys. Rev. B 58: 4713
Brus L.E., (1984). J. Chem. Phys. 80: 4403
Fu H. and Zunger A., (1997). Phys. Rev. B 55: 1642
Jacobs, I.S. & C.P. Bean, 1963. In: Rado G.T. & Suhl H. eds. Magnetism. Academic Press, New York, 271ff pp.
Lamparth I., Szabó D.V. and Vollath D., (2002). Marcomolecular Symposia 181: 107
MacDonald A.D. (1966). Microwave Breakdown in Gases. John Wiley & Sons, New York
Micic O.I., Sprague J., Lu Z. and Nozik J., (1996). Appl. Phys. Lett. 73: 3150
Monticone S., Tufeu R. and Kanaev A.V., (1998). J. Phys. Chem. B 102: 2854–2862
Néel L., (1949). Comt. Rend. 228: 664
Roth P. 2000, private communication
Sun C.Q., Chen T.P., Tay B.K., Li S., Huang H., Zhang Y.B., Pan L.K., Lau S.P. and Sun X.W., (2001). J. Phys. D: 34: 3470–3479
Vollath D. and Sickafus K.E., (1992). NanoStructured Materials 1: 427
Vollath D., (1994). Mat. Res. Soc. Symp. Proc. 347: 629
Vollath D. and Szabó D.V., (1994). NanoStructured Materials 4: 927
Vollath, D., D.V. Szabó & B. Seith, 1998. German Patent DE19638601C1
Vollath, D. & D.V. Szabó, 2002. In: K.L. Choy, ed. Innovative Processing of Films and Nanocrystalline Powders. Imperial College Press, London
Vollath D., Lamparth I. and Szabó D.V., (2002). Mat. Res. Soc. Symp. Proc. 703: V7.8.1
Vollath D., Szabó D.V. and Schlabach S., (2004). J. Nanoparticle Research 6: 181
Ziemann P.J., Kittelson D.B. and McMurry P.H., (1996). J. Aerosol Sci. 27 (4): 587
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Vollath, D., Szabó, D.V. The Microwave plasma process – a versatile process to synthesise nanoparticulate materials. J Nanopart Res 8, 417–428 (2006). https://doi.org/10.1007/s11051-005-9014-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11051-005-9014-0