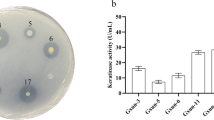
Abstract
A novel feather-degrading bacterium named CA-1 was isolated from the gut of the spider Chilobrachys guangxiensis, which degrades native whole chicken feathers within 20 h. The CA-1 was confirmed to belong to Stenotrophomonas maltophilia based on morphologic and molecular analysis. Maximum feather degradation activity of the bacterium was observed at 37 °C in basal feather medium (NaCl 0.5 g/L, KH2PO4 0.3 g/L, K2HPO4 0.4 g/L, feather powder 10.0 g/L, pH 8.0), which was inhibited when glucose and ammonium nitrate were added in the medium. Furthermore, the purified enzymes under the optimal and suppressive conditions were analyzed respectively by SDS-PAGE and LC–MS/MS. Three enzymes, namely alkaline serine protease (29.1 kDa), ABC transporter permease (27.5 kDa), and alkaline phosphatase (40.8 kDa), were isolated and identified from the supernatant of the optimal culture and were considered to play principal roles. On the other hand, the potential synergic effects of the three proteins in S. maltophilia CA-1 feather degradation system were analyzed theoretically. CA-1 may product outer-membrane vesicles comprised of membranes and periplasmic proteins in the feather medium. The newly identified CA-1 and its synergic enzymes provide a new insight into further understanding the molecular mechanism of feather degradation by microbes. They also have potential application in cost-effectively degrading feathers into feeds and fertilizers through careful optimization and engineering of the three newly identified enzymes.





Similar content being viewed by others
References
Riffel A et al (2007) Purification and characterization of a keratinolytic metalloprotease from Chryseobacterium sp. kr6. J Biotechnol 128(3):693–703
Zaghloul TI, Embaby AM, Elmahdy AR (2011) Biodegradation of chicken feathers waste directed by Bacillus subtilis recombinant cells: scaling up in a laboratory scale fermentor. Bioresour Technol 102(3):2387–2393
Paul T, Halder SK, Das A, Bera S, Maity C, Mandal A, Das PS, Mohapatra PKD, Pati BR, Mondal KC (2013) Exploitation of chicken feather waste as a plant growth promoting agent using keratinase producing novel isolate Paenibacillus woosongensis TKB2. Biocatal Agric Biotechnol 2:50–57
Eslahi N, Dadashian F, Nejad NH (2013) An investigation on keratin extraction from wool and feather waste by enzymatic hydrolysis. Prep Biochem Biotechnol 43(7):624–648
Gupta R, Ramnani P (2006) Microbial keratinases and their prospective applications: an overview. Appl Microbiol Biotechnol 70(1):21–33
Daroit DJ, Brandelli A (2014) A current assessment on the production of bacterial keratinases. Crit Rev Biotechnol 34(4):372–384
Gupta R, Sharma R, Beg QK (2013) Revisiting microbial keratinases: next generation proteases for sustainable biotechnology. Crit Rev Biotechnol 33(2):216–228
Vesela M, Friedrich J (2009) Amino acid and soluble protein cocktail from waste keratin hydrolysed by a fungal keratinase of Paecilomyces marquandii. Biotechnol Bioprocess Eng 14(1):84–90
Poopathi S et al (2014) Purification and characterization of keratinase from feather degrading bacterium useful for mosquito control: a new report. Trop Biomed 31(1):97–109
Sharma R, Gupta R (2012) Coupled action of gamma-glutamyl transpeptidase-glutathione and keratinase effectively degrades feather keratin and surrogate prion protein, Sup 35NM. Bioresour Technol 120:314–317
Fang Z et al (2017) Keratinolytic protease: a green biocatalyst for leather industry. Appl Microbiol Biotechnol 101(21):7771–7779
Cheng SW et al (1995) Production and characterization of keratinase of a feather-degrading Bacillus licheniformis PWD-1. Biosci Biotechnol Biochem 59(12):2239–2243
Bernal C, Diaz I, Coello N (2006) Response surface methodology for the optimization of keratinase production in culture medium containing feathers produced by Kocuria rosea. Can J Microbiol 52(5):445–450
Lee H et al (2002) Characterization of a keratinolytic metalloprotease from Bacillus sp. SCB-3. Appl Biochem Biotechnol 97(2):123–133
Thys RC, Brandelli A (2006) Purification and properties of a keratinolytic metalloprotease from Microbacterium sp. J Appl Microbiol 101(6):1259–1268
Prakash P, Jayalakshmi SK, Sreeramulu K (2010) Purification and characterization of extreme alkaline, thermostable keratinase, and keratin disulfide reductase produced by Bacillus halodurans PPKS-2. Appl Microbiol Biotechnol 87(2):625–633
Cao ZJ et al (2009) Characterization of a novel Stenotrophomonas isolate with high keratinase activity and purification of the enzyme. J Ind Microbiol Biotechnol 36(2):181–188
Yamamura S et al (2002) Keratin degradation: a cooperative action of two enzymes from Stenotrophomonas sp. Biochem Biophys Res Commun 294(5):1138–1143
Fang Z et al (2013) Biodegradation of wool waste and keratinase production in scale-up fermenter with different strategies by Stenotrophomonas maltophilia BBE11-1. Bioresour Technol 140:286–291
Fang Z, Zang J, Du G, Chen J (2017) Rational protein engineering approaches to further improve the keratinolytic activity and thermostability of engineered keratinase KerSMD. Biochem Eng J 127:7
Ramnani P, Singh R, Gupta R (2005) Keratinolytic potential of Bacillus licheniformis RG1: structural and biochemical mechanism of feather degradation. Can J Microbiol 51(3):191–196
Liu QY et al (2014) Purification and characterization of four key enzymes from a feather-degrading Bacillus subtilis from the gut of tarantula Chilobrachys guangxiensis. Int Biodeterior Biodegradation 96:26–32
Tamura K et al (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28(10):2731–2739
Kaul S, Sumbali G (1997) Keratinolysis by poultry farm soil fungi. Mycopathologia 139(3):137–140
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227(5259):680–685
Olsen JV et al (2007) Higher-energy C-trap dissociation for peptide modification analysis. Nat Methods 4(9):709–712
Neilson KA et al (2011) Less label, more free: approaches in label-free quantitative mass spectrometry. Proteomics 11(4):535–553
Ribitsch D et al (2012) Extracellular serine proteases from Stenotrophomonas maltophilia: screening, isolation and heterologous expression in E. coli. J Biotechnol 157(1):140–147
Bhange K, Chaturvedi V, Bhatt R (2016) Feather degradation potential of Stenotrophomonas maltophilia KB13 and feather protein hydrolysate (FPH) mediated reduction of hexavalent chromium. 3 Biotech 6(1):42
Jankiewicz U, Larkowska E, Brzezinska MS (2016) Production, characterization, gene cloning, and nematocidal activity of the extracellular protease from Stenotrophomonas maltophilia N4. J Biosci Bioeng 121(6):614–618
Gouveia C et al (2008) Study on the bacterial midgut microbiota associated to different Brazilian populations of Lutzomyia longipalpis (Lutz & Neiva) (Diptera: Psychodidae). Neotrop Entomol 37(5):597–601
Hughes GL et al., Genome sequence of Stenotrophomonas maltophilia strain SmAs1, isolated from the Asian malaria mosquito Anopheles stephensi. Genome Announc (2016) 4(2)
Indiragandhi P et al (2007) Cultivable bacteria associated with larval gut of prothiofos-resistant, prothiofos-susceptible and field-caught populations of diamondback moth, Plutella xylostella and their potential for, antagonism towards entomopathogenic fungi and host insect nutrition. J Appl Microbiol 103(6):2664–2675
Morales-Jimenez J et al (2009) Bacterial community and nitrogen fixation in the red turpentine beetle, Dendroctonus valens LeConte (Coleoptera: Curculionidae: Scolytinae). Microb Ecol 58(4):879–891
Wang Z et al (2016) The optimization of fermentation conditions and enzyme properties of Stenotrophomonas maltophilia for protease production. Biotechnol Appl Biochem 63(2):292–299
Blow F, Vontas J, Darby AC (2016) Draft genome sequence of Stenotrophomonas maltophilia SBo1 isolated from Bactrocera oleae. Genome Announc 4(5)
Fang Z, Zhang J, Liu B, Du G, Chen J (2013) Biochemical characterization of three keratinolytic enzymes from Stenotrophomonas maltophilia BBE11-1 for biodegrading keratin wastes. Int Biodeterior Biodegradation 82:166–172
Parrado J et al (2014) Proteomic analysis of enzyme production by Bacillus licheniformis using different feather wastes as the sole fermentation media. Enzyme Microb Technol 57:1–7
Riessen S, Antranikian G (2001) Isolation of Thermoanaerobacter keratinophilus sp. nov., a novel thermophilic, anaerobic bacterium with keratinolytic activity. Extremophiles 5(6):399–408
Maranhao FC et al (2009) Membrane transporter proteins are involved in Trichophyton rubrum pathogenesis. J Med Microbiol 58(Pt 2):163–168
Jan AT (2017) Outer membrane vesicles (OMVs) of gram-negative bacteria: a perspective update. Front Microbiol 8:1053
Acknowledgements
This work was supported by grants from the Educational Department Foundation of Hunan Province (Grant No. 16A098), Key R & D project of Hunan Provincial Science and Technology Department (Grant No. 2017NK2311) and National Natural Science Foundation of China (Grant No. 31772865). We thank Dr. Fangjia Lu in Purdue University for the professional edit.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
All experimental protocols used in the study were carried out in accordance with the guidelines and regulations of College of Bioscience and Biotechnology, Hunan Agricultural University, China. The feathers were collected from a local poultry-processing store. Here, the poultry were slaughtered, dissected and after carcass evaluation, meat was intended for consumption. Therefore, our research does not require the approval of Animal Experimentation committee.
Rights and permissions
About this article
Cite this article
Qu, F., Chen, Q., Ding, Y. et al. Isolation of a feather-degrading strain of bacterium from spider gut and the purification and identification of its three key enzymes. Mol Biol Rep 45, 1681–1689 (2018). https://doi.org/10.1007/s11033-018-4311-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-018-4311-8