Abstract
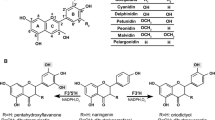
Dihydroflavonol 4-reductase (DFR), which catalyzes the reduction of dihydroflavonols to leucoanthocyanins, is a key enzyme in the biosynthesis of anthocyanidins, proanthocyanidins, and other flavonoids of importance in plant development and human nutrition. This study isolated a full length cDNA encoding DFR, designated as SmDFR (GenBank Accession No. EF600682), by screening a cDNA library from a red callus line of Saussurea medusa, which is an endangered, traditional Chinese medicinal plant with high pharmacological value. SmDFR was functionally expressed in yeast (Saccharomyces cerevisiae) to confirm that SmDFR can readily reduce dihydroquercetin (DHQ) and dihydrokampferol (DHK), but it could not reduce dihydromyricetin (DHM). The deduced SmDFR structure shared extensive sequence similarity with previously characterized plant DFRs and phylogenetic analysis showed that it belonged to the plant DFR super-family. SmDFR also possessed flavanone 4-reductase (FNR) activity and can catalyze the conversion of eridictyol to luteoforol. Real-time PCR analysis showed that the expression level of SmDFR was higher in flowers compared with both leaves and roots. This work greatly enhances our knowledge of flavonoid biosynthesis in S. medusa and marks a major advance that could facilitate future genetic modification of S. medusa.




Similar content being viewed by others
Abbreviations
- ANS:
-
Anthocyanidin synthase
- BA:
-
Benzyladenine
- CHI:
-
Chalcone isomerase
- DHK:
-
Dihydrokaempferol
- DHM:
-
Dihydromyricetin
- DHQ:
-
Dihydroquercetin
- DFR:
-
Dihydroflavonol 4-reductase
- HPLC-MS:
-
High performance liquid chromatography-mass spectrometry
- F3H:
-
Flavanone 3-hydroxylase
- F3′H:
-
Flavanonoid 3′ hydroxylase
- FNR:
-
Flavanone 4-reductase
- NAA:
-
Naphthaleneacetic Acid
- NADPH:
-
Reduced nicotinamide adenine dinucleotide phosphate
- ORF:
-
Open reading frame
- PAL:
-
Phenylalanine ammonia-lyase
- UTR:
-
Untranslated region
References
Li FX, Jin ZP, Zhao DX, Cheng LQ, Fu CX, Ma FS (2006) Overexpression of the Saussurea medusa chalcone isomerase gene in Saussurea involucrata hairy root cultures enhances their biosynthesis of apigenin. Phytochemistry 67:553–560
Law W, Salick J (2005) Human-induced dwarfing of Himalayan snow lotus, Saussurea laniceps (Asteraceae). Proc Natl Acad Sci USA 102:10218–10220
Takasaki M, Konoshima T, Komatsu K, Tokuda H, Nishino H (2000) Anti-tumor-promoting activity of lignans from the aerial part of Saussurea medusa. Cancer Lett 158:53–59
Zhao L, Wang X (2003) Research on chemical composition f pharmacology and its clinic application of Saussurea involucrata. J. Southwest National Coll 29:424–428
Fan CQ, Yue JM (2003) Biologically active phenols from Saussurea medusa. Bioorg Med Chem 11:703–708
Jia JM, Wu CF, Liu W, Yu H, Hao Y, Zheng JH, Ji YR (2005) Antiinflammatory and analgesic activities of the tissue culture of Saussurea involucrata. Biol Pharm Bull 28:1612–1614
Cheng LQ, Xu YJ, Grotewold E, Jin ZP, Wu FY, Fu CX, Zhao DX (2007) Characterization of Anthocyanidin Synthase (ANS) Gene and anthocyanidin in rare medicinal plant-Saussurea medusa. Plant Cell Tiss Organ Cult 89:63–73
Duan HQ, Takaishi Y, Momota H, Ohmoto Y, Taki T (2002) Immunosuppressive constituents from Saussurea medusa. Phytochemistry 59:85–90
Almeida JRM, D’Amico E, Preuss A, Carbone F, de Vos CHR, Deiml B, Mourgues F, Perrotta G, Fischer TC, Bovy AG, Martens S, Rosati C (2007) Characterization of major enzymes and genes involved in flavonoid and proanthocyanidin biosynthesis during fruit development in strawberry (Fragaria x ananassa). Arch Biochem Biophys 465:61–71
Forkmann G, Heller W (1999) Biosynthesis of flavonoids. Compr Nat Prod Chem 1:713–748
Harborne JB, Williams CA (2000) Advances in flavonoid research since 1992. Phytochemistry 55:481–504
Singh K, Kumar S, Yadav SK, Ahuja PS (2009) Characterization of dihydroflavonol 4-reductase cDNA in tea [Camellia sinensis (L.) O. Kuntze]. Plant Biotechnol Rep 3:95–101
Mayr U, Michalek S, Treutter D, Feucht W (1997) Phenolic compounds of apple and their relationship to scab resistance. J Phytopathol–Phytopathol Z 145:69–75
Khanizadeh S, Tsao R, Rekika D, Yang R, De Ell J (2007) Phenolic composition and antioxidant activity of selected apple genotypes. J Food Agricult Environ 5:61–66
Treutter D (2005) Significance of flavonoids in plant resistance and enhancement of their biosynthesis. Plant Biol 7:581–591
Fischer TC, Halbwirth H, Meisel B, Stich K, Forkmann G (2003) Molecular cloning, substrate specificity of the functionally expressed dihydroflavonol 4-reductases from Malus domestica and Pyrus communis cultivars and the consequences for flavonoid metabolism. Arch Biochem Biophys 412:223–230
Polashock JJ, Griesbach RJ, Sullivan RF, Vorsa N (2002) Cloning of a cDNA encoding the cranberry dihydroflavonol-4-reductase (DFR) and expression in transgenic tobacco. Plant Sci 163:241–251
Shimada N, Sasaki R, Sato S, Kaneko T, Tabata S, Aoki T, Ayabe S (2005) A comprehensive analysis of six dihydroflavonol 4-reductases encoded by a gene cluster of the Lotus japonicus genome. J Exp Bot 56:2573–2585
Forkmann G, Martens S (2001) Metabolic engineering and applications of flavonoids. Curr Opin Biotechnol 12:155–160
Martens S, Teeri T, Forkmann G (2002) Heterologous expression of dihydroflavonol 4-reductases from various plants. Febs Lett 531:453–458
Jin ZP, Grotewold E, Qu WQ, Fu GX, Zhao DX (2005) Cloning and characterization of a flavanone 3-hydroxylase gene from Saussurea medusa. DNA Seq 16:121–129
Xing JM, Zhao DX, Li MY, Ye HC, Li GF, Li ZH (1998) Cell growth and flavonoids production in suspension culture of Saussurea medusa. Acta Botanica Sinica 40:836–841
Zhao DX, Qiao CL, Wang Y (1998) Cell culture and selection of high flavonoids-producing cell lines in Saussurea medusa. Acta Botanica Sinica 40:515–520
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A, Speleman F (2002) Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 3:research 0034
Li HH, Flachowsky H, Fischer TC, Hanke MV, Forkmann G, Treutter D, Schwab W, Hoffmann T, Szankowski I (2007) Maize Lc transcription factor enhances biosynthesis of anthocyanins, distinct proanthocyanidins and phenylpropanoids in apple (Malus domestica Borkh.). Planta 226:1243–1254
Lo Piero AR, Puglisi I, Petrone G (2006) Gene characterization, analysis of expression and in vitro synthesis of dihydroflavonol 4-reductase from Citrus sinensis (L.) Osbeck. Phytochemistry 67:684–695
Xie DY, Jackson LA, Cooper JD, Ferreira D, Paiva NL (2004) Molecular and biochemical analysis of two cDNA clones encoding dihydroflavonol-4-reductase from Medicago truncatula. Plant Physiol 134:979–994
Petit P, Granier T, d’Estaintot BL, Manigand C, Bathany K, Schmitter JM, Lauvergeat V, Hamdi S, Gallois B (2007) Crystal structure of grape dihydroflavonol 4-reductase, a key enzyme in flavonoid biosynthesis. J Mol Biol 368:1345–1357
Treutter D, Feucht W (1990) The pattern of flavan-3-ols in relation to scab resistance of apple cultivars. J Horticult Sci 65:511–517
Knekt P, Jarvinen R, Seppanen R, Heliovaara M, Teppo L, Pukkala E, Aromaa A (1997) Dietary flavonoids and the risk of lung cancer and other malignant neoplasms. Am J Epidemiol 146:223–230
Hirpara KV, Aggarwal P, Mukherjee AJ, Joshi N, Burman AC (2009) Quercetin and its derivatives: synthesis, pharmacological uses with special emphasis on anti-tumor properties and prodrug with enhanced bio-availability. Anti-Cancer Ag Med Chem 9:138–161
Murakami A, Ashida H, Terao J (2008) Multitargeted cancer prevention by quercetin. Cancer Lett 269:315–325
Shi M, Wang FS, Wu ZZ (2003) Synergetic anticancer effect of combined quercetin and recombinant adenoviral vector expressing human wild-type p53, GM-CSF and B7-1 genes on hepatocellular carcinoma cells in vitro. World J Gastroenterol 9:73–78
Szankowski I, Flachowsky H, Li HH, Halbwirth H, Treutter D, Regos I, Hanke MV, Stich K, Fischer TC (2009) Shift in polyphenol profile and sublethal phenotype caused by silencing of anthocyanidin synthase in apple (Malus sp.). Planta 229:681–692
Acknowledgments
This work was supported by the National Science Foundation of China (No. 30873452; 30701089). The authors thank Dr. Duncan E. Jackson for critical review and comments to improve the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, H., Qiu, J., Chen, F. et al. Molecular characterization and expression analysis of dihydroflavonol 4-reductase (DFR) gene in Saussurea medusa . Mol Biol Rep 39, 2991–2999 (2012). https://doi.org/10.1007/s11033-011-1061-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-011-1061-2