Abstract
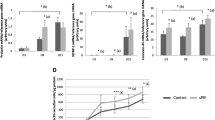
Recent evidence points to a stromal decidualization-like response in the pregnant porcine uterus. The objective of this study was to evaluate expression of tissue inhibitors of metalloproteinase-3 (TIMP-3), a sensitive indicator of endometrial stromal decidualization, in endometrium of pregnant sows and to further investigate this phenomenon. Real-time PCR, Western blot and immunostaining analysis were used to study TIMP-3 expression between/at attachment sites of endometrium of Days 13, 18 and 24 pregnant sows. The results indicate that TIMP-3 protein expression was lowest by Day 13 compared with Day 18 (P < 0.01) and 24 (P < 0.01), and the expression was higher at attachment sites than between attachment sites on Day 13 (P < 0.01) and 18 (P < 0.01). TIMP-3 intensive immunostaining was observed in stroma of endometrium on Days 13, 18 and 24, and the staining at attachment sites was stronger than between attachment sites. Collectively, these results suggest the crucial role of TIMP-3 in successful implantation and embryo survival and indicate the endometrial stromal decidualization-like in pigs.




Similar content being viewed by others
References
Kyriazakis I, Whittemore CT (2006) Reproduction whittemore’s science and practice of pig production. Blackwell Publishing, Oxford
Bischof P, Campana A (1996) A model for implantation of the human blastocyst and early placentation. Hum Reprod Update 2:262–270
Fisher SJ, Cui TY, Zhang L et al (1989) Adhesive and degradative properties of human placental cytotrophoblast cells in vitro. J Cell Biol 109:891–902
Damsky CH, Fitzgerald ML, Fisher SJ (1992) Distribution patterns of extracellular matrix components and adhesion receptors are intricately modulated during first trimester cytotrophoblast differentiation along the invasive pathway, in vivo. J Clin Invest 89:210–222
Jr Curry TE, Osteen KG (2003) The matrix metalloproteinase system: changes, regulation, and impact throughout the ovarian and uterine reproductive cycle. Endocr Rev 24:428–465
Reponen P, Leivo I, Sahlberg C et al (1995) 92-kDa type IV collagenase and TIMP-3, but not 72-kDa type IV collagenase or TIMP-1 or TIMP-2, are highly expressed during mouse embryo implantation. Dev Dyn 202:388–396
Alexander CM, Hansell EJ, Behrendtsen O (1996) Expression and function of matrix metalloproteinases and their inhibitors at the maternal-embryonic boundary during mouse embryo implantation. Development 122:1723–1736
Johnson GA, Burghardt RC, Joyce MM et al (2003) Osteopontin expression in uterine stroma indicates a decidualization-like differentiation during ovine pregnancy. Biol Reprod 68:1951–1958
Corner GW (1921) The ovarian cycle of swine. Science 53:420–421
Bjorkman N (1973) Fine structure of the fetal-maternal area of exchange in the epitheliochorial and endotheliochorial types of placentation. Acta Anat Suppl (Basel) 61:1–22
Jr Menino AR, Hogan A, Schultz GA et al (1997) Expression of proteinases and proteinase inhibitors during embryo-uterine contact in the pig. Dev Genet 21:68–74
Higuchi T, Kanzaki H, Nakayama H et al (1995) Induction of tissue inhibitor of metalloproteinase 3 gene expression during in vitro decidualization of human endometrial stromal cells. Endocrinology 136:4973–4981
Lin HC, Wang X, Liu GF, Fu JL, Wang AG (2007) Expression of alphaV and beta3 integrin subunits during implantation in pig. Mol Reprod Dev 74:1379–1385
Lord E, Murphy BD, Desmarais JA et al (2006) Modulation of peroxisome proliferator-activated receptor delta and gamma transcripts in swine endometrial tissue during early gestation. Reproduction 131:929–942
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C (T)) Method. Methods 25:402–408
Schmittgen TD, Livak KJ (2008) Analyzing real-time PCR data by the comparative C (T) method. Nat Protoc 3:1101–1108
Huang MC, Chen HY, Huang HC et al (2006) C2GnT-M is downregulated in colorectal cancer and its re-expression causes growth inhibition of colon cancer cells. Oncogene 25:3267–3276
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Leco KJ, Edwards DR, Schultz GA (1996) Tissue inhibitor of metalloproteinases-3 is the major metalloproteinase inhibitor in the decidualizing murine uterus. Mol Reprod Dev 45:458–465
Graham CH, Lala PK (1991) Mechanism of control of trophoblast invasion in situ. J Cell Physiol 148:228–234
Harvey MB, Leco KJ, Arcellana-Panlilio MY et al (1995) Proteinase expression in early mouse embryos is regulated by leukaemia inhibitory factor and epidermal growth factor. Development 121:1005–1014
Hurskainen T, Hoyhtya M, Tuuttila A, Oikarinen A, Autio-Harmainen H (1996) mRNA expressions of TIMP-1, -2, and -3 and 92-kD type IV collagenase in early human placenta and decidual membrane as studied by in situ hybridization. J Histochem Cytochem 44:1379–1388
Burghardt RC, Johnson GA, Jaeger LA et al (2002) Integrins and extracellular matrix proteins at the maternal–fetal interface in domestic animals. Cells Tissues Organs 172:202–217
Wynn RM (1974) Ultrastructural development of the human decidua. Am J Obstet Gynecol 118:652–670
Samuel CA, Perry JS (1972) The ultrastructure of pig trophoblast transplanted to an ectopic site in the uterine wall. J Anat 113:139–149
Cunha GR, Chung LW, Shannon JM, Taguchi O, Fujii H (1983) Hormone-induced morphogenesis and growth: role of mesenchymal-epithelial interactions. Recent Prog Horm Res 39:559–598
Cunha GR, Bigsby RM, Cooke PS, Sugimura Y (1985) Stromal-epithelial interactions in adult organs. Cell Differ 17:137–148
Guillomot M, Flechon JE, Wintenberger-Torres S (1981) Conceptus attachment in the ewe: an ultrastructural study. Placenta 2:169–182
Kirby DR (1965) Endocrinological effects of experimentally induced extra-uterine pregnancies in virgin mice. J Reprod Fertil 10:403–412
Denker HW (1993) Implantation: a cell biological paradox. J Exp Zool 266:541–558
Chai H, Yu DL, Zhang B, Fu Y, Hu SN (2009) Analysis of expressed sequence tags in porcine uterus tissue. Biochem Genet 47:117–125
Acknowledgments
The authors greatly appreciate Beijing Huadu Swine Breeding Company, LTD for collecting samples. This study was supported by National High-Technology Research Development Program of China (Nos. 2007AA10Z166 & 2008AA101008) and National Natural Science Foundation of China (No. 30771540).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Ren, Q., Guan, S., Fu, J. et al. Expression of tissue inhibitor of metalloproteinases-3 messenger RNA and protein in porcine endometrium during implantation. Mol Biol Rep 38, 3829–3837 (2011). https://doi.org/10.1007/s11033-010-0498-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-010-0498-z