Abstract
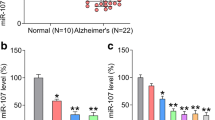
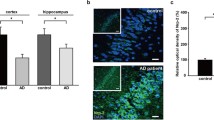
Accumulation of extracellular amyloid-β (Aβ) in hippocampal subregions is a hallmark of Alzheimer’s disease (AD), which promotes neuronal apoptosis, potentiates cognitive decline and play a causative role in AD pathogenesis. However, whether this process is controlled by distinct miRNAs at the posttranscriptional level remain fascinating but poorly understood. Using post mortem hippocampal samples from human AD patients and 3xTg-AD mouse, we demonstrate that miR-342-3p expression was significantly induced during the AD development. With the aid of intrahippocampal injection of miR-342-3p antagomir, we further show that in vivo miR-342-3p inhibition synergistically improved cognitive deficits in 3xTg-AD mice. The hippocampal Aβ-plaque burden in 3xTg-AD mice, as revealed by immunohistochemical analysis with 4G8 antibody, was attenuated also. Mechanistically, the upregulation of neuronal miR-342-3p is linked to an increase in the activation of the stress kinase c-Jun N-terminal kinase with the subsequent death of the neurons in Aβ-challenged HT22 hippocampal neuronal cells. These findings support the model that derangement of hippocampus signal transduction and subsequent neuronal apoptosis in AD arises as a consequence of increased Aβ burden and chronic activation of the JNK MAPK cascade in a miR-342-3p-dependent manner. Overall, we described for the first time the regulatory activity of miR-342-3p on relevant Aβ metabolism pathways in Alzheimer’s disease.




Similar content being viewed by others
References
Bhowmick S, D'Mello V, Abdul-Muneer PM (2018) Synergistic inhibition of ERK1/2 and JNK, not p38, phosphorylation ameliorates neuronal damages after traumatic brain injury. Mol Neurobiol. https://doi.org/10.1007/s12035-018-1132-7
Bonham LW et al (2018) Insulin-like growth factor binding protein 2 is associated with biomarkers of Alzheimer's disease pathology and shows differential expression in transgenic mice. Front Neurosci 12:476. https://doi.org/10.3389/fnins.2018.00476
Branca C et al (2017) Dyrk1 inhibition improves Alzheimer's disease-like pathology. Aging Cell 16:1146–1154. https://doi.org/10.1111/acel.12648
Chartoumpekis DV, Zaravinos A, Ziros PG, Iskrenova RP, Psyrogiannis AI, Kyriazopoulou VE, Habeos IG (2012) Differential expression of microRNAs in adipose tissue after long-term high-fat diet-induced obesity in mice. PLoS One 7:e34872. https://doi.org/10.1371/journal.pone.0034872
Coronas-Samano G, Portillo W, Beltran Campos V, Medina-Aguirre GI, Paredes RG, Diaz-Cintra S (2014) Deficits in odor-guided behaviors in the transgenic 3xTg-AD female mouse model of Alzheimers disease. Brain Res 1572:18–25. https://doi.org/10.1016/j.brainres.2014.05.013
Dong YS et al (2016) Unexpected requirement for a binding partner of the syntaxin family in phagocytosis by murine testicular Sertoli cells. Cell Death Differ 23:787–800. https://doi.org/10.1038/cdd.2015.139
El Fatimy R et al (2018) MicroRNA-132 provides neuroprotection for tauopathies via multiple signaling pathways. Acta Neuropathol 136:537–555. https://doi.org/10.1007/s00401-018-1880-5
Ferrante M, Conti GO (2017) Environment and neurodegenerative diseases: an update on miRNA. Role Microrna 6:157–165. https://doi.org/10.2174/2211536606666170811151503
Lei X, Lei L, Zhang Z, Cheng Y (2015) Downregulated miR-29c correlates with increased BACE1 expression in sporadic Alzheimer's disease. Int J Clin Exp Pathol 8:1565–1574
McLean FH et al (2018) Rapid and reversible impairment of episodic memory by a high-fat diet in mice. Sci Rep 8:11976. https://doi.org/10.1038/s41598-018-30265-4
Murakami K, McCaffrey TA, Livingstone MB (2013) Associations of dietary glycaemic index and glycaemic load with food and nutrient intake and general and central obesity in British adults. Br J Nutr 110:2047–2057. https://doi.org/10.1017/S0007114513001414
Muthaiyah B, Essa MM, Chauhan V, Chauhan A (2011) Protective effects of walnut extract against amyloid beta peptide-induced cell death and oxidative stress in PC12 cells. Neurochem Res 36:2096–2103. https://doi.org/10.1007/s11064-011-0533-z
Peng J, Andersen JK (2003) The role of c-Jun N-terminal kinase (JNK) in Parkinson's disease. IUBMB Life 55:267–271. https://doi.org/10.1080/1521654031000121666
Ren Y et al (2008) Endophilin I expression is increased in the brains of Alzheimer disease patients. J Biol Chem 283:5685–5691. https://doi.org/10.1074/jbc.M707932200
Savage MJ, Lin YG, Ciallella JR, Flood DG, Scott RW (2002) Activation of c-Jun N-terminal kinase and p38 in an Alzheimer's disease model is associated with amyloid deposition. J Neurosci 22:3376–3385. https://doi.org/10.1523/JNEUROSCI.22-09-03376.2002
Shah H et al (2016) Research priorities to reduce the global burden of dementia by 2025. Lancet Neurol 15:1285–1294. https://doi.org/10.1016/S1474-4422(16)30235-6
Tan L, Yu JT, Tan MS, Liu QY, Wang HF, Zhang W, Jiang T (2014) Genome-wide serum microRNA expression profiling identifies serum biomarkers for Alzheimer's disease. J Alzheimers Dis 40:1017–1027. https://doi.org/10.3233/JAD-132144
Wang L, Xu L, Xu M, Liu G, Xing J, Sun C, Ding H (2015) Obesity-associated MiR-342-3p promotes Adipogenesis of mesenchymal stem cells by suppressing CtBP2 and releasing C/EBPalpha from CtBP2 binding. Cell Physiol Biochem 35:2285–2298. https://doi.org/10.1159/000374032
Wang LL et al (2017) Profiling microRNA from brain by microarray in a transgenic mouse model of Alzheimer's disease. Biomed Res Int 2017:8030369. https://doi.org/10.1155/2017/8030369
Wen MM (2016) Getting miRNA therapeutics into the target cells for neurodegenerative diseases: a mini-review. Front Mol Neurosci 9:129. https://doi.org/10.3389/fnmol.2016.00129
Zhang C, Lai JH, Hu B, Zhang S, Zhao J, Li W (2014) A chromatin modifier regulates Sertoli cell response to mono-(2-ethylhexyl) phthalate (MEHP) via tissue inhibitor of metalloproteinase 2 (TIMP2) signaling. Biochim Biophys Acta 1839:1170–1182. https://doi.org/10.1016/j.bbagrm.2014.08.006
Zhu X, Castellani RJ, Takeda A, Nunomura A, Atwood CS, Perry G, Smith MA (2001) Differential activation of neuronal ERK, JNK/SAPK and p38 in Alzheimer disease: the 'two hit' hypothesis. Mech Ageing Dev 123:39–46
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Supplemental Table 1
(PDF 335 kb)
Supplemental Table 2
(PDF 169 kb)
Supplemental Fig. 1
Working protocols for the in vivo intrahippocampal injection study. (B) qPCR analyses of miR-342-3p expression in hippocampal tissues of 3xTg-AD mice two months after intrahippocampal injection with miR-342-3p antagomir (n = 10/group). (PDF 95 kb)
Supplemental Fig. 2
Transfection with miR-342-3p mimics or mimics-NC alone exhibits no effects on JNK activation. 48 h after transfection with miR-342-3p mimics or NC, HT22 cells were subjected to Western blot analysis of JNK, p-JNK, c-Jun and p-c-Jun expression levels. (PDF 169 kb)
Rights and permissions
About this article
Cite this article
Fu, Y., Hu, X., Zheng, C. et al. Intrahippocampal miR-342-3p inhibition reduces β-amyloid plaques and ameliorates learning and memory in Alzheimer’s disease. Metab Brain Dis 34, 1355–1363 (2019). https://doi.org/10.1007/s11011-019-00438-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-019-00438-9