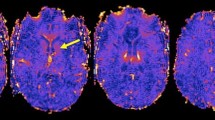
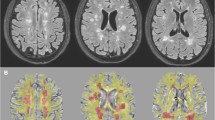
Abstract
Multiple sclerosis (MS) is a disorder related to myelin damage, which can be investigated by neuroimaging techniques such as fractional anisotropy (FA), a measure of microstructural white matter properties. The objectives of this study were to investigate (1) the relationship between FA and disability using an extremes of outcome approach, and (2) whether blood iron parameters were associated with FA and/or disability. Patients diagnosed with MS (n = 107; 14 males and 93 females) had iron parameter tests and disability determinations using the Expanded Disability Status Scale (EDSS). FA was recorded in 48 white matter tracts in 11 of the female patients with MS and 12 female controls. Results: In patients with high disability scores the mean FA was significantly lower (0.34 ± 0.067) than in the control group (0.45 ± 0.036; p = 0.04), while patients with low disability had mean FA values (0.44 ± 0.014) similar to controls (p = 0.5). Positive associations were found between FA and the iron parameters serum iron, ferritin and percentage transferrin saturation (%Tfsat) in all the white matter tracts. For % Tfsat, the associations were highly significant in 14 tracts (p < 0.01; r-values 0.74–0.84) and p < 0.001 (r = 0.83) in the superior fronto occipital fasciculus (LH). In the whole patient group a trend was found towards an inverse association between the EDSS and the %Tfsat (r = −0.26, p = 0.05) after excluding male gender and smoking as confounders, suggesting reduced disability in the presence of higher blood iron parameters. Additionally, significant inverse associations between disease duration and haemoglobin (p = 0.04) as well as %Tfsat (p = 0.02) suggested that patients with MS may experience a decrease in blood iron concentrations over time.




Similar content being viewed by others
References
Anderson G, Rodriguez M (2015) Multiple sclerosis: the role of melatonin and N-acetylserotonin. Mult Scler Relat Disord 4:112–123. https://doi.org/10.1016/j.msard.2014.12.001
Anderson GJ, Frazer DM, McLaren GD (2009) Iron absorption and metabolism. Curr Opin Gastroenterol 25:129–135. https://doi.org/10.1097/MOG.0b013e32831ef1f7
Asaf A, Evan S, Anat A (2015) Injury to white matter tracts in relapsing–remitting multiple sclerosis: A possible therapeutic window within the first 5 years from onset using diffusion-tensor imaging tract-based spatial statistics. Neuro image Clin 8:261–266. https://doi.org/10.1016/j.nicl.2015.04.020
Ascherio A, Munger K, White R, Köchert K, Simon K, Polman CH, Freedman MS, Hartung HP, Miller DH, Montalbán X, Edan G, Barkhof F, Pleimes D, Radü EW, Sandbrink R, Kappos L, Pohl C (2014) Vitamin D as an early predictor of multiple sclerosis activity and progression. JAMA Neurol 71:306. https://doi.org/10.1001/jamaneurol.2013.5993
Barnett M, Prineas J (2004) Relapsing and remitting multiple sclerosis: Pathology of the newly forming lesion. Ann Neurol 55:458–468. https://doi.org/10.1002/ana.20016
Bartzokis G (2011) Neuroglial pharmacology: white matter pathophysiologies and psychiatric treatments. Front Biosci 16:2695. https://doi.org/10.2741/3881
Bengtsson SL, Nagy Z, Skare S, Forsman L, Forssberg H, Ullén F (2005) Extensive piano practicing has regionally specific effects on white matter development. Nat Neurosci 8:1148–1150. https://doi.org/10.1038/nn1516
Bjartmar C, Wujek J, Trapp B (2003) Axonal loss in the pathology of MS: consequences for understanding the progressive phase of the disease. J Neurol Sci 206:165–171. https://doi.org/10.1016/S0022-510X(02)00069-2
Burgetova A, Seidl Z, Krasensky J, Horakova D, Vaneckova M (2010) Multiple sclerosis and the accumulation of iron in the Basal Ganglia: quantitative assessment of brain iron using MRI t(2) relaxometry. Eur Neurol 63:136–143. https://doi.org/10.1159/000279305
Cesar B, Dwyer MG, Shucard JL, Polak P, Bergsland N, Benedict RH, Weinstock-Guttman B, Shucard DW, Zivadinov R (2015) Cognitive and white matter tract differences in MS and diffuse neuropsychiatric systemic lupus erythematosus. AJNR Am J Neuroradiol 36:1874–1883. https://doi.org/10.3174/ajnr.A4354
Connor JR, Menzies SL (1996) Relationship of iron to oligondendrocytes and myelination. Glia 17:83–93. https://doi.org/10.1002/(SICI)1098-1136(199606)17:2<83::AID-GLIA1>3.0.CO;2-7
Curtiss F (2007) Pharmacoeconomic Modeling of Drug Therapies for Multiple Sclerosis - Are We Building Houses on Sand? J Manag Care Pharm 13:287–289. https://doi.org/10.18553/jmcp.2007.13.3.287
D’hooghe MB, Haentjens P, Nagels G, De Keyser J (2012) Alcohol, coffee, fish, smoking and disease progression in multiple sclerosis. Eur J Neurol 19:616–624. https://doi.org/10.1111/j.1468-1331.2011.03596.x
Davis W, van Rensburg SJ, Cronje FJ, Whati L, Fisher LR, van der Merwe L, Geiger D, Hassan MS, Matsha T, Erasmus RT, Kotze MJ (2014) The fat mass and obesity-associated FTO rs9939609 polymorphism is associated with elevated homocysteine levels in patients with multiple sclerosis screened for vascular risk factors. Metab Brain Dis 29:409–419. https://doi.org/10.1007/s11011-014-9486-7
DeLuca GC, Ramagopalan SV, Herrera BM, Dyment DA, Lincoln MR, Montpetit A, Pugliatti M, Barnardo MC, Risch NJ, Sadovnick AD, Chao M (2007) An extremes of outcome strategy provides evidence that multiple sclerosis severity is determined by alleles at the HLA-DRB1 locus. PNAS 104:20896–20901. https://doi.org/10.1073/pnas.0707731105
Evans RW, Kong X, Hider RC (2012) Iron mobilization from transferrin by therapeutic iron chelating agents. Biochim Biophys Acta 1820:282–290. https://doi.org/10.1016/j.bbagen.2011.11.007
Faivre A, Robinet E, Guye M, Rousseau C, Maarouf A, Le Troter A, Zaaraoui W, Rico A, Crespy L, Soulier E, Confort-Gouny S, Pelletier J, Achard S, Ranjeva JP, Audoin B (2016) Depletion of brain functional connectivity enhancement leads to disability progression in multiple sclerosis: A longitudinal resting-state fMRI study. Mult Scler 22:1695–1708. https://doi.org/10.1177/1352458516628657
Hametner S, Wimmer I, Haider L, Pfeifenbring S, Brück W, Lassmann H (2013) Iron and neurodegeneration in the multiple sclerosis brain. Ann Neurol 74:848–861. https://doi.org/10.1002/ana.23974
Hasan KM, Gupta RK, Santos RM, Wolinsky JS, Narayana PA (2005) Diffusion tensor fractional anisotropy of the normal-appearing seven segments of the corpus callosum in healthy adults and relapsing-remitting multiple sclerosis patients. J Magn Reson Imaging 21:735–743. https://doi.org/10.1002/jmri.20296
Herbert EP (2016) Magnetic resonance imaging and biochemical markers to assess disability in female subjects with multiple sclerosis. Dissertation, Cape Peninsula University of Technology
Jahanshad N, Kohannim O, Hibar DP, Stein JL, McMahon KL, de Zubicaray GI, Medland SE, Montgomery GW, Whitfield JB, Martin NG, Wright MJ, Toga AW, Thompson PM (2012) Brain structure in healthy adults is related to serum transferrin and the H63D polymorphism in the HFE gene. Proc Natl Acad Sci U S A 109:E851–E859. https://doi.org/10.1073/pnas.1105543109
Karim ME, Gustafson P, Petkau J, Zhao Y, Shirani A, Kingwell E, Evans C, van der Kop M, Oger J, Tremlett H (2014) Marginal structural Cox models for estimating the association between β-interferon exposure and disease progression in a multiple sclerosis cohort. Am J Epidemiol 180:160–171. https://doi.org/10.1093/aje/kwu125
Keser Z, Hasan KM, Mwangi B, Gabr RE, Steinberg JL, Wilken J, Wolinsky JS, Nelson FM (2017) Limbic Pathway Correlates of Cognitive Impairment in Multiple Sclerosis. J Neuroimaging 27:37–42. https://doi.org/10.1111/jon.12381
Khalil M, Langkammer C, Pichler A, Pinter D, Gattringer T, Bachmaier G, Ropele S, Fuchs S, Enzinger C, Fazekas F (2015) Dynamics of brain iron levels in multiple sclerosis: A longitudinal 3T MRI study. Neurology 84:2396–2402. https://doi.org/10.1212/WNL.0000000000001679
Koenig KA, Sakaie KE, Lowe MJ, Lin J, Stone L, Bermel RA, Beall EB, Rao SM, Trapp BD, Phillips MD (2013) High spatial and angular resolution diffusion-weighted imaging reveals forniceal damage related to memory impairment. Magn Reson Imaging 31:695–699. https://doi.org/10.1016/j.mri.2012.10.030
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis an expanded disability status scale (EDSS). Neurology 33:1444–1452. https://doi.org/10.1212/WNL.33.11.1444
Lane DJ, Merlot AM, Huang MH, Bae DH, Jansson PJ, Sahni S, Kalinowski DS, Richardson DR (2015) Cellular iron uptake, trafficking and metabolism: Key molecules and mechanisms and their roles in disease. Biochim Biophys Acta 1853:1130–1144. https://doi.org/10.1016/j.bbamcr.2015.01.021
Lassmann H (1998) Neuropathology in multiple sclerosis: new concepts. Mult Scler 4:93–98. https://doi.org/10.1177/135245859800400301
Lebel C, Gee M, Camicioli R, Wieler M, Martin W, Beaulieu C (2012) Diffusion tensor imaging of white matter tract evolution over the lifespan. NeuroImage 60:340–352. https://doi.org/10.1016/j.neuroimage.2011
Limson J, Nyokong T, Daya S (1998) The interaction of melatonin and its precursors with aluminium, cadmium, copper, iron, lead, and zinc: an adsorptive voltammetric study. J Pineal Res 24:15–21. https://doi.org/10.1111/j.1600-079X.1998.tb00361.x
Liu Y, Duan Y, He Y, Yu C, Wang J, Huang J, Ye J, Parizel PM, Li K, Shu N (2012) Whole brain white matter changes revealed by multiple diffusion metrics in multiple sclerosis: a TBSS study. Biomed Res Int 81:2826–2832. https://doi.org/10.1016/j.ejrad.2011.11.022
Llufriu S, Blanco Y, Martinez-Heras E, Casanova-Molla J, Gabilondo I, Sepulveda M, Falcon C, Berenguer J, Bargallo N, Villoslada P, Graus F (2012) Influence of corpus callosum damage on cognition and physical disability in multiple sclerosis: a multimodal study. PLoS One 7:e37167. https://doi.org/10.1371/journal.pone.0037167
Lochner C, Fouché JP, du Plessis S, Spottiswoode B, Seedat S, Fineberg N, Chamberlain SR, Stein DJ (2012) Evidence for fractional anisotropy and mean diffusivity white matter abnormalities in the internal capsule and cingulum in patients with obsessive-compulsive disorder. J Psychiatry Neurosci 37:193–199. https://doi.org/10.1503/jpn.110059
Lyksborg M, Siebner H, Sørensen P, Blinkenberg M, Parker G, Dogonowski GE, Larsen R, Dyrby TB (2014) Secondary progressive and relapsing remitting multiple sclerosis leads to motor-related decreased anatomical connectivity. PLoS One 9:e95540. https://doi.org/10.1371/journal.pone.0095540
Manouchehrinia A, Tench C, Maxted J, Bibani R, Britton J, Constantinescu C (2013) Tobacco smoking and disability progression in multiple sclerosis: United Kingdom cohort study. Brain 136:2298–2304. https://doi.org/10.1093/brain/awt139
McDonald WI, Compston A, Edan G, Goodkin D, Hartung HP, Lublin FD, McFarland HF, Paty DW, Polman CH, Reingold SC, Sandberg-Wollheim M, Sibley W, Thompson A, van den Noort S, Weinshenker BY, Wolinsky JS (2001) Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol 50:121–127. https://doi.org/10.1002/ana.1032
Moccia M, Lanzillo R, Palladino R, Maniscalco G, De Rosa A, Russo C, Massarelli M, Carotenuto A, Postiglione E, Caporale O, Triassi M, Brescia Morra V (2015) The Framingham cardiovascular risk score in multiple sclerosis. Eur J Neurol 22:1176–1183. https://doi.org/10.1111/ene.12720
Moremi K, van Rensburg SJ, Fisher LR, Davis W, Cronje FJ, Dashti M, Sefid J, Gamieldien J, Geiger D, Rensburg M, van Toorn R (2013) Association of an iron-related TMPRSS6 genetic variant c. 2207 C> T (rs855791) with functional iron deficiency and its effect on multiple sclerosis risk in the South African population. South African Journal of Psychiatry 19:120–121
Mori S, Wakana S, Nagae-Poetscher LM, van Zijl PC (2005) MRI atlas of human white matter. Elsevier, Amsterdam
Nelson MC, Isaacs F, Hassan MS, Kidd M, Cronje FJ, Van Rensburg SJ (2014) Prevalence of abnormal bloodflow patterns and effects of biochemistry and lifestyle factors on the major neck vessels in patients with Multiple Sclerosis in the Western Cape, South Africa. Medical Technology SA 28:28–33
Onu M, Roceanu A, Sboto-Frankenstein U, Bendic R, Tarta E, Preoteasa F, Bajenaru O (2012) Diffusion abnormality maps in demyelinating disease: correlations with clinical scores. Eur J Radiol 81:e386–e391. https://doi.org/10.1016/j.ejrad.2011.12.014
Phatlhane DV, Zemlin AE, Matsha TE, Hoffmann M, Naidoo N, Ichihara K, Smit F, Erasmus RT (2016) The iron status of a healthy South African adult population. Clin Chim Acta 460:240–245. https://doi.org/10.1016/j.cca.2016.06.019
Phillips O, Squitieri F, Sanchez-Castaneda C, Elifani F, Caltagirone C, Sabatini U, Di Paola M (2014) Deep white matter in Huntington's disease. PLoS One 9:e109676. https://doi.org/10.1371/journal.pone.0109676
Pittock SJ, Rodriguez M (2008) Benign multiple sclerosis. Curr Top Microbiol Immunol 318:1–7. https://doi.org/10.1007/978-3-540-73677-6_1
Rashid W, Hadjiprocopis A, Davies G, Griffin C, Chard D, Tiberio M, Altmann D, Wheeler-Kingshott C, Tozer D, Thompson A, Miller DH (2008) Longitudinal evaluation of clinically early relapsing-remitting multiple sclerosis with diffusion tensor imaging. J Neurol 255:390–397. https://doi.org/10.1007/s00415-008-0678-0
Rensvold JW, Krautkramer KA, Dowell JA, Denu JM, Pagliarini DJ (2016) Iron Deprivation Induces Transcriptional Regulation of Mitochondrial Biogenesis. J Biol Chem 291:20827–20837. https://doi.org/10.1074/jbc.M116.727701
Rodrigo L, Hernández-Lahoz C, Fuentes D, Alvarez N, López-Vázquez A, González S (2011) Prevalence of celiac disease in multiple sclerosis. BMC Neurol 11:31. https://doi.org/10.1186/1471-2377-11-31
Rouault TA, Zhang DL, Jeong SY (2009) Brain iron homeostasis, the choroid plexus, and localization of iron transport proteins. Metab Brain Dis 24:673–684. https://doi.org/10.1007/s11011-009-9169-y
Sbardella E, Tona F, Petsas N, Pantano P (2013) DTI measurements in multiple sclerosis: evaluation of brain damage and clinical implications. Mult Scler Int 2013:671730. https://doi.org/10.1155/2013/671730
Schmithorst VJ, Wilke M (2002) Differences in white matter architecture between musicians and non-musicians: a diffusion tensor imaging study. Neurosci Lett 321:57–60. https://doi.org/10.1016/S0304-3940(02)00054-X
Shirani A, Zhao Y, Karim ME, Evans C, Kingwell E, van der Kop ML, Oger J, Gustafson P, Petkau J, Tremlett H (2012) Association between use of interferon beta and progression of disability in patients with relapsing-remitting multiple sclerosis. JAMA 308:247–256. https://doi.org/10.1001/jama.2012.7625
Simpson IA, Ponnuru P, Klinger ME, Myers RL, Devraj K, Coe CL, Lubach GR, Carruthers A, Connor JR (2015) A novel model for brain iron uptake: introducing the concept of regulation. J Cereb Blood Flow Metab 35:48–57. https://doi.org/10.1038/jcbfm.2014.168
Smith SM, Jenkinson M, Woolrich MW, Beckmann CF, Behrens TE, Johansen-Berg H, Bannister PR, De Luca M, Drobnjak I, Flitney DE, Niazy RK, Saunders J, Vickers J, Zhang Y, De Stefano N, Brady JM, Matthews PM (2004) Advances in functional and structural MR image analysis and implementation as FSL. NeuroImage 23(Suppl 1):S208–S219. https://doi.org/10.1016/j.neuroimage.2004.07.051
Smith SM, Jenkinson M, Johansen-Berg H, Rueckert D, Nichols TE, Mackay CE, Watkins KE, Ciccarelli O, Cader MZ, Matthews PM, Behrens TE (2006) Tract-based spatial statistics: voxelwise analysis of multi-subject diffusion data. NeuroImage 31:1487–1505. https://doi.org/10.1016/j.neuroimage.2006.02.024
Swank RL, Dugan BB (1990) Effect of low saturated fat diet in early and late cases of multiple sclerosis. Lancet 336:37–39
Syka M, Keller J, Klempíř J, Rulseh AM, Roth J, Jech R, Vorisek I, Vymazal J (2015) Correlation between relaxometry and diffusion tensor imaging in the globus pallidus of Huntington's disease patients. PLoS One 10:e0118907. https://doi.org/10.1371/journal.pone.0118907.eCollection2015
Temel Ş, Kekliğkoğlu H, Vural G, Deniz O, Ercan K (2013) Diffusion tensor magnetic resonance imaging in patients with multiple sclerosis and its relationship with disability. Neuroradiol J 26:3–17
Tettey P, Simpson S, Taylor B, van der Mei I (2014) Vascular comorbidities in the onset and progression of multiple sclerosis. J Neurol Sci 347:23–33. https://doi.org/10.1016/j.jns.2014.10.020
Thouvenot E, Orsini M, Daures J, Camu W (2014) Vitamin D is associated with degree of disability in patients with fully ambulatory relapsing-remitting multiple sclerosis. Eur J Neurol 22:564–569. https://doi.org/10.1111/ene.12617
Todorich B, Zhang X, Connor JR (2011) H-ferritin is the major source of iron for oligodendrocytes. Glia 59:927–935. https://doi.org/10.1002/glia.21164
Trapp B, Bö L, Mörk S, Chang A (1999) Pathogenesis of tissue injury in MS lesions. J Neuroimmunol 98:49–56. https://doi.org/10.1016/S0165-5728(99)00081-8
Tremlett H, Zhu F, Petkau J, Oger J, Zhao Y (2012) Natural, innate improvements in multiple sclerosis disability. Mult Scler 18:1412–1421. https://doi.org/10.1177/1352458512439119
Unger EL, Earley CJ, Thomsen LL, Jones BC, Allen RP (2013) Effects of IV iron isomaltoside-1000 treatment on regional brain iron status in an iron-deficient animal. Neuroscience 246:179–185. https://doi.org/10.1016/j.neuroscience.2013.04.049
van Rensburg SJ, van Toorn R (2010) The controversy of CCSVI and iron in multiple sclerosis: is ferritin the key? Neurology 75:1581–1582. https://doi.org/10.1212/WNL.0b013e3181fb44f0
van Rensburg S, Kotze M, van Toorn R (2012) The conundrum of iron in multiple sclerosis – time for an individualised approach. Metab Brain Dis 27:239–253. https://doi.org/10.1007/s11011-012-9290-1
van Rensburg SJ, Davis W, Geiger D, Cronje FJ, Whati L, Kidd M, Kotze MJ (2015) Effects of diet, smoking and alcohol consumption on disability (EDSS) in people diagnosed with multiple sclerosis. S Afr J Psychiatr 21:121
van Rensburg SJ, van Toorn R, Moremi KE, Peeters AV, Oguniyi A, Kotze MJ (2016) Multiple sclerosis-like diagnosis as a complication of previously treated malaria in an iron and vitamin D deficient Nigerian patient. Metab Brain Dis 31:197–204. https://doi.org/10.1007/s11011-015-9788-4
van Toorn R, Schoeman JF, Solomons R, Rensburg MA, van Rensburg SJ (2010) Iron status in children with recurrent episodes of tumefactive cerebral demyelination. J Child Neurol 25:1401–1407. https://doi.org/10.1177/0883073810366179
Vollmann H, Ragert P, Conde V, Villringer A, Classen J, Witte OW, Steele CJ (2014) Instrument specific use-dependent plasticity shapes the anatomical properties of the corpus callosum: a comparison between musicians and non-musicians. Front Behav Neurosci 8:245. https://doi.org/10.3389/fnbeh.2014.00245
Weigel KJ, Lynch SG, LeVine SM (2014) Iron chelation and multiple sclerosis. ASN neuro 6:e00136. https://doi.org/10.1042/AN20130037
Weinberg ED (2009) Tobacco smoke iron: an initiator/promoter of multiple diseases. Biometals 22:207–210. https://doi.org/10.1007/s10534-008-9156-5
Williams R, Rohr AM, Wang WT, Choi IY, Lee P, Berman NE, Lynch SG, LeVine SM (2011) Iron deposition is independent of cellular inflammation in a cerebral model of multiple sclerosis. BMC Neurosci 12:59. https://doi.org/10.1186/1471-2202-12-59
World Medical Association Declaration of Helsinki (2013) http://www.ub.edu/recerca/Bioetica/doc/Declaracio_Helsinki_2013.pdf
Worthington V, Killestein J, Eikelenboom MJ, Teunissen CE, Barkhof F, Polman CH, Uitdehaag BM, Petzold A (2010) Normal CSF ferritin levels in MS suggest against etiologic role of chronic venous insufficiency. Neurology 75:1617–1622. https://doi.org/10.1212/WNL.0b013e3181fb449e
Xu B, Chasteen ND (1991) Iron oxidation chemistry in ferritin. Increasing Fe/O2 stoichiometry during core formation. J Biol Chem 266:19965–19970
Xu X, Wang Q, Zhong J, Zhang M (2015) Iron deposition influences the measurement of water diffusion tensor in the human brain: a combined analysis of diffusion and iron-induced phase changes. Neuroradiology 57:1169–1178. https://doi.org/10.1007/s00234-015-1579-4
Zeman D, Adam P, Kalistová H, Sobek O, Kelbich P, Andel J, Andel M (2000) Transferrin in patients with multiple sclerosis: a comparison among various subgroups of multiple sclerosis patients. Acta Neurol Scand 101:89–94. https://doi.org/10.1034/j.1600-0404.2000.101002089.x
Zhang T, Shirani A, Zhao Y, Karim ME, Gustafson P, Petkau J, Evans C, Kingwell E, van der Kop M, Zhu F, Oger J, Tremlett H, (2015) Beta-interferon exposure and onset of secondary progressive multiple sclerosis. European Journal Neurology 22(6):990–1000
Acknowledgements
We wish to thank Dr. Heloise Avenant for the EDSS measurements. We also thank Mr. Shafick Hassan for his involvement with the project. We gratefully acknowledge the financial support given by the National Research Foundation (NRF), Winetech, and the National Health Laboratory Service.
Funding
This study was funded by the National Research Foundation Award Number 2011 and Award Number CPR20110726000022168; Winetech Award Number N07/09/203 and the National Health Laboratory Service Award Number 94139.
Author information
Authors and Affiliations
Contributions
Performed the DTI and Collected the data: EH, CL. Analyzed the data: JPF, MK, CH. Wrote the paper: EH, SJVR. Interpreted the data: JPF, CH, SJVR, EH, MK, MJK. Revised the manuscript: PEH, MJK, MK, CH, CL.
Corresponding author
Ethics declarations
Disclosure of potential conflict of interest
Professors SJ van Rensburg and MJ Kotze are listed as inventors on patent number 2010/00058. Prof Kotze is also a director and shareholder of Gknowmix (Pty) Ltd. that has developed a database tool for research translation under the auspices of the Innovation Centre of the South African Medical Research Council. Prof SJ van Rensburg applied the Gknowmix database tool and open innovation genetic testing service delivery model for development of the Demyelinating Diseases GeneScreenTM. A treatment regimen was developed for application with this genomic solution (patent number 2012/07347). The remaining authors declared no conflict of interest. No writing assistance was utilised in the preparation of this manuscript.
Electronic supplementary material
ESM 1
(DOCX 54 kb)
Rights and permissions
About this article
Cite this article
Herbert, E., Engel-Hills, P., Hattingh, C. et al. Fractional anisotropy of white matter, disability and blood iron parameters in multiple sclerosis. Metab Brain Dis 33, 545–557 (2018). https://doi.org/10.1007/s11011-017-0171-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-017-0171-5