Abstract
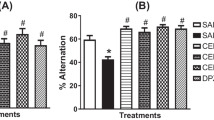
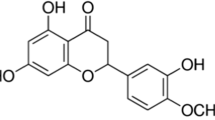
In Alzheimer’s disease (AD) basal forebrain cholinergic neurons appear to be targeted primarily in early stages of the disease. Scopolamine (muscarinic receptor antagonist) has been used for decades to induce working and reference memory impairment in rodents. In this study, we evaluated the protective effect of kolaviron, a biflavonoid complex isolated from Garcinia kola seeds extract against scopolamine-induced memory impairment/oxidative stress. Rats were pretreated with kolaviron (25, 50 or 100 mg/kg p.o.) for 3 consecutive days, scopolamine (3 mg/kg, i.p.) was administered 1 h post-treatment on day 3. Five minutes post-scopolamine injection, memory function was assessed using the Y-maze or Morris water maze tests (MWM) in rats. The rats were sacrificed and brains isolated on the 8th day after the MWM test for estimation of acetylcholinesterase activity and nitrosative/oxidative stress status. Scopolamine injection induced deficit (P < 0.05) in percentage alternation behaviour in the Y-maze test indicating memory impairment which was ameliorated by kolaviron in a dose-dependent manner. Also, pre-training treatment with kolaviron significantly improved spatial learning evidenced in the session-dependent and more efficient localization of the hidden platform in the MWM test. Moreover, scopolamine injection induced significant increase in lipid peroxidation (prefrontal cortex), nitrite generation (striatum and hippocampus) and a decrease in glutathione (prefrontal cortex, striatum and hippocampus) and superoxide dismutase (striatum and hippocampus) level which was attenuated by kolaviron pre-treatment. These findings showed that kolaviron possesses cognition enhancing effect through enhancement of antioxidant defense and cholinergic systems.








Similar content being viewed by others
Reference
Adaramoye OA (2010) Protective effect of kolaviron, a biflavonoid from Garcinia kola seeds, in brain of Wistar albino rats exposed to gamma-radiation. Biol Pharm Bull 33(2):260–266
Adaramoye OA, Adeyemi EO (2006) Hepatoprotection of D-galactosamine-induced toxicity in mice by purified fractions from Garcinia kola seeds. Basic Clin Pharmacol Toxicol 98(2):135–141
Akinmoladun AC, Akinrinola BL, Olaleye MT, Farombi EO (2015) Kolaviron, a Garcinia kola biflavonoid complex, protects against ischemia/reperfusion injury: pertinent mechanistic insights from biochemical and physical evaluations in rat brain. Neurochem Res 40(4):777–787
Blokland A (1995) Acetylcholine: a neurotransmitter for learning and memory? Brain Res Brain Res Rev 21(3):285–300
Bonda DJ, Wang X, Perry G, Nunomura A, Tabaton M, Zhu X, Smith MA (2010) Oxidative stress in Alzheimer disease: a possibility for prevention. Neuropharmacol 59(4–5):290–294
Budzynska B, Boguszewska-Czubara A, Kruk-Slomka M, Skalicka-Wozniak K, Michalak A, Musik I, Biala G (2015) Effects of imperatorin on scopolamine-induced cognitive impairment and oxidative stress in mice. Psychopharmacology 232(5):931–942
Cyril-Olutayo CM, Oladele AT, Elufioye TO (2012) Ethnobotanical survey of plants used as memory enhancer and antiaging in Ondo state, Nigeria. Int J Pharm 2(1):26–32
Ellman GL, Courtney KD, Andres V Jr., Feather-Stone RM. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 1961;7:88–95
Farombi EO, Nwaokeafor IA (2005) Anti-oxidant mechanisms of kolaviron: studies on serum lipoprotein oxidation, metal chelation and oxidative membrane damage in rats. Clin Exp Pharmacol Physiol 32(8):667–674
Farombi EO, Shrotriya S, Surh YJ (2009) Kolaviron inhibits dimethyl nitrosamine-induced liver injury by suppressing COX-2 and iNOS expression via NF-kB and AP-1. Life Sci 84:149–155
Fisher A (2012) Cholinergic modulation of amyloid precursor protein processing with emphasis on M1 muscarinic receptor: perspectives and challenges in treatment of Alzheimer’s disease. J Neurochem 120(Suppl 1):22–33
Green LC, Wagner DA, Glogowski J, Skipper PL, Wishnok JS, Tannenbaum SR (1982) Analysis of nitrate, nitrite, and [15N]nitrate in biological fluids. Anal Biochem 126(1):131–8
Hasselmo ME (2006) The role of acetylcholine in learning and memory. Curr Opin Neurobiol 16(6):710–715
Howes LG (2014) Cardiovascular effects of drugs used to treat Alzheimer’s disease. Drug Saf 37(6):391–395
Hritcu L, Clicinschi M, Nabeshima T (2007) Brain serotonin depletion impairs short-term memory, but not long-term memory in rats. Physiol Behav 91(5):652–657
Hritcu L, Foyet HS, Stefan M, Mihasan M, Asongalem AE, Kamtchouing P (2011) Neuroprotective effect of the methanolic extract of Hibiscus Asper leaves in 6-hydroxydopamine-lesioned rat model of Parkinson’s disease. J Ethnopharmacol 137:585–591
Ijomone OM, Obi AU (2013) Kolaviron, isolated from Garcinia kola, inhibits acetylcholinesterase activities in the hippocampus and striatum of Wistar rats. Ann Neurosci 20(2):42–46
Ishola IO, Tota S, Adeyemi OO, Agbaje EO, Narender T, Shukla R (2013) Protective effect of Cnestis ferruginea and its active constituent on scopolamine-induced memory impairment in mice: a behavioral and biochemical study. Pharm Biol 51(7):825–835
Iwu MM (1985) Antihepatoxic constituents of Garcinia kola seeds. Experientia 41(5):699–700
Iwu MM, Igboko OA, Onwuchekwa UA, Okunji CO (1987) Evaluation of the antihepatotoxic activity of the biflavonoids of Garcinia kola seed. J Ethnopharmacol 21(2):127–138
Klinkeberg I, Blokland A (2010) The validity of scopolamine as a pharmacological model for cognitive impairment: a review of animal behavioural studies. Neurosci Biobehav Rev 34(8):1307–1350
Kröger E, Van Marum R, Souverein P, Carmichael PH, Egberts T (2015) Treatment with rivastigmine or galantamine and risk of urinary incontinence: results from a Dutch database study. Pharmacoepidemiol Drug Saf 24(3):276–285
Lowry OH, Rosebrough NJ, Rarr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193(1):265–75
Mancuso C, Bates TE, Butterfield DA, Calafato S, Cornelius C, De Lorenzo A, Dinkova Kostova AT, Calabrese V (2007) Natural antioxidants in Alzheimer’s disease. Expert Opin Investig Drugs 16(12):1921–1931
Morris RG (1984) Development of a water maze procedure for studying spatial learning in the rat. J Neurosci Methods 11:47–60
Natalie IK, Heather M, Wilkins I, Linseman DA (2010) Nutraceutical antioxidants as novel neuroprotective agents. Mol 15:7B
Ohkawa H, Ohishi N, Yagi K (1979) Assay for lipid peroxides in animal tissues by thiobarbituric acid reaction. Anal Biochem 95(2):351–8
Olajide OJ, Enaibe BU, Bankole OO, Akinola OB, Laoye BJ, Ogundele OM (2016) Kolaviron was protective against sodium azide (NaN3) induced oxidative stress in the prefrontal cortex. Metab Brain Dis 31(1):25–35
Olaleye SB, Onasanwo SA, Ige AO, KK W, Cho CH (2010) Antiinflammatory activities of kolaviron-inhibition of nitric oxide, prostaglandin E2 and tumor necrosis factor-alpha production in activated macrophage-like cell line. Afr J Med Med Sci 39:41–46
Onasanwo SA, Velagapudi R, El-Bakoush A, Olajide OA (2016) Inhibition of neuroinflammation in BV2 microglia by the biflavonoid kolaviron is dependent on the Nrf2/ARE antioxidant protective mechanism. Mol Cell Biochem 414:23–36
Parihar MS, Hemnani T (2004) Alzheimer’s disease pathogenesis and therapeutic interventions. J Clin Neurosci 11(5):456–467
Rudi DP, De Deyn P (2001) Applications of the Morris water maze in the study of learning and memory. Brain Res Rev 36:60–90
Sarter M, Bodewitz G, Stephens DN (1988) Attenuation of scopolamine-induced impairment of spontaneous alteration behaviour by antagonist but not inverse agonist and agonist beta-carbolines. Psychopharmacology 94(4):491–495
Schliebs R, Arendt T (2011) The cholinergic system in aging and neuronal degeneration. Behav Brain Res 221(2):555–563
Sedlak J, Lindsay RH (1968) Estimation of total, protein-bound, and nonprotein sulfhydryl groups in tissue with Ellman’s reagent. Anal Biochem 25(1):192–205
Souza AC, Bruning CA, Acker CI, Neto JS, Nogueira CW (2013) 2-Phenylethynylbutyltellurium enhances learning and memory impaired by scopolamine in mice. Behav Pharmacol 24:249–254
Sultana M, Perluigi DA, Butterfield R (2013) Lipid peroxidation triggers neurodegeneration: a redox proteomics view into the Alzheimer disease brain. Free Radic Biol Med 62:157–169
Tabet N, Mantle D, Orrell M (2000) Free radicals as mediators of toxicity in Alzheimer’s disease: a review and hypothesis. Adverse drug reaction and toxicology. Review 19:127–152
Tariot PN, Federoff HJ (2003) Current treatment for Alzheimer’s disease and future prospects. Alzheimer’s Dis Assoc 17(suppl 4):S105–S113
Tota S, Goel R, Pachauri SD, Rajasekar N, Najmi AK, Hanif K, Nath C (2013) Effect of angiotensin II on spatial memory, cerebral blood flow, cholinergic neurotransmission, and brain derived neurotrophic factor in rats. Psychopharmacology 226(2):357–369
Uttara B, Singh AW, Zamboni P, Mahajan RT (2009) Oxidative stress and neurodegenerative diseases: a review of upstream and downstream antioxidant therapeutic options. Curr Neuropharmacol 7:65–74
Wang X, Wang W, Li L, Perry G, Lee HG, Zhu X (2014) Oxidative stress and mitochondrial dysfunction in Alzheimer’s disease. Biochim Biophys Acta 1842(8):1240–1247
Watkins PB, Zimmerman HJ, Knapp MJ, Gracon SI, Lewis KW (1994) Hepatotoxic effects of tacrine administration in patients with Alzheimer’s disease. JAMA 271(13):992–998
Wimo A, Jönsson L, Bond J, Prince M, Winblad B (2013) The worldwide economic impact of dementia 2010. Alzheimer disease international. Alzheimers Dement 9(1):1–11 e3
Winterbourn CC, Peskin AV, Parsons-Mair HN (2002) Thiol oxidase activity of copper, zinc superoxide dismutase. J Biol Chem 277(3):1906–1911
Acknowledgment
The authors are grateful to Mr. C. Micah of the Department of Pharmacology, Therapeutics and Toxicology, and Mr. S.A. Adenekan of the Department of Biochemistry, College of Medicine of the University of Lagos for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No conflict of interest to declare.
Rights and permissions
About this article
Cite this article
Ishola, I.O., Adamson, F.M. & Adeyemi, O.O. Ameliorative effect of kolaviron, a biflavonoid complex from Garcinia kola seeds against scopolamine-induced memory impairment in rats: role of antioxidant defense system. Metab Brain Dis 32, 235–245 (2017). https://doi.org/10.1007/s11011-016-9902-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-016-9902-2