Abstract
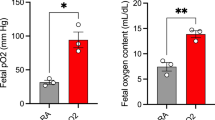
Increasing evidence indicates that fetal metabolic stress may result in a variety of post–natal perturbations during brain development. The goal of the study was to determine the duration of hypoxia/ischemia that would elicit a demonstrable regional depression of metabolism in the fetal brain and further to examine several end–points to determine if the metabolic stress affects the developing brain. The uterine artery and uterine branch of the ovarian artery were occluded with aneurysm clamps for a period of 45 min, the clips removed and the metabolites in five regions of the perinatal brain were measured at 0, 2 and 6 h of reflow. Regional P–creatine, ATP and glucose levels were significantly depleted at the end of the 45 min occlusion. The levels of glycogen and glutamate at the end of the occlusion indicated a decreasing trend which was not significant. The concentration of citrate remained essentially unchanged at the end of the occlusion. To ensure that the insult was not lethal to the tissue, the recovery of the metabolites was examined at 2 and 6 h of reflow and generally the concentrations of the high–energy phosphates and glucose were normal or near–normal by 6 h of reperfusion in the five regions of the brain examined. The changes in the metabolites indicate that 45 min of hypoxia/ischemia is an appropriate model for studying neonatal development after fetal metabolic stress.



Similar content being viewed by others
References
Altman J, Bayer S (1995) Atlas of prenatal rat brain development. CRC Press, Ann Arbor: pp. 389–387
Anderson ML, Wolpert SM, Kaye EM (1992) Vascular disease and trauma. In: Wolpert SM, Barnes PD (eds) MRI in pediatric neuroradiology, pp. 177–173
Andine P, Sandberg M, Bagenholm R, Lehmann A, Hagberg H (1991) Intra- and extra-cellular changes of amino acids in the cerebral cortex of the neonatal rat during hypoxic-ischemia. Brain Res Dev Brain Res 64:115–120
Blair E, Stanley F (1990) Intrauterine growth and spastic cerebral palsy. I. Association with birth weight for gestational age. Am J Obstet Gynecol 162:229–7
Bulterys M, Greenland S, Krau J (1990) Chronic fetal hypoxia and sudden infant death syndrome: Interaction between maternal smoking and low hematocrit during pregnancy. PedB 86:535–540
Burke RE, Kenyon N (1991) The effect of neonatal hypoxia—ischemia on striatal cholinergic neuropil: A quantitative morphologic analysis. Exp Neurol 113:63–73
Dwyer BE, Nishimura RN, Powell CL, Mailheau SL (1987) Focal protein synthesis inhibition in a model of neonatal hypoxic-ischemic brain injury. Exp Neurol 95:277–289
Dwyer BE, Nishimura RN, and Brown IR (1989) Synthesis of the major inducible heat shock protein in rat hippocampus after neonatal hypoxia-ischemia. Exp Neurol 104:28–31
Ferriero DM, Arcavi LJ, Sagar SM, McIntosh TK, Simon RP (1988) Selective sparing of NADPH—diaphorase neurons in neonatal hypoxia-ischemia. Ann Neurol 24:670–676
Ferriero DM, Soberano HQ, Simon RP, Sharp FR (1990) Hypoxia-ischemia induces heat shock protein—like (HSP72) immunoreactivity in neonatal rat brain. Brain Res Dev Brain Res 53:145–150
Lindahl E, Michelson K, Helenius M, Parre M (1988) Neonatal risk factors and later neurodevelopmental disturbances. Dev Med Child Neurol 30:571–589
Lowry O, Passonneau J (1972). A flexible system of enzymatic ananlysis. Academic Press Inc., New York
Lust WD, Feussner G, Barbehenn E et al (1981) The enzymatic measurement of adenine nucleotides and P-creatine in picomole amounts. Anal Biochem 110:258–266
Lust WD, Pundik S, Zechel J, Zhou Y, Buczek M, Selman WR (2003) Changing metabolic and energy profiles in fetal, neonatal, and adult rat brain. Metab Brain Dis 18:195–206
Mann LI (1986) Pregnancy events and brain damage. Am J Obstet Gynecol 155:6–9
Naeye R, Peters E. (1987) Antenatal hypoxia and low IQ values. Am J Dis Child 141:50–54
Nehlig A, Pereira de Vasconcelos A (1993) Glucose and ketone body utilization by the brain of neonatal rats. Prog Neurobiol 40:163–221
Nehlig A (1996) Respective roles of glucose and ketone bodies as substrates for cerebral energy metabolism in the suckling rat. Dev Neurosci 18:426–433
Nelson K (1989) Relationship of intrapartum and delivery room events to long-term neurologic outcome. Clin Perinatol 16:995–1007
Papile LA (1992) Central nervous disturbances, part 4. In: Fanaroff AA, Martin MJ (eds) Neonatal-perinatal medicine-diseases of the fetus and infant. Mosby-Year Book, Inc, pp. 719–728
Rizzo N, Gabrielli S, Perolo A, Pilu G, Cacciari A, Domini R, Bovicelli L (1989) Prenatal diagnosis and management of fetal ovarian cysts. Prenat Diagn 9:97–103
Robinson S, Petelenz K., Li Q, Cohen ML, Dechant A, Tabrizi N, Bucek M, Lust D, Miller RH (2005) Developmental changes induced by graded prenatal systemic hypoxic—ischemic insults in rats. Neurobiol Dis 18:568–581
Squire M., Keeling J (1991) The incidence of prenatal brain injury. Neuropathol Appl Neurobiol 17:29–38
Uvebran P, Hagberg G (1992) Intrauterine growth in children with cerebral palsy. Acta Paediatric 81:407–412
Acknowledgements
Study supported in part by R01 NS 32683 and Philip Morris External Research Program.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pundik, S., Robinson, S., Lust, W.D. et al. Regional metabolic status of the E–18 rat fetal brain following transient hypoxia/ischemia. Metab Brain Dis 21, 309–317 (2006). https://doi.org/10.1007/s11011-006-9031-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-006-9031-4