Abstract
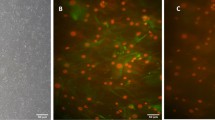
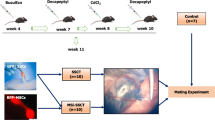
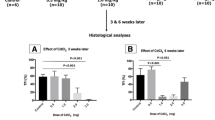
Currently, azoospermia is one of the most common diseases of male infertility. Stem cell research is the new hope for novel therapy with a higher degree of safety and lower cost. This study aimed to investigate the effect of umbilical cord blood-derived stem cells (” and mesenchymal “UCB-MSCs”) and mono-cell layer implanted into the induced azoospermic mice testis. Stem cells were isolated from umbilical cord blood and CD34+ve cells were separated from negative one by Mini MACs column. At 5th week after single injection of busulfan, stained mesenchymal (CD34-ve), hematopoietic stem cells (CD34+ve) and their conjugate (mono-cell layer) were injected locally into testis. At the end of the study, MSCs group showed that mRNA levels of genes related to meiosis (Vasa, SCP3, and PgK2) were increased with significant decrease of FSH and LH levels, compared to control group. Histologically, most of the tubules restored normal architecture. In contrast, HSCs and mono-cell layer groups showed statically insignificant change of FSH, LH, and gene expression, compared to control group. Histologically, distorted seminiferous tubules, with reduction in sperm content, and interstitial mononuclear cellular infiltration were seen. There was significant increase in the optical density of PCNA immune reaction in MSCs group than azoospermia, HSCs, and mono-cell layer, while there was non-significant difference between MSCs and control group. The present study suggested that injection of MSCs into chemotherapeutic-induced azoospermia in mice improved testicular failure; histologically and functionally, by restoration of spermatogenic gene expression while HSC and mono-cell layer showed no effect on spermatogenesis added to that mono-cell layer may induce testicular tissue damage.






Similar content being viewed by others
References
Harton GL, Tempest HG (2012) Chromosomal disorders and male infertility. Asian J Androl 14:32–39
Silber SJ, Barbey N (2012) Scientific molecular basis for treatment of reproductive failure in the human: an insight into the future. Biochim Biophys Acta 1822.
Joyce N, Annett G, Wirthlin L, Olson S, Bauer G (2010) Mesenchymal stem cells for the treatment of neurodegenerative disease. Regen Med 5:933–946
Martino G, Franklin RJ, Baron Van Evercooren A, Kerr DA (2010) Stem Cells in Multiple Sclerosis (STEMS) Consensus Group. Stem cell transplantation in multiple sclerosis:current status and future prospects. Nat. Rev Neurol 6:247–255
Jacobs SA, Roobrouck VD, Verfaillie CM, Goo V (2013) Immunological characteristics of human mesenchymal stem cells and multipotent adult progenitor cells. Immunol Cell Biol 91:32–39
Sanders JE, Hawley J, Levy W, Gooley T, Buckner CD, Deeg HJ, Doney K, Storb R, Sullivan K, Witherspoon R, Appelbaum FR (1996) Pregnancies following high-dose cyclophosphamide with or without high-dose busulfan or total-body irradiation and bone marrow transplantation. Blood 87:3045–3052
Heller RH, Jones HW (1964) Production of ovarian dysgenesis in the rat and human by busulphan. Am J Obstet Gynecol 89:414–420
Saffman EE, Lasko P (1999) Germline development in vertebrates and invertebrates. Cell Mol Life Sci 55(8–9):1141–1163
Xue-Xia Liu X, Zhang H, Shen X, Liu F, Liu J, Wang W (2016) Characteristics of testis-specific phosphoglycerate kinase 2 and its association with human sperm quality. Hum Reprod 31:273–279
Sage J, Martin L, Rassoulzadegan M, Cuzin F (1989) Germ cell-specific enhancer activity of a repeated element in a variable region of the mouse genome. Gene 9(1):85–92
Duangporn J, Moira K (2010) A Mouse models in male fertility research. Asian J Androl 13(1):139–151·
Schuler US, Renner UD, Kroschinsky F, Johne C, Jenke A et al (2001) Intravenous busulphan for conditioning before autologous or allogeneic human blood stem cell transplantation. Br J Haematol 114:944–950
Yang R, Liu T, Zhao K, Xiong C (2014) Enhancement of mouse germ cell-associated genes expression by injection of human umbilical cord mesenchymal stem cells into the testis of Chemical-induced azoospermic mice. Asian J Androl 16: 1–7.
Giarratana MC et al (2005) Ex vivo generation of fully mature human red blood cells from hematopoietic stem cells. Nat Biotechnol 23:69–74
Secunda R, Vennila R, Mohanashankar AM, Rajasundari M, Jeswanth S, Surendran R (2015) Isolation, expansion and characterisation of mesenchymal stem cells from human bone marrow, adipose tissue, umbilical cord blood and matrix: a comparative study. CytoTechnology 67:793–807
Bancroft J, Layton C (2014) The Hematoxylin and eosin. In: Suvarna SK, Layton C and Bancroft JD (ed). Theory and Practice of histological techniques. 7th ed. Churchill Livingstone of El Sevier, Philadelphia Ch. 10 and 11 pp, 172–214.
Elias JM, Margiotta M, Gaborc D (1989) Sensitivity and detection efficiency of the peroxidase antiperoxidase (PAP), avidin–biotin peroxidase complex (ABC) and peroxidase-labeled avidin–biotin (LAB) methods. Am J Clin Pathol 92(1):62–67
McLaren A (2000) Germ and somatic cell lineages in the developing gonad. Mol Cell Endocrinol 163: 3–9.
Gilbert S (2010) Developmental biology. 8th Ed, USA: Sinauer Associates Inc.
Yazawa T, Mizutani T, Yamada K, Kawata H, Umezawa K, Miyamoto K, Sekiguchi T, Yoshino M, Kajitani T, Shou Z, Umezawa A, Miyamoto K (2006) Differentiation of adult stem cells derived from bone marrow stroma into Leydig or adrenocortical cells. Endocrinology 147:4104–4111
Sabbaghi MA, Bahrami AR, Feizzade B, Kalantar SM, et al (2012) Trial evaluation of bone marrow derived mesenchymal stem cells (MSCs) transplantation in revival of spermatogenesis in testicular torsion. Middle East Fertil Soc J 17: 243–249.
Nayernia K, Lee JH, Drusenheimer N, Nolte J, Wulf G, Dressel R, Gromoll J, Engel W(2006) Derivation of male germ cells from bone marrow stem cells. Lab Invest 86: 654–63
Drusenheimer N, Wulf G, Nolte J et al (2007) Putative human male germ cells from bone marrow stem cells. Soc Reprod Fertil Suppl 63:69–76
Cakici C, Buyrukcu B, Duruksu G, Haliloglu AH, Aksoy A, Isik A, Uludag O, Ustun H, Subasi C (2013) Recovery of fertility in azoospermia rats after injection of adipose-tissue derived mesenchymal stem cells: the sperm generation. Biomed Res. 529–589
Lue Y, Erkkila K, Liu PY, Ma K, Wang C, Hikim AS, Swerdloff RS (2007) Fate of bone marrow stem cells transplanted into the testis: potential implication for men with testicular failure. Am J Pathol 170:899–908
Liu FH, Yang DZ, Wang YF, Liang XP, Peng WM, Cao CA, Chen XG, Guo ZM (2007) Mesenchymal stem cells do not differentiate into “quasi-sperm”. Nat J Androl 13(4):309–311.
Van Saen D, Goossens E, De Block G, Tournaye H (2009) Bone marrow stem cells transplanted to the testis of sterile mice do not differentiate into spermatogonial stem cells and have no protective effect on fertility. Fertil Steril 91:1549–1152
Meistrich, ML, Kangasniemi M (1997) Treatment after irradiation stimulates recovery of rat spermatogenesis from surviving spermatogonia. Andrology 18: 80–87.
Schrader M, Muller M, Straub B, Miller K (2001) The impact of chemotherapy on male fertility: a survey of the biologic basis and clinical aspects. Reprod Toxicol 15:611
Fan QR, Hendrickson WA (2005) Structure of human follicle-stimulating hormone in complex with its receptor. Nature 433:269–277
Xie L, Lin L, Tang Q, Li W, Huang T, Huo X, Liu X, Jiang J (2015) Sertoli cell-mediated differentiation of male germ cell-like cells from human umbilical cord Wharton’s jelly-derived mesenchymal stem cells in an in vitro co-culture system. Eur J Med Res 20:9
Ebata KT, Yeh JR, Zhang X, Nagano MC (2011) Soluble growth factors stimulate spermatogonial stem cell divisions that maintain a stem cell pool and produce progenitors. Cell Res 317:1319–1329
Ma L, Zhou Z, Zhang D, Yang S, Wang J et al (2012) Immunosuppressive function of mesenchymal stem cells from human umbilical cord matrix in immune thrombocytopenia patients. Thromb Haemost 107:937–950
Zahkook SAM, Atwa A, Shahat MM, Mansour AM, Bakry S (2014) Mesenchymal stem cells restore fertility in induced azoospermic rats following chemotherapy administration. J Reproduc Infertil 5(2):50–57
Monsefi M, Fereydouni B, Rohani L, Talae T (2013) Mesenchymal stem cells repair germinal cells of seminiferous tubules of sterile rats. Iran. J Reprod Med 11:537–544
Easley CA, Phillips BT, McGuire MM, Barringer JM, Valli H, Hermann BP, Simerly CR, Rajkovic A, Miki T, Orwig KE, Schatten GP (2012) Direct Differentiation of Human Pluripotent Stem Cells into Haploid Spermatogenic Cells. Cell Rep 27:440–446.
Horn MM, Paz AH, Duarte ME, Baldo G, Belardinelli MC, Matte U, Lima EO, Passos EP (2008) Germinative testicular cells and bone marrow mononuclear cells transplanted to a rat model of testicular degeneration. Cloning Stem Cells 10:543–546
Lee IC, Wang JH, Lee YT, Young TH (2007) The differentiation of mesenchymal stem cells by mechanical stress or/and co-culture system. Biochem Biophys Res Commun 352:147–152
Ball SG, Shuttleworth AC, Kielty CM (2004) Direct cell contact influences bone marrow mesenchymal stem cell fate”. Int J Biochem Cell Biol 36:714–727
D’Andrea MR, Lawrence D, Nagele RG, Wang CY, Damiano BP (2008) PCNA indexing as a preclinical immunohistochemical biomarker for testicular toxicity. Biotech Histochem 83:211–220
Chapman DL, Wolgemuth DJ (1994) Expression of proliferating cell nuclear antigen in the mouse germ line and surrounding somatic cells suggests both proliferation-dependent and -independent modes of function. Int J Dev Biol 38:491–497
Acknowledgement
No source of funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Abd Allah, S.H., Pasha, H.F., Abdelrahman, A.A. et al. Molecular effect of human umbilical cord blood CD34-positive and CD34-negative stem cells and their conjugate in azoospermic mice . Mol Cell Biochem 428, 179–191 (2017). https://doi.org/10.1007/s11010-016-2928-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-016-2928-2