Abstract
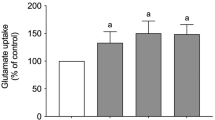
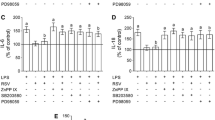
Resveratrol is a dietary polyphenol that displays neuroprotective properties in several in vivo and in vitro experimental models, by modulating oxidative and inflammatory responses. Glutathione (GSH) is a key antioxidant in the central nervous system (CNS) that modulates several cellular processes, and its depletion is associated with oxidative stress and inflammation. Therefore, this study sought to investigate the protective effects of resveratrol against GSH depletion pharmacologically induced by buthionine sulfoximine (BSO) in C6 astroglial cells, as well as its underlying cellular mechanisms. BSO exposure resulted in several detrimental effects, decreasing glutamate-cysteine ligase (GCL) activity, cystine uptake, GSH intracellular content and the activities of the antioxidant enzymes glutathione peroxidase (GPx) and glutathione reductase (GR). Moreover, BSO increased reactive oxygen/nitrogen species (ROS/RNS) levels and pro-inflammatory cytokine release. Resveratrol prevented these effects by protecting astroglial cells against BSO-induced cytotoxicity, by modulating oxidative and inflammatory responses. Additionally, we observed that pharmacological inhibition of heme oxygenase 1 (HO-1), an essential cellular defense against oxidative and inflammatory injuries, abolished all the protective effects of resveratrol. These observations suggest HO-1 pathway as a cellular effector in the mechanism by which resveratrol protects astroglial cells against GSH depletion, a condition that may be associated to neurodegenerative diseases.





Similar content being viewed by others
References
Baur JA, Sinclair DA (2006) Therapeutic potential of resveratrol: the in vivo evidence. Nat Rev Drug Discov 5:493–506. doi:10.1038/nrd2060
Bhullar KS, Hubbard BP (2015) Lifespan and healthspan extension by resveratrol. Biochim Biophys Acta 1852:1209–1218. doi:10.1016/j.bbadis.2015.01.012
Bellaver B, Souza DG, Souza DO, Quincozes-Santos A (2014) Resveratrol increases antioxidant defenses and decreases proinflammatory cytokines in hippocampal astrocyte cultures from newborn, adult and aged Wistar rats. Toxicol Vitro Int J Publ Assoc BIBRA 28:479–484. doi:10.1016/j.tiv.2014.01.006
Delmas D, Jannin B, Latruffe N (2005) Resveratrol: preventing properties against vascular alterations and ageing. Mol Nutr Food Res 49:377–395. doi:10.1002/mnfr.200400098
Quincozes-Santos A, Gottfried C (2011) Resveratrol modulates astroglial functions: neuroprotective hypothesis: Resveratrol modulates astroglial functions. Ann N Y Acad Sci 1215:72–78. doi:10.1111/j.1749-6632.2010.05857.x
Sakata Y, Zhuang H, Kwansa H et al (2010) Resveratrol protects against experimental stroke: putative neuroprotective role of heme oxygenase 1. Exp Neurol 224:325–329. doi:10.1016/j.expneurol.2010.03.032
Rege SD, Geetha T, Griffin GD, et al (2014) Neuroprotective effects of resveratrol in Alzheimer disease pathology. Front Aging Neurosci 6:218. doi:10.3389/fnagi.2014.00218
Ferretta A, Gaballo A, Tanzarella P et al (2014) Effect of resveratrol on mitochondrial function: implications in parkin-associated familiar Parkinson’s disease. Biochim Biophys Acta 1842:902–915. doi:10.1016/j.bbadis.2014.02.010
Bastianetto S, Ménard C, Quirion R (2015) Neuroprotective action of resveratrol. Biochim Biophys Acta 1852:1195–1201. doi:10.1016/j.bbadis.2014.09.011
Bastianetto S, Quirion R (2010) Heme oxygenase 1: another possible target to explain the neuroprotective action of resveratrol, a multifaceted nutrient-based molecule. Exp Neurol 225:237–239. doi:10.1016/j.expneurol.2010.06.019
Kulkarni SS, Cantó C (2015) The molecular targets of resveratrol. Biochim Biophys Acta 1852:1114–1123. doi:10.1016/j.bbadis.2014.10.005
Quincozes-Santos A, Bobermin LD, Latini A et al (2013) Resveratrol protects C6 Astrocyte cell line against hydrogen peroxide-induced oxidative stress through Heme oxygenase 1. PLoS One 8:e64372. doi:10.1371/journal.pone.0064372
Motterlini R, Foresti R (2014) Heme oxygenase-1 as a target for drug discovery. Antioxid Redox Signal 20:1810–1826. doi:10.1089/ars.2013.5658
Scapagnini G, Butterfield DA, Colombrita C et al (2004) Ethyl ferulate, a lipophilic polyphenol, induces HO-1 and protects rat neurons against oxidative stress. Antioxid Redox Signal 6:811–818. doi:10.1089/ars.2004.6.811
Loboda A, Damulewicz M, Pyza E, et al (2016) Role of Nrf2/HO-1 system in development, oxidative stress response and diseases: an evolutionarily conserved mechanism. Cell Mol Life Sci CMLS 73:3221–3247. doi:10.1007/s00018-016-2223-0
Wakabayashi N, Slocum SL, Skoko JJ et al (2010) When NRF2 talks, who’s listening? Antioxid Redox Signal 13:1649–1663. doi:10.1089/ars.2010.3216
Dringen R (2000) Metabolism and functions of glutathione in brain. Prog Neurobiol 62:649–671
Dringen R, Brandmann M, Hohnholt MC, Blumrich E-M (2015) Glutathione-dependent detoxification processes in Astrocytes. Neurochem Res 40:2570–2582. doi:10.1007/s11064-014-1481-1
Hertz L, Zielke HR (2004) Astrocytic control of glutamatergic activity: astrocytes as stars of the show. Trends Neurosci 27:735–743. doi:10.1016/j.tins.2004.10.008
Lu SC (2013) Glutathione synthesis. Biochim Biophys Acta 1830:3143–3153. doi:10.1016/j.bbagen.2012.09.008
Lewerenz J, Hewett SJ, Huang Y et al (2013) The cystine/glutamate antiporter system x(c)(-) in health and disease: from molecular mechanisms to novel therapeutic opportunities. Antioxid Redox Signal 18:522–555. doi:10.1089/ars.2011.4391
Seib TM, Patel SA, Bridges RJ (2011) Regulation of the system x(C)- cystine/glutamate exchanger by intracellular glutathione levels in rat astrocyte primary cultures. Glia 59:1387–1401. doi:10.1002/glia.21176
Niture SK, Khatri R, Jaiswal AK (2014) Regulation of Nrf2-an update. Free Radic Biol Med 66:36–44. doi:10.1016/j.freeradbiomed.2013.02.008
Bobermin LD, Hansel G, Scherer EBS, et al (2015) Ammonia impairs glutamatergic communication in astroglial cells: protective role of resveratrol. Toxicol Vitro Int J Publ Assoc BIBRA 29:2022–2029. doi:10.1016/j.tiv.2015.08.008
de Almeida LMV, Piñeiro CC, Leite MC et al (2007) Resveratrol increases glutamate uptake, glutathione content, and S100B secretion in cortical astrocyte cultures. Cell Mol Neurobiol 27:661–668. doi:10.1007/s10571-007-9152-2
Bellaver B, Bobermin LD, Souza DG, et al (2016) Signaling mechanisms underlying the glioprotective effects of resveratrol against mitochondrial dysfunction. Biochim Biophys Acta BBA 1862:1827–1838. doi:10.1016/j.bbadis.2016.06.018
Souza DG, Bellaver B, Souza DO, Quincozes-Santos A (2013) Characterization of adult rat astrocyte cultures. PloS One 8:e60282. doi:10.1371/journal.pone.0060282
Schreiner B, Romanelli E, Liberski P, et al (2015) Astrocyte depletion impairs redox homeostasis and triggers neuronal loss in the adult CNS. Cell Rep 12:1377–1384. doi:10.1016/j.celrep.2015.07.051
Bakshi R, Zhang H, Logan R et al (2015) Neuroprotective effects of urate are mediated by augmenting astrocytic glutathione synthesis and release. Neurobiol Dis 82:574–579. doi:10.1016/j.nbd.2015.08.022
Lee M, Cho T, Jantaratnotai N, et al (2010) Depletion of GSH in glial cells induces neurotoxicity: relevance to aging and degenerative neurological diseases. FASEB J Off Publ Fed Am Soc Exp Biol 24:2533–2545. doi:10.1096/fj.09-149997
Halliwell B (2007) Biochemistry of oxidative stress. Biochem Soc Trans 35:1147–1150. doi:10.1042/BST0351147
Halliwell B (2006) Oxidative stress and neurodegeneration: where are we now? J Neurochem 97:1634–1658. doi:10.1111/j.1471-4159.2006.03907.x
Tanabe K, Matsushima-Nishiwaki R, Yamaguchi S et al (2010) Mechanisms of tumor necrosis factor-alpha-induced interleukin-6 synthesis in glioma cells. J Neuroinflammation 7:16. doi:10.1186/1742-2094-7-16
Shen H-M, Pervaiz S (2006) TNF receptor superfamily-induced cell death: redox-dependent execution. FASEB J Off Publ Fed Am Soc Exp Biol 20:1589–1598. doi:10.1096/fj.05-5603rev
Fischer R, Maier O (2015) Interrelation of oxidative stress and inflammation in neurodegenerative disease: role of TNF. Oxid Med Cell Longev 2015:610813. doi:10.1155/2015/610813
Radford RA, Morsch M, Rayner SL, et al (2015) The established and emerging roles of astrocytes and microglia in amyotrophic lateral sclerosis and frontotemporal dementia. Front Cell Neurosci 9:414. doi:10.3389/fncel.2015.00414
McBean GJ, López MG, Wallner FK (2016) Redox-based therapeutics in neurodegenerative disease. Br J Pharmacol. doi:10.1111/bph.13551
Chamorro Á, Dirnagl U, Urra X, Planas AM (2016) Neuroprotection in acute stroke: targeting excitotoxicity, oxidative and nitrosative stress, and inflammation. Lancet Neurol 15:869–881. doi:10.1016/S1474-4422(16)00114-9
Vargas F, Rodríguez-Gómez I, Pérez-Abud R et al (2012) Cardiovascular and renal manifestations of glutathione depletion induced by buthionine sulfoximine. Am J Hypertens 25:629–635. doi:10.1038/ajh.2011.240
Ibi M, Sawada H, Kume T et al (1999) Depletion of intracellular glutathione increases susceptibility to nitric oxide in mesencephalic dopaminergic neurons. J Neurochem 73:1696–1703
Gegg ME, Clark JB, Heales SJR (2005) Co-culture of neurones with glutathione deficient astrocytes leads to increased neuronal susceptibility to nitric oxide and increased glutamate-cysteine ligase activity. Brain Res 1036:1–6. doi:10.1016/j.brainres.2004.11.064
dos Santos AQ, Nardin P, Funchal C et al (2006) Resveratrol increases glutamate uptake and glutamine synthetase activity in C6 glioma cells. Arch Biochem Biophys 453:161–167. doi:10.1016/j.abb.2006.06.025
Quincozes-Santos A, Bobermin LD, Souza DG et al (2014) Guanosine protects C6 astroglial cells against azide-induced oxidative damage: a putative role of Heme oxygenase 1. J Neurochem 130:61–74. doi:10.1111/jnc.12694
Steiner J, Schroeter ML, Schiltz K et al (2010) Haloperidol and clozapine decrease S100B release from glial cells. Neuroscience 167:1025–1031. doi:10.1016/j.neuroscience.2010.03.010
Bobermin LD, Arús BA, Leite MC et al (2016) Gap Junction Intercellular Communication Mediates Ammonia-Induced Neurotoxicity. Neurotox Res 29:314–324. doi:10.1007/s12640-015-9581-5
Bobermin LD, Quincozes-Santos A, Guerra MC, et al (2012) Resveratrol prevents ammonia toxicity in astroglial cells. PloS One 7:e52164. doi:10.1371/journal.pone.0052164
Seelig GF, Meister A (1985) Glutathione biosynthesis; gamma-glutamylcysteine synthetase from rat kidney. Methods Enzymol 113:379–390
Wegrzynowicz M, Hilgier W, Dybel A et al (2007) Upregulation of cerebral cortical glutathione synthesis by ammonia in vivo and in cultured glial cells: the role of cystine uptake. Neurochem Int 50:883–889. doi:10.1016/j.neuint.2006.12.003
Browne RW, Armstrong D (1998) Reduced glutathione and glutathione disulfide. Methods Mol Biol Clifton NJ 108:347–352. doi:10.1385/0-89603-472-0:347
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Iglesias J, Morales L, Barreto GE (2016) Metabolic and inflammatory adaptation of reactive astrocytes: role of PPARs. Mol Neurobiol. doi:10.1007/s12035-016-9833-2
Parpura V, Heneka MT, Montana V, et al (2012) Glial cells in (patho)physiology: glial cells in (patho) physiology. J Neurochem 121:4–27. doi:10.1111/j.1471-4159.2012.07664.x
Verkhratsky A, Rodríguez JJ, Parpura V (2014) Neuroglia in ageing and disease. Cell Tissue Res 357:493–503. doi:10.1007/s00441-014-1814-z
Bélanger M, Allaman I, Magistretti PJ (2011) Brain energy metabolism: focus on astrocyte-neuron metabolic cooperation. Cell Metab 14:724–738. doi:10.1016/j.cmet.2011.08.016
Quincozes-Santos A, Bobermin LD, de Assis AM et al (2016) Fluctuations in glucose levels induce glial toxicity with glutamatergic, oxidative and inflammatory implications. Biochim Biophys Acta. doi:10.1016/j.bbadis.2016.09.013
Anderson ME (1998) Glutathione: an overview of biosynthesis and modulation. Chem Biol Interact 111–112:1–14
Vargas MR, Johnson DA, Sirkis DW et al (2008) Nrf2 activation in astrocytes protects against neurodegeneration in mouse models of familial amyotrophic lateral sclerosis. J Neurosci Off J Soc Neurosci 28:13574–13581. doi:10.1523/JNEUROSCI.4099-08.2008
Calkins MJ, Johnson DA, Townsend JA et al (2009) The Nrf2/ARE pathway as a potential therapeutic target in neurodegenerative disease. Antioxid Redox Signal 11:497–508. doi:10.1089/ars.2008.2242
Aoyama K, Nakaki T (2015) Glutathione in Cellular Redox Homeostasis: Association with the Excitatory Amino Acid Carrier 1 (EAAC1). Mol Basel Switz 20:8742–8758. doi:10.3390/molecules20058742
Escartin C, Won SJ, Malgorn C et al (2011) Nuclear factor erythroid 2-related factor 2 facilitates neuronal glutathione synthesis by upregulating neuronal excitatory amino acid transporter 3 expression. J Neurosci Off J Soc Neurosci 31:7392–7401. doi:10.1523/JNEUROSCI.6577-10.2011
Biswas C, Shah N, Muthu M et al (2014) Nuclear Heme oxygenase-1 (HO-1) modulates subcellular distribution and activation of Nrf2, impacting metabolic and anti-oxidant defenses. J Biol Chem 289:26882–26894. doi:10.1074/jbc.M114.567685
Chi P-L, Lin C-C, Chen Y-W et al (2014) CO induces Nrf2-dependent Heme oxygenase-1 transcription by cooperating with Sp1 and c-Jun in rat brain astrocytes. Mol Neurobiol 52:277–292. doi:10.1007/s12035-014-8869-4
Backos DS, Franklin CC, Reigan P (2012) The role of glutathione in brain tumor drug resistance. Biochem Pharmacol 83:1005–1012. doi:10.1016/j.bcp.2011.11.016
Park H-A, Khanna S, Rink C et al (2009) Glutathione disulfide induces neural cell death via a 12-lipoxygenase pathway. Cell Death Differ 16:1167–1179. doi:10.1038/cdd.2009.37
Huang T-C, Lu K-T, Wo Y-YP, et al (2011) Resveratrol protects rats from Aβ-induced neurotoxicity by the reduction of iNOS expression and lipid peroxidation. PloS One 6:e29102. doi:10.1371/journal.pone.0029102
Yuste JE, Tarragon E, Campuzano CM, Ros-Bernal F (2015) Implications of glial nitric oxide in neurodegenerative diseases. Front Cell Neurosci 9:322. doi:10.3389/fncel.2015.00322
Xia N, Strand S, Schlufter F et al (2013) Role of SIRT1 and FOXO factors in eNOS transcriptional activation by resveratrol. Nitric Oxide. Biol Chem 32:29–35. doi:10.1016/j.niox.2013.04.001
Sarady JK, Zuckerbraun BS, Bilban M, et al (2004) Carbon monoxide protection against endotoxic shock involves reciprocal effects on iNOS in the lung and liver. FASEB J Off Publ Fed Am Soc Exp Biol 18:854–856. doi:10.1096/fj.03-0643fje
Olmos G, Lladó J (2014) Tumor necrosis factor alpha: a link between neuroinflammation and excitotoxicity. Mediators Inflamm 2014:861231. doi:10.1155/2014/861231
Rossi D (2015) Astrocyte physiopathology: At the crossroads of intercellular networking, inflammation and cell death. Prog Neurobiol 130:86–120. doi:10.1016/j.pneurobio.2015.04.003
Kaur U, Banerjee P, Bir A et al (2015) Reactive oxygen species, redox signaling and neuroinflammation in Alzheimer’s disease: the NF-κB connection. Curr Top Med Chem 15:446–457
Nakajima S, Kitamura M (2013) Bidirectional regulation of NF-κB by reactive oxygen species: a role of unfolded protein response. Free Radic Biol Med 65:162–174. doi:10.1016/j.freeradbiomed.2013.06.020
Acknowledgements
This study was supported by the Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS), Financiadora de Estudos e Projetos (FINEP)-Instituto Brasileiro de Neurociências (IBN Net) 01.06.0842-00, and Universidade Federal do Rio Grande do Sul and Instituto Nacional de Ciência e Tecnologia para Excitotoxicidade e Neuroproteção (INCTEN/CNPq).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Arús, B.A., Souza, D.G., Bellaver, B. et al. Resveratrol modulates GSH system in C6 astroglial cells through heme oxygenase 1 pathway. Mol Cell Biochem 428, 67–77 (2017). https://doi.org/10.1007/s11010-016-2917-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-016-2917-5