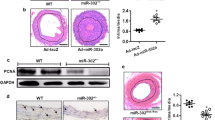
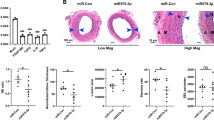
Abstract
In the recent decades, carotid angioplasty and stenting (CAS) has been developed into a credible option for the patients with carotid stenosis. However, restenosis remains a severe and unsolved issue after CAS treatment. Restenosis is characterized by neointimal hyperplasia, which is partially caused by vascular smooth muscle cells (VSMC) proliferation. However, the molecular mechanism involved in the restenosis is still unclear. In this study, we demonstrated a functional crosstalk between two TGF-β superfamily signaling pathway members, Smad3 and BMPR2, in VSMC proliferation. Smad3 plays an important role in the TGF-β/Smad3 signaling pathway, and is significantly upregulated in the carotid artery with restenosis to promote VSMC proliferation. In contrast, BMP receptor II (BMPR2), an inhibitor of VSMC proliferation is downregulated in carotid restenosis. We further found that BMPR2 downregulation is mediated by miR-17–92 cluster, which is transcriptionally regulated by Smad3. Thus, Smad3 upregulation and Smad3/miR-17–92 cluster-dependent BMPR2 downregulation are likely to promote VSMC proliferation and restenosis. Taken together, our results may provide novel clues for early diagnosis of carotid restenosis and developing new therapeutic strategy.




Similar content being viewed by others
References
Meissner I, Meyer FB (1994) Carotid stenosis and carotid endarterectomy. Cerebrovasc Brain Metab Rev 6(2):163–179
Berman SS, Bernhard VM, Erly WK, McIntyre KE, Erdoes LS, Hunter GC (1994) Critical carotid artery stenosis: diagnosis, timing of surgery, and outcome. J Vasc Surg 20(4):499–508 Discussion 508–410
Kelly R (1992) Selections from current literature: prevention of stroke in non-rheumatic atrial fibrillation and carotid artery stenosis. Fam Pract 9(2):231–236
Sherman DG (1989) The carotid artery and stroke. Am Fam Physician 40 (5 Suppl):41S–44S, 47S–49S
Parnetti L, Mercuri M, Susta A, Lupattelli G, Ciuffetti G, Senin U (1988) Extracranial carotid atherosclerosis evaluation and stroke occurrence: role of the echotomographic analysis. Angiology 39(8):705–713
Wasserman BA, Haacke EM, Li D (1994) Carotid plaque formation and its evaluation with angiography, ultrasound, and MR angiography. J Magn Reson Imaging 4(4):515–527
Fanelli F, Boatta E, Cannavale A, Corona M, Lucatelli P, Wlderk A, Cirelli C, Salvatori FM (2012) Carotid artery stenting: analysis of a 12-year single-center experience. J Endovasc Ther 19(6):749–756. doi:10.1583/JEVT-12-3944MR.1
Gahremanpour A, Perin EC, Silva G (2012) Carotid artery stenting versus endarterectomy: a systematic review. Tex Heart Inst J 39(4):474–487
AbuRahma AF, Abu-Halimah S, Hass SM, Nanjundappa A, Stone PA, Mousa A, Lough E, Dean LS (2010) Carotid artery stenting outcomes are equivalent to carotid endarterectomy outcomes for patients with post-carotid endarterectomy stenosis. J Vasc Surg 52(5):1180–1187. doi:10.1016/j.jvs.2010.06.074
Cosottini M, Michelassi MC, Bencivelli W, Lazzarotti G, Picchietti S, Orlandi G, Parenti G, Puglioli M (2010) In stent restenosis predictors after carotid artery stenting. Stroke Res Treat. doi:10.4061/2010/864724
Costa MA, Simon DI (2005) Molecular basis of restenosis and drug-eluting stents. Circulation 111(17):2257–2273. doi:10.1161/01.CIR.0000163587.36485.A7
Inoue T, Node K (2009) Molecular basis of restenosis and novel issues of drug-eluting stents. Circ J 73(4):615–621
Suwanabol PA, Seedial SM, Shi X, Zhang F, Yamanouchi D, Roenneburg D, Liu B, Kent KC (2012) Transforming growth factor-beta increases vascular smooth muscle cell proliferation through the Smad3 and extracellular signal-regulated kinase mitogen-activated protein kinases pathways. J Vasc Surg 56(2):446–454. doi:10.1016/j.jvs.2011.12.038
Suwanabol PA, Kent KC, Liu B (2011) TGF-beta and restenosis revisited: a Smad link. J Surg Res 167(2):287–297. doi:10.1016/j.jss.2010.12.020
Tsai S, Hollenbeck ST, Ryer EJ, Edlin R, Yamanouchi D, Kundi R, Wang C, Liu B, Kent KC (2009) TGF-beta through Smad3 signaling stimulates vascular smooth muscle cell proliferation and neointimal formation. Am J Physiol Heart Circ Physiol 297(2):H540–H549. doi:10.1152/ajpheart.91478.2007
Rasmussen LM, Wolf YG, Ruoslahti E (1995) Vascular smooth muscle cells from injured rat aortas display elevated matrix production associated with transforming growth factor-beta activity. Am J Pathol 147(4):1041–1048
Ryer EJ, Hom RP, Sakakibara K, Nakayama KI, Nakayama K, Faries PL, Liu B, Kent KC (2006) PKCdelta is necessary for Smad3 expression and transforming growth factor beta-induced fibronectin synthesis in vascular smooth muscle cells. Arterioscler Thromb Vasc Biol 26(4):780–786. doi:10.1161/01.ATV.0000209517.00220.cd
Massague J, Seoane J, Wotton D (2005) Smad transcription factors. Genes Dev 19(23):2783–2810. doi:10.1101/gad.1350705
Derynck R, Zhang Y, Feng XH (1998) Smads: transcriptional activators of TGF-beta responses. Cell 95(6):737–740
Matzuk MM (1995) Functional analysis of mammalian members of the transforming growth factor-beta superfamily. Trends Endocrinol Metab 6(4):120–127
Miyazono K, Kusanagi K, Inoue H (2001) Divergence and convergence of TGF-beta/BMP signaling. J Cell Physiol 187(3):265–276. doi:10.1002/jcp.1080
Chen D, Zhao M, Mundy GR (2004) Bone morphogenetic proteins. Growth Factors 22(4):233–241. doi:10.1080/08977190412331279890
Sieber C, Kopf J, Hiepen C, Knaus P (2009) Recent advances in BMP receptor signaling. Cytokine Growth Factor Rev 20(5–6):343–355. doi:10.1016/j.cytogfr.2009.10.007
Cogan J, Austin E, Hedges L, Womack B, West J, Loyd J, Hamid R (2012) Role of BMPR2 alternative splicing in heritable pulmonary arterial hypertension penetrance. Circulation 126(15):1907–1916. doi:10.1161/CIRCULATIONAHA.112.106245
Li W, Dunmore BJ, Morrell NW (2010) Bone morphogenetic protein type II receptor mutations causing protein misfolding in heritable pulmonary arterial hypertension. Proc Am Thorac Soc 7(6):395–398. doi:10.1513/pats.201002-024AW
Nasim MT, Ogo T, Chowdhury HM, Zhao L, Chen CN, Rhodes C, Trembath RC (2012) BMPR-II deficiency elicits pro-proliferative and anti-apoptotic responses through the activation of TGFbeta-TAK1-MAPK pathways in PAH. Hum Mol Genet 21(11):2548–2558. doi:10.1093/hmg/dds073
Jost D, Unmuth SJ, Meissner H, Henn-Beilharz A, Henkes H, Hupp T (2012) Surgical treatment of carotid in-stent-restenosis: novel strategy and current management. Thorac Cardiovasc Surg 60(8):517–524. doi:10.1055/s-0032-1311535
Filipowicz W, Bhattacharyya SN, Sonenberg N (2008) Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nat Rev Genet 9(2):102–114. doi:10.1038/nrg2290
Lewis BP, Burge CB, Bartel DP (2005) Conserved seed pairing, often flanked by adenosines, indicates that thousands of human genes are microRNA targets. Cell 120(1):15–20. doi:10.1016/j.cell.2004.12.035
Chen Y, Gorski DH (2008) Regulation of angiogenesis through a microRNA (miR-130a) that down-regulates antiangiogenic homeobox genes GAX and HOXA5. Blood 111(3):1217–1226. doi:10.1182/blood-2007-07-104133
Wu WH, Hu CP, Chen XP, Zhang WF, Li XW, Xiong XM, Li YJ (2011) MicroRNA-130a mediates proliferation of vascular smooth muscle cells in hypertension. Am J Hypertens 24(10):1087–1093. doi:10.1038/ajh.2011.116
Maegdefessel L, Azuma J, Toh R, Deng A, Merk, Raiesdana A, Leeper NJ, Raaz U, Schoelmerich AM, McConnell MV, Dalman RL, Spin JM, Tsao PS (2012) MicroRNA-21 blocks abdominal aortic aneurysm development and nicotine-augmented expansion. Sci Transl Med 4(122):122ra122. doi:10.1126/scitranslmed.3003441
McDonald RA, White KM, Wu J, Cooley BC, Robertson KE, Halliday CA, McClure JD, Francis S, Lu R, Kennedy S, George SJ, Wan S, van Rooij E, Baker AH (2013) miRNA-21 is dysregulated in response to vein grafting in multiple models and genetic ablation in mice attenuates neointima formation. Eur Heart J 34(22):1636–1643. doi:10.1093/eurheartj/eht105
Perlman H, Luo Z, Krasinski K, Le Roux A, Mahfoudi A, Smith RC, Branellec D, Walsh K (1999) Adenovirus-mediated delivery of the Gax transcription factor to rat carotid arteries inhibits smooth muscle proliferation and induces apoptosis. Gene Ther 6(5):758–763. doi:10.1038/sj.gt.3300893
Furgeson SB, Simpson PA, Park I, Vanputten V, Horita H, Kontos CD, Nemenoff RA, Weiser-Evans MC (2010) Inactivation of the tumour suppressor, PTEN, in smooth muscle promotes a pro-inflammatory phenotype and enhances neointima formation. Cardiovasc Res 86(2):274–282. doi:10.1093/cvr/cvp425
Kroh EM, Parkin RK, Mitchell PS, Tewari M (2010) Analysis of circulating microRNA biomarkers in plasma and serum using quantitative reverse transcription-PCR (qRT-PCR). Methods 50(4):298–301. doi:10.1016/j.ymeth.2010.01.032
Kosaka N, Iguchi H, Ochiya T (2010) Circulating microRNA in body fluid: a new potential biomarker for cancer diagnosis and prognosis. Cancer Sci 101(10):2087–2092. doi:10.1111/j.1349-7006.2010.01650.x
Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL, Peterson A, Noteboom J, O’Briant KC, Allen A, Lin DW, Urban N, Drescher CW, Knudsen BS, Stirewalt DL, Gentleman R, Vessella RL, Nelson PS, Martin DB, Tewari M (2008) Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci USA 105(30):10513–10518. doi:10.1073/pnas.0804549105
Zhang Y, Liu D, Chen X, Li J, Li L, Bian Z, Sun F, Lu J, Yin Y, Cai X, Sun Q, Wang K, Ba Y, Wang Q, Wang D, Yang J, Liu P, Xu T, Yan Q, Zhang J, Zen K, Zhang CY (2010) Secreted monocytic miR-150 enhances targeted endothelial cell migration. Mol Cell 39(1):133–144. doi:10.1016/j.molcel.2010.06.010
Vickers KC, Palmisano BT, Shoucri BM, Shamburek RD, Remaley AT (2011) MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins. Nat Cell Biol 13(4):423–433. doi:10.1038/ncb2210
Chen X, Ba Y, Ma L, Cai X, Yin Y, Wang K, Guo J, Zhang Y, Chen J, Guo X, Li Q, Li X, Wang W, Wang J, Jiang X, Xiang Y, Xu C, Zheng P, Zhang J, Li R, Zhang H, Shang X, Gong T, Ning G, Zen K, Zhang CY (2008) Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res 18(10):997–1006. doi:10.1038/cr.2008.282
Ahn SM, Cha JY, Kim J, Kim D, Trang HT, Kim YM, Cho YH, Park D, Hong S (2012) Smad3 regulates E-cadherin via miRNA-200 pathway. Oncogene 31(25):3051–3059. doi:10.1038/onc.2011.484
Acknowledgments
This study was supported by the National Institute of Health (CA132755 and CA130899 to X.Y.), the University of Michigan Cancer Center and GI Peptide Research Center. X.Y. is a recipient of the Era of Hope Scholar Award from the Department of Defense.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Tao Luo and Shijun Cui have contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Luo, T., Cui, S., Bian, C. et al. Crosstalk between TGF-β/Smad3 and BMP/BMPR2 signaling pathways via miR-17–92 cluster in carotid artery restenosis. Mol Cell Biochem 389, 169–176 (2014). https://doi.org/10.1007/s11010-013-1938-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1938-6