Abstract
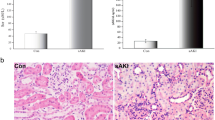
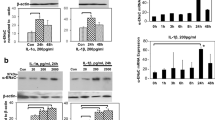
Nuclear factor κB (NF-κB) plays an important role in the regulation of inflammatory proteins. However, it is unclear whether the NF-κB/intercellular adhesion molecule-1 (ICAM-1) pathway is involved in the adhesion of neutrophils and renal injury after hypoxia–ischemia (HI) in neonates. In this report we investigated whether NF-κB and its downstream molecule ICAM-1 were involved in renal injury induced by postasphyxial serum (PS) from neonates. Human renal proximal tubular (HK-2) cells were preincubated with 10 % fetal calf serum (control), 20 % neonatal PS, or 20 % PS plus pyrolidine dithiocarbamate (PDTC). The expression of IκBα, NF-κB p65, and ICAM-1 in HK-2 cells was determined by Western blot and/or immunohistochemistry. Nuclear translocation of NF-κB p65 in HK-2 cells was detected by immunofluorescence and Western blot. The ICAM-1 mRNA was determined by RT-PCR. Then HK-2 cells were cultured with neutrophils from neonates with asphyxia. After HK-2 cells had been cultured with neutrophils, we detected myeloperoxidase (MPO) activity, the leakage rate of lactate dehydrogenase (LDH), and cell viability. We found that PS preincubation resulted in significantly decreased IκBα expression and increased expression of NF-κB and ICAM-1, and facilitated the nuclear translocation of NF-κB in HK-2 cells. PS preincubation increased MPO activity, leading to elevated leakage rates of LDH and decreased cell viability after neutrophil exposure. Furthermore, the inhibition of NF-κB activity by PDTC significantly upregulated IκBα expression, decreased NF-κB and ICAM-1 expression, downregulated the nuclear translocation of NF-κB, and decreased MPO activity. This leads to decreased leakage rates of LDH and increased cell viability after neutrophil exposure. Our findings suggest that NF-κB/ICAM-1 pathway may be involved in neutrophil–endothelial interactions and neonatal renal injury after HI.








Similar content being viewed by others
References
Hankins GD, Koen S, Gei AF, Lopez SM, van Hook JW, Anderson GD (2002) Neonatal organ system injury in acute birth asphyxia sufficient to result in neonatal encephalopathy. Obstet Gynecol 99:688–691
Beekhuizen H, van de Gevel JS (1998) Endothelial cell adhesion molecules in inflammation and postischemic reperfusion injury. Transplant Proc 30:4251–4256
Li L, Huang L, Vergis AL, Ye H, Bajwa A, Narayan V, Strieter RM, Rosin DL, Okusa MD (2010) IL-17 produced by neutrophils regulates IFN-gamma-mediated neutrophil migration in mouse kidney ischemia-reperfusion injury. J Clin Invest 120:331–342
Hasegawa T, Ito Y, Wijeweera J, Liu J, Malle E, Farhood A, McCuskey RS, Jaeschke H (2007) Reduced inflammatory response and increased microcirculatory disturbances during hepatic ischemia-reperfusion injury in steatotic livers of ob/ob mice. Am J Physiol Gastrointest Liver Physiol 292:G1385–G1395
Spandou E, Tsouchnikas I, Karkavelas G, Dounousi E, Simeonidou C, Guiba-Tziampiri O, Tsakiris D (2006) Erythropoietin attenuates renal injury in experimental acute renal failure ischaemic/reperfusion model. Nephrol Dial Transplant 21:330–336
Benard C, Cultrone A, Michel C, Rosales C, Segain JP, Lahaye M, Galmiche JP, Cherbut C, Blottiere HM (2010) Degraded carrageenan causing colitis in rats induces TNF secretion and ICAM-1 upregulation in monocytes through NF-kappaB activation. PLoS One 5:e8666
Mizuno S, Nakamura T (2005) Prevention of neutrophil extravasation by hepatocyte growth factor leads to attenuations of tubular apoptosis and renal dysfunction in mouse ischemic kidneys. Am J Pathol 166:1895–1905
Hadad N, Tuval L, Elgazar-Carmom V, Levy R (2011) Endothelial ICAM-1 protein induction is regulated by cytosolic phospholipase A2{alpha} via both NF-{kappa}B and CREB transcription factors. J Immunol 186:1816–1827
Min JK, Kim YM, Kim SW, Kwon MC, Kong YY, Hwang IK, Won MH, Rho J, Kwon YG (2005) TNF-related activation-induced cytokine enhances leukocyte adhesiveness: induction of ICAM-1 and VCAM-1 via TNF receptor-associated factor and protein kinase C-dependent NF-kappaB activation in endothelial cells. J Immunol 175:531–540
Winning S, Splettstoesser F, Fandrey J, Frede S (2010) Acute hypoxia induces HIF-independent monocyte adhesion to endothelial cells through increased intercellular adhesion molecule-1 expression: the role of hypoxic inhibition of prolyl hydroxylase activity for the induction of NF-kappa B. J Immunol 185:1786–1793
Srichai MB, Hao C, Davis L, Golovin A, Zhao M, Moeckel G, Dunn S, Bulus N, Harris RC, Zent R, Breyer MD (2008) Apoptosis of the thick ascending limb results in acute kidney injury. J Am Soc Nephrol 19:1538–1546
Dong WB, Tang ZH, Chen HY, Chen SQ, Wang XY, Wang SH, Zhai XS, Xiao DC (2003) Change in the level of inflammatory cytokines and their relationship to the indicator in evaluating renal tubules injury of urinary in neonates with asphyxia. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue 15:94–96
Dong WB, Du YT, Chen Y, Hang YL, Chen F, Deng CL (2005) The relationship between TNF-alpha and the injury of renal tubular cells caused by anoxia/reoxygenation. Xi Bao Yu Fen Zi Mian Yi Xue Za Zhi 21:690–692
Wen-bin D, Min C, Ming-yong W, Cun-liang D, Feng C, Kai-gui X (2005) Effect of postasphyxial-serum in neonate inducing apoptosis of renal tubular cells [J]. J Appl Clin Pediatr 20:1207–1209
Zhang Y, Dong WB, Li QP, Deng CL, Xiong T, Lei XP, Guo L (2009) Role of Omi/HtrA2 in renal tubular cells apoptosis induced by post asphyxial serum of neonate. Zhongguo Wei Zhong Bing Ji Jiu Yi Xue 21:346–348
Li L, Qu Y, Li J, Xiong Y, Mao M, Mu D (2007) Relationship between HIF-1alpha expression and neuronal apoptosis in neonatal rats with hypoxia-ischemia brain injury. Brain Res 1180:133–139
Vranyac-Tramoundanas A, Harrison JC, Clarkson AN, Kapoor M, Winburn IC, Kerr DS, Sammut IA (2008) Domoic acid impairment of cardiac energetics. Toxicol Sci 105:395–407
Huang Y, Rabb H, Womer KL (2007) Ischemia-reperfusion and immediate T cell responses. Cell Immunol 248:4–11
Lee HT, Kim M, Kim N, FTt Billings, D’Agati VD, Emala CW Sr (2007) Isoflurane protects against renal ischemia and reperfusion injury and modulates leukocyte infiltration in mice. Am J Physiol Renal Physiol 293:F713–F722
Lee HT, Kim M, Jan M, Emala CW (2006) Anti-inflammatory and antinecrotic effects of the volatile anesthetic sevoflurane in kidney proximal tubule cells. Am J Physiol Renal Physiol 291:F67–F78
Burne MJ, Elghandour A, Haq M, Saba SR, Norman J, Condon T, Bennett F, Rabb H (2001) IL-1 and TNF independent pathways mediate ICAM-1/VCAM-1 up-regulation in ischemia reperfusion injury. J Leukoc Biol 70:192–198
Akcay A, Nguyen Q, Edelstein CL (2009) Mediators of inflammation in acute kidney injury. Mediators Inflamm 2009:137072
Frommhold D, Kamphues A, Hepper I, Pruenster M, Lukic IK, Socher I, Zablotskaya V, Buschmann K, Lange-Sperandio B, Schymeinsky J, Ryschich E, Poeschl J, Kupatt C, Nawroth PP, Moser M, Walzog B, Bierhaus A, Sperandio M (2010) RAGE and ICAM-1 cooperate in mediating leukocyte recruitment during acute inflammation in vivo. Blood 116:841–849
Sutton TA, Fisher CJ, Molitoris BA (2002) Microvascular endothelial injury and dysfunction during ischemic acute renal failure. Kidney Int 62:1539–1549
Koo DD, Welsh KI, West NE, Channon KM, Penington AJ, Roake JA, Morris PJ, Fuggle SV (2001) Endothelial cell protection against ischemia/reperfusion injury by lecithinized superoxide dismutase. Kidney Int 60:786–796
Graham KM, Singh R, Millman G, Malnassy G, Gatti F, Bruemmer K, Stefanski C, Curtis H, Sesti J, Carlson CG (2010) Excessive collagen accumulation in dystrophic (mdx) respiratory musculature is independent of enhanced activation of the NF-kappaB pathway. J Neurol Sci 294:43–50
Wan X, Fan L, Hu B, Yang J, Li X, Chen X, Cao C (2011) Small interfering RNA targeting IKK{beta} prevents renal ischemia- reperfusion injury in rats. Am J Physiol Renal Physiol 300(4):857–863
Chen HH, Chen TW, Lin H (2009) Prostacyclin-induced peroxisome proliferator-activated receptor-alpha translocation attenuates NF-kappaB and TNF-alpha activation after renal ischemia-reperfusion injury. Am J Physiol Renal Physiol 297:F1109–F1118
Acknowledgments
This research was supported by Sichuan Youth Science and Technology Foundation (No. 04ZQ026-033 to Wenbin Dong), and the National Natural Science Foundation of China (No. 81300525 to Tao Xiong). The authors are also grateful for support provided by the confocal microscope facility at the Institute of Myocardium Electrophysiology of Luzhou Medical College.
Conflict of interest
The authors declare that they have no conflicts of interest concerning this article.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xiong, T., Dong, W., Fu, H. et al. Involvement of the nuclear factor-κB pathway in the adhesion of neutrophils to renal tubular cells after injury induced by neonatal postasphyxial serum. Mol Cell Biochem 388, 85–94 (2014). https://doi.org/10.1007/s11010-013-1901-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1901-6