Abstract
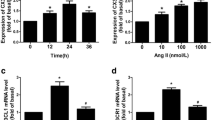
Vascular remodeling is characterized by the aggregation of vascular smooth muscle cells (VSMCs) in intima. Previous studies have demonstrated that dehydroepiandrosterone (DHEA), a steroid hormone, can reverse vascular remodeling. However, it is still far clear that whether and how DHEA participates in the modulation of VSMCs activation and vascular remodeling. VSMCs were obtained from the thoracic aorta of SD rats. Cell proliferation was evaluated by CCK-8 assay and BrdU assay. To measure VSMCs migration activity, a transwell chamber assay was performed. Quantitative real-time RT-PCR and western blot were used to explore the molecular mechanisms. ROS generation by VSMCs was measured by DCF fluorescence. NADPH oxidase activity and SOD activity were measured by the corresponding kits. NF-κB activity was detected by NF-κB luciferase reporter gene assay. A rat carotid artery balloon injury model was built to evaluate the neointimal formation, and plasma PGF2 was measured by ELISA. Our results showed that DHEA significantly inhibited VSMCs proliferation after angiotensin (Ang II) stimulation by down-regulation of NADPH oxidase activity and ERK1/2 phosphorylation. Ang II can increase IL-6 and MCP-1 expression, but DHEA reverses these changes via inhibiting p38-MAPK/NF-κB (p65) signaling pathway. DHEA has no significant effects on VSMCs phenotype transition, but can reduce the neointimal to media area ratio after balloon injury. DHEA can alleviate oxidative stress and inflammation in VSMCs via ERK1/2 and NF-κB signaling pathway, but has no effect on VSMCs phenotype transition. Furthermore, DHEA attenuates VSMCs activation and neointimal formation after carotid injury in vivo. Taken together, DHEA might be a promising treatment for vascular injury under pathological condition.






Similar content being viewed by others
References
Pugh PJ, Jones RD, Jones TH, Channer KS (2002) Heart failure as an inflammatory condition: potential role for androgens as immune modulators. Eur J Heart Fail 4(6):673–680
English KM, Mandour O, Steeds RP, Diver MJ, Jones TH, Channer KS (2000) Men with coronary artery disease have lower levels of androgens than men with normal coronary angiograms. Eur Heart J 21(11):890–894
Jankowska EA, Drohomirecka A, Ponikowska B, Witkowska A, Lopuszanska M, Szklarska A, Borodulin-Nadzieja L, Banasiak W, Poole-Wilson PA, Ponikowski P (2010) Deficiencies in circulating testosterone and dehydroepiandrosterone sulphate, and depression in men with systolic chronic heart failure. Eur J Heart Fail 12(9):966–973
Feldman HA, Johannes CB, Araujo AB, Mohr BA, Longcope C, McKinlay JB (2001) Low dehydroepiandrosterone and ischemic heart disease in middle-aged men: prospective results from the Massachusetts male aging study. Am J Epidemiol 153(1):79–89
LaCroix AZ, Yano K, Reed DM (1992) Dehydroepiandrosterone sulfate, incidence of myocardial infarction, and extent of atherosclerosis in men. Circulation 86(5):1529–1535
Savastano S, Valentino R, Belfiore A, De Luca N, de Alteriis A, Orio F Jr, Palomba S, Villani AM, Falconi C, Lupoli G, Lombardi G (2003) Early carotid atherosclerosis in normotensive severe obese premenopausal women with low DHEA(S). J Endocrinol Invest 26(3):236–243
Bonnet S, Dumas-de-La-Roque E, Bégueret H, Marthan R, Fayon M, Dos Santos P, Savineau JP, Baulieu EE (2003) Dehydroepiandrosterone (DHEA) prevents and reverses chronic hypoxic pulmonary hypertension. Proc Natl Acad Sci USA 100(16):9488–9493
Hayashi T, Esaki T, Muto E, Kano H, Asai Y, Thakur NK, Sumi D, Jayachandran M, Iguchi A (2000) Dehydroepiandrosterone retards atherosclerosis formation through its conversion to estrogen: the possible role of nitric oxide. Arterioscler Thromb Vasc Biol 20(3):782–792
Kiechl S, Willeit J, Bonora E, Schwarz S, Xu Q (2000) No association between dehydroepiandrosterone sulfate and development of atherosclerosis in a prospective population study (Bruneck Study). Arterioscler Thromb Vasc Biol 20(4):1094–1100
Barrett-Connor E, Goodman-Gruen D (1995) Dehydroepiandrosterone sulfate does not predict cardiovascular death in postmenopausal women: the Rancho Bernardo Study. Circulation 91(6):1757–1760
Nedeljkovic ZS, Gokce N, Loscalzo J (2003) Mechanisms of oxidative stress and vascular dysfunction. Postgrad Med J 79(930):195–199 quiz 198–200
Tedgui A, Mallat Z (2006) Cytokines in atherosclerosis: pathogenic and regulatory pathways. Physiol Rev 86(2):515–581
Koenig W, Khuseyinova N (2007) Biomarkers of atherosclerotic plaque instability and rupture. Arterioscler Thromb Vasc Biol 27(1):15–26
Packard RR, Libby P (2008) Inflammation in atherosclerosis: from vascular biology to biomarker discovery and risk prediction. Clin Chem 54(1):24–38
Morrow D, Guha S, Sweeney C, Birney Y, Walshe T, O’Brien C, Walls D, Redmond EM, Cahill PA (2008) Notch and vascular smooth muscle cell phenotype. Circ Res 103(12):1370–1382
Fuster JJ, Fernández P, González-Navarro H, Silvestre C, Nabah YN, Andrés V (2010) Control of cell proliferation in atherosclerosis: insights from animal models and human studies. Cardiovasc Res 86(2):254–264
Tokunou T, Shibata R, Kai H, Ichiki T, Morisaki T, Fukuyama K, Ono H, Iino N, Masuda S, Shimokawa H, Egashira K, Imaizumi T, Takeshita A (2003) Apoptosis induced by inhibition of cyclic AMP response element-binding protein in vascular smooth muscle cells. Circulation 108(10):1246–1252
Chen J, Zhang J, Xu L, Xu C, Chen S, Yang J, Jiang H (2012) Inhibition of neointimal hyperplasia in the rat carotid artery injury model by a HMGB1 inhibitor. Atherosclerosis 224(2):332–339
Han M, Wen JK, Zheng B, Cheng Y, Zhang C (2006) Serum deprivation results in redifferentiation of human umbilical vascular smooth muscle cells. Am J Physiol Cell Physiol 291:C50–C58
Liu J, Xiu J, Cao J, Gao Q, Ma D, Fu L (2011) Berberine cooperates with adrenal androgen dehydroepiandrosterone sulfate to attenuate PDGF-induced proliferation of vascular smooth muscle cell A7r5 through Skp2 signaling pathway. Mol Cell Biochem 355(1–2):127–134
Urata Y, Goto S, Kawakatsu M, Yodoi J, Eto M, Akishita M, Kondo T (2010) DHEA attenuates PDGF-induced phenotypic proliferation of vascular smooth muscle A7r5 cells through redox regulation. Biochem Biophys Res Commun 396(2):489–494
Ii M, Hoshiga M, Negoro N, Fukui R, Nakakoji T, Kohbayashi E, Shibata N, Furutama D, Ishihara T, Hanafusa T, Losordo DW, Ohsawa N (2009) Adrenal androgen dehydroepiandrosterone sulfate inhibits vascular remodeling following arterial injury. Atherosclerosis 206(1):77–85
Berry C, Touyz R, Dominiczak AF, Webb RC, Johns DG (2001) Angiotensin receptors: signaling, vascular pathophysiology, and interactions with ceramide. Am J Physiol Heart Circ Physiol 281(6):H2337–H2365
Mugabe BE, Yaghini FA, Song CY, Buharalioglu CK, Waters CM, Malik KU (2010) Angiotensin II-induced migration of vascular smooth muscle cells (VSMCs) is mediated by both 72-kDa spleen tyrosine kinase (Syk) via p38-MAPK activated c-Src and by ERK1/2 via c-Src-induced EGFR transactivation. J Pharmacol Exp Ther 332(1):116–124
Luo X, Xiao Y, Song F, Yang Y, Xia M, Ling W (2012) Increased plasma S-adenosyl-homocysteine levels induce the proliferation and migration of VSMCs through an oxidative stress-ERK1/2 pathway in apoE(−/−) mice. Cardiovasc Res 95(2):241–250
Camporez JP, Akamine EH, Davel AP, Franci CR, Rossoni LV, Carvalho CR (2011) Dehydroepiandrosterone protects against oxidative stress-induced endothelial dysfunction in ovariectomized rats. J Physiol 589(Pt 10):2585–2596
Jia C, Chen X, Li X, Li M, Miao C, Sun B, Fan Z, Ren L (2011) The effect of DHEA treatment on the oxidative stress and myocardial fibrosis induced by Keshan disease pathogenic factors. J Trace Elem Med Biol 25(3):154–159
Steele VE, Arnold JT, Lei H, Izmirlian G, Blackman MR (2006) Comparative effects of DHEA and DHT on gene expression in human LNCaP prostate cancer cells. Anticancer Res 26(5A):3205–3215
Raza K, Thambyrajah J, Townend JN, Exley AR, Hortas C, Filer A, Carruthers DM, Bacon PA (2000) Suppression of inflammation in primary systemic vasculitis restores vascular endothelial function: lessons for atherosclerotic disease? Circulation 102(13):1470–1472
Taube A, Schlich R, Sell H, Eckardt K, Eckel J (2012) Inflammation and metabolic dysfunction: links to cardiovascular diseases. Am J Physiol Heart Circ Physiol 302(11):H2148–H2165
Brasier AR (2010) The nuclear factor-κB–interleukin-6 signalling pathway mediating vascular inflammation. Cardiovasc Res 86(2):211–218
Chen J, Jiang H, Yang J, Chen SS, Xu L (2012) Down-regulation of CREB-binding protein expression blocks thrombin-mediated endothelial activation by inhibiting acetylation of NF-κB. Int J Cardiol 154(2):147–152
Monaco C, Paleolog E (2004) Nuclear factor κB: a potential therapeutic target in atherosclerosis and thrombosis. Cardiovasc Res 61(4):671–682
Yang J, Jiang H, Chen SS, Chen J, Xu SK, Li WQ, Wang JC (2010) CBP knockdown inhibits angiotensin II-induced vascular smooth muscle cells proliferation through downregulating NF-κB transcriptional activity. Mol Cell Biochem 340(1–2):55–62
Natoli G, Chiocca S (2008) Nuclear ubiquitin ligases, NF-κB degradation, and the control of inflammation. Sci Signal 1(1):pe1
Patel DN, King CA, Bailey SR, Holt JW, Venkatachalam K, Agrawal A, Valente AJ, Chandrasekar B (2007) Interleukin-17 stimulates C-reactive protein expression in hepatocytes and smooth muscle cells via p38 MAPK and ERK1/2-dependent NF-κB and C/EBP β Activation. J Biol Chem 282(37):27229–27238
Saha RN, Jana M, Pahan K (2007) MAPK p38 regulates transcriptional activity of NF-κB in primary human astrocytes via acetylation of p65. J Immunol 179(10):7101–7109
van Kleef EM, Fingerle J, Daemen MJ (1996) Angiotensin II-induced progression of neointimal thickening in the balloon-injured rat carotid artery is AT1 receptor mediated. Arterioscler Thromb Vasc Biol 16(7):857–863
Li F, Zhang C, Schaefer S, Estes A, Malik KU (2005) ANG II-induced neointimal growth is mediated via cPLA2- and PLD2-activated Akt in balloon-injured rat carotid artery. Am J Physiol Heart Circ Physiol 289(6):H2592–H2601
Eguchi S, Dempsey PJ, Frank GD, Motley ED, Inagami T (2001) Activation of MAPKs by angiotensin II in vascular smooth muscle cells. Metalloprotease-dependent EGF receptor activation is required for activation of ERK and p38 MAPK but not for JNK. J Biol Chem 276(11):7957–7962
Dugourd C, Gervais M, Corvol P, Monnot C (2003) Akt is a major downstream target of PI3-kinase involved in angiotensin II-induced proliferation. Hypertension 41(4):882–890
Kaplan-Albuquerque N, Bogaert YE, Van Putten V, Weiser-Evans MC, Nemenoff RA (2005) Patterns of gene expression differentially regulated by platelet-derived growth factor and hypertrophic stimuli in vascular smooth muscle cells: markers for phenotypic modulation and response to injury. J Biol Chem 280(20):19966–19976
Han M, Wen JK, Zheng B, Cheng Y, Zhang C (2006) Serum deprivation results in redifferentiation of human umbilical vascular smooth muscle cells. Am J Physiol Cell Physiol 291(1):C50–C58
Acknowledgments
This work was supported by a grant from Fundamental Research Funds for the Central Universities (No. 302274023).
Author information
Authors and Affiliations
Corresponding author
Additional information
Jiangbin Chen and Lin Xu have contributed equally to this study.
Rights and permissions
About this article
Cite this article
Chen, J., Xu, L. & Huang, C. DHEA inhibits vascular remodeling following arterial injury: a possible role in suppression of inflammation and oxidative stress derived from vascular smooth muscle cells. Mol Cell Biochem 388, 75–84 (2014). https://doi.org/10.1007/s11010-013-1900-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1900-7