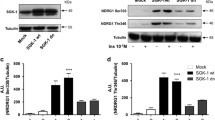
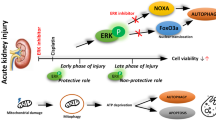
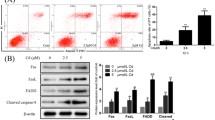
Abstract
It remains elusive whether crosstalk exists among mitochondrial Bax, caspases, and mitogen-activated protein kinases (MAPKs), and whether epidermal growth factor (EGF), which may activate MAPKs, affects ceramide-induced apoptosis through the crosstalk in renal proximal tubular cells (RPTCs). Effect of ceramide on expression of mitochondrial Bax and phosphorylated (p)-ERK, p38MAPK and JNK, that of MAPKs inhibition, and of EGF in the presence or absence of MAPKs inhibition on ceramide-induced apoptosis were examined in HK-2 cells. Apoptosis and expression of mitochondrial Bax and p-MAPKs were measured by Hoechst 33258 staining and Western blotting. C2-ceramide, but not dihydroC2-ceramide, inactive C2-ceramide, induced apoptosis at 24 h. C2-ceramide enhanced the mitochondrial Bax expression at 1 h, which was peaked at 3–6 h and decreased at 24 h, but remained increased, compared to control. An inhibitor of caspases, zVAD-fmk, ameliorated ceramide-induced apoptosis, suggesting a role of caspases for ceramide-induced apoptosis. C2-ceramide enhanced the expression of p-ERK and p-p38MAPK, but not p-JNK, at 1 h, which was increased till 24 h. An inhibitor of ERK, PD98059, or of p38MAPK, SB202190, failed to affect C2-ceramide-induced apoptosis. EGF, which enhanced the expression of p-ERK and p-p38MAPK but not p-JNK, ameliorated C2-ceramide-induced apoptosis without affecting mitochondrial Bax. Inhibition of ERK or p38MAPK failed to abolish the protective effect of EGF on C2-ceramide-induced apoptosis. Mitochondrial Bax and caspases, but not MAPKs, play a role for ceramide-induced apoptosis in RPTCs. EGF ameliorates ceramide-induced apoptosis in Bax- and MAPKs-independent pathways. The mechanism of ceramide-induced apoptosis and anti-apoptotic effect of EGF deserves further investigations.







Similar content being viewed by others
References
Morales A, Lee H, Goñi FM, Kolesnick R, Fernandez-Checa JC (2007) Sphingolipids and cell death. Apoptosis 12:923–939
Ueda N, Kaushal GP, Hong X, Shah SV (1998) Role of enhanced ceramide generation in DNA damage and cell death in chemical hypoxic injury to LLC-PK1 cells. Kidney Int 54:399–406
Ueda N, Camargo SM, Hong X, Basnakian AG, Walker PD, Shah SV (2001) Role of ceramide synthase in oxidant injury to renal tubular epithelial cells. J Am Soc Nephrol 12:2384–2391
Basnakian AG, Ueda N, Hong X, Galitovsky VE, Yin X, Shah SV (2005) Ceramide synthase is essential for endonuclease-mediated death of renal tubular epithelial cells induced by hypoxia-reoxygenation. Am J Physiol Renal Physiol 288:F308–F314
Autret A, Martin SJ (2009) Emerging role for members of the Bcl-2 family in mitochondrial morphogenesis. Mol Cell 36:355–363
Zhang TH, Liu JF, Zhang Y, Li YL, Lu HT, Murata NM, Yamakawa T (2007) Ceramide induces apoptosis in human lung adenocarcinoma A549 cells through mitogen-activated protein kinases. Acta Pharmacol Sin 28:439–445
Kim HJ, Oh JE, Kim SW, Chun YJ, Kim MY (2008) Ceramide induces p38 MAPK-dependent apoptosis and Bax translocation via inhibition of Akt in HL-60 cells. Cancer Lett 260:88–95
Blázquez C, Galve-Roperh I, Guzmán M (2000) De novo-synthesized ceramide signals apoptosis in astrocytes via extracellular signal-regulated kinase. FASEB J 14:2315–2322
Willaime S, Vanhoutte P, Caboche J, Lemaigre-Dubreuil Y, Mariani J, Brugg B (2001) Ceramide-induced apoptosis in cortical neurons is mediated by an increase in p38 phosphorylation and not by the decrease in ERK phosphorylation. Eur J Neurosci 13:2037–2046
Oh HL, Seok JY, Kwon CH, Kang SK, Kim YK (2006) Role of MAPK in ceramide-induced cell death in primary cultured astrocytes from mouse embryonic brain. Neurotoxicology 27:31–38
Jarvis WD, Fornari FA Jr, Auer KL, Freemerman AJ, Szabo E, Birrer MJ, Johnson CR, Barbour SE, Dent P, Grant S (1997) Coordinate regulation of stress- and mitogen-activated protein kinases in the apoptotic actions of ceramide and sphingosine. Mol Pharmacol 52:935–947
Stoica BA, Movsesyan VA, Knoblach SM, Faden AI (2005) Ceramide induces neuronal apoptosis through mitogen-activated protein kinases and causes release of multiple mitochondrial proteins. Mol Cell Neurosci 29:355–371
Kong JY, Klassen SS, Rabkin SW (2005) Ceramide activates a mitochondrial p38 mitogen-activated protein kinase: a potential mechanism for loss of mitochondrial transmembrane potential and apoptosis. Mol Cell Biochem 278:39–51
Falluel-Morel A, Aubert N, Vaudry D, Basille M, Fontaine M, Fournier A, Vaudry H, Gonzalez BJ (2004) Opposite regulation of the mitochondrial apoptotic pathway by C2-ceramide and PACAP through a MAP-kinase-dependent mechanism in cerebellar granule cells. J Neurochem 91:1231–1243
Qiu L, Zhou C, Sun Y, Di W, Scheffler E, Healey S, Wanebo H, Kouttab N, Chu W, Wan Y (2006) Paclitaxel and ceramide synergistically induce cell death with transient activation of EGFR and ERK pathway in pancreatic cancer cells. Oncol Rep 16:907–913
Willaime-Morawek S, Brami-Cherrier K, Mariani J, Caboche J, Brugg B (2003) c-Jun N-terminal kinases/c-Jun and p38 pathways cooperate in ceramide-induced neuronal apoptosis. Neuroscience 119:387–397
Donato NJ, Klostergaard J (2004) Distinct stress and cell destruction pathways are engaged by TNF and ceramide during apoptosis of MCF-7 cells. Exp Cell Res 294:523–533
Henson ES, Gibson SB (2006) Surviving cell death through epidermal growth factor (EGF) signal transduction pathways: implications for cancer therapy. Cell Signal 18:2089–2097
Sheng G, Guo J, Warner BW (2007) Epidermal growth factor receptor signaling modulates apoptosis via p38α MAPK-dependent activation of Bax in intestinal epithelial cells. Am J Physiol Gastrointest Liver Physiol 293:G599–G606
Iwayama H, Sakamoto T, Nawa A, Ueda N (2011) Crosstalk between Smad and MAPKs for the regulation of apoptosis in cyclosporine A-induced renal tubular injury. Nephron Extra 1:178–189
Ganesan V, Colombini M (2010) Regulation of ceramide channels by Bcl-2 family proteins. FEBS Lett 584:2128–2134
Martinou JC, Youle RJ (2011) Mitochondria in apoptosis: Bcl-2 family members and mitochondrial dynamics. Dev Cell 21:92–101
Wolter KG, Hsu YT, Smith CL, Nechushtan A, Xi XG, Youle RJ (1997) Movement of Bax from the cytosol to mitochondria during apoptosis. J Cell Biol 139:1281–1292
Er E, Oliver L, Cartron PF, Juin P, Manon S, Vallette FM (2006) Mitochondria as the target of the pro-apoptotic protein Bax. Biochim Biophys Acta 1757:1301–1311
Düssmann H, Rehm M, Concannon CG, Anguissola S, Würstle M, Kacmar S, Völler P, Huber HJ, Prehn JH (2010) Single-cell quantification of Bax activation and mathematical modeling suggest pore formation on minimal mitochondrial Bax accumulation. Cell Death Differ 17:278–290
Leber B, Lin J, Andrews DW (2007) Embedded together: the life and death consequences of interaction of the Bcl-2 family with membranes. Apoptosis 12:897–911
Sawada M, Nakashima S, Banno Y, Yamakawa H, Takenaka K, Shinoda J, Nishimura Y, Sakai N, Nozawa Y (2000) Influence of Bax or Bcl-2 overexpression on the ceramide-dependent apoptotic pathway in glioma cells. Oncogene 19:3508–3520
Sawai H, Kawai S, Domae N (2004) Reduced expression of Bax in ceramide-resistant HL-60 subline. Biochem Biophys Res Commun 319:46–49
Poppe M, Reimertz C, Münstermann G, Kögel D, Prehn JH (2002) Ceramide-induced apoptosis of D283 medulloblastoma cells requires mitochondrial respiratory chain activity but occurs independently of caspases and is not sensitive to Bcl-xL overexpression. J Neurochem 82:482–494
Lee H, Rotolo JA, Mesicek J, Penate-Medina T, Rimner A, Liao WC, Yin X, Ragupathi G, Ehleiter D, Gulbins E, Zhai D, Reed JC, Haimovitz-Friedman A, Fucks Z, Kolesnick R (2011) Mitochondrial ceramide-rich macrodomains functionalize Bax upon irradiation. Plos One 6:e19783
Tikhomirov O, Carpenter G (2005) Bax activation and translocation to mitochondria mediate EGF-induced programmed cell death. J Cell Sci 118:5681–5690
Payne SG, Brindley DN, Guilbert LJ (1999) Epidermal growth factor inhibits ceramide-induced apoptosis and lowers ceramide levels in primary placental trophoblasts. J Cell Physiol 180:263–270
Acknowledgments
We thank Mrs. M. Miyake for her technical assistance. This study has been supported by the Aichi Kidney Foundation and the Nagono Medical Foundation.
Conflict of interest
None is declared.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Iwayama, H., Ueda, N. Role of mitochondrial Bax, caspases, and MAPKs for ceramide-induced apoptosis in renal proximal tubular cells. Mol Cell Biochem 379, 37–42 (2013). https://doi.org/10.1007/s11010-013-1624-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-013-1624-8