Abstract
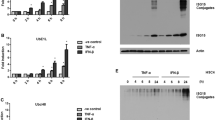
Interferon-stimulated gene15 (ISG15) is the first characterized ubiquitin-like protein, which is strongly induced by type I interferons (IFN-α/β), bacterial endotoxin, and cellular stress. Up-regulation of ISG15 is observed in several cancer cell types and is associated with cancer progression. As many cytokines can influence all stages of tumorigenesis, the elevated expression of ISG15 system may be regulated in cancer cells by inflammatory cytokines. In this study, we showed that TNF-α, but not TGF-β and IL-6, up-regulates levels of both ISG15 and its conjugates in human lung carcinoma A549 and human squamous carcinoma HSC4 cell lines. Induction of ISG15 and its conjugates by TNF-α was dose-dependent and required mediation of p38 MAP kinase and Jak1 through up-regulation of endogenous type I interferon expression. SB202190 (p38 MAPK inhibitor) and Jak1 inhibitor suppressed TNF-α-induced expression of ISG15 and its conjugates. However, only SB202190 inhibited the expression of type I interferons by TNF-α. Although B18R, a soluble type I interferon receptor, totally abolished the effect of exogenous IFN-β, it was unable to inhibit completely the TNF-α-induced ISG15 production. In addition, the initiation of ISG15 induction by TNF-α was detected earlier than that of IFN-β induction. Taken together, TNF-α elicits the induction of ISG15 and ISG15 conjugates not only via the autocrine stimulation of type I interferon expression, but also through a type I interferon-independent pathway. These data provide a possible link between inflammatory response and cancer progression.





Similar content being viewed by others
References
Kumar S, Yoshida Y, Noda M (1993) Cloning of a cDNA which encodes a novel ubiquitin-like protein. Biochem Biophys Res Commun 195:393–399
Haas AL, Ahrens P, Bright PM, Ankel H (1987) Interferon induces a 15-kilodalton protein exhibiting marked homology to ubiquitin. J Biol Chem 262:11315–11323
Zhang D, Zhang DE (2011) Interferon-stimulated gene 15 and the protein ISGylation system. J Interferon Cytokine Res 31:119–130
Yuan W, Krug RM (2001) Influenza B virus NS1 protein inhibits conjugation of the interferon (IFN)-induced ubiquitin-like ISG15 protein. EMBO J 20:362–371
Zhao C, Beaudenon SL, Kelley ML, Waddell MB, Yuan W, Schulman BA, Huibregtse JM, Krug RM (2004) The UbcH8 ubiquitin E2 enzyme is also the E2 enzyme for ISG15, an IFN-alpha/beta-induced ubiquitin-like protein. Proc Natl Acad Sci USA 101:7578–7582
Kim KI, Giannakopoulos NV, Virgin HW, Zhang DE (2004) Interferon-inducible ubiquitin E2, Ubc8, is a conjugating enzyme for protein ISGylation. Mol Cell Biol 24:9592–9600
Zou W, Zhang DE (2006) The interferon-inducible ubiquitin-protein isopeptide ligase (E3) EFP also functions as an ISG15 E3 ligase. J Biol Chem 281:3989–3994
Dastur A, Beaudenon S, Kelley M, Krug RM, Huibregtse JM (2006) Herc5, an interferon-induced HECT E3 enzyme, is required for conjugation of ISG15 in human cells. J Biol Chem 281:4334–4338
Pestka S, Krause CD, Walter MR (2004) Interferons, interferon-like cytokines, and their receptors. Immunol Rev 202:8–32
Silvennoinen O, Ihle JN, Schlessinger J, Levy DE (1993) Interferon-induced nuclear signalling by Jak protein tyrosine kinases. Nature 366:583–585
Kessler DS, Levy DE, Darnell JE Jr (1988) Two interferon-induced nuclear factors bind a single promoter element in interferon-stimulated genes. Proc Natl Acad Sci USA 85:8521–8525
Au WC, Moore PA, Lowther W, Juang YT, Pitha PM (1995) Identification of a member of the interferon regulatory factor family that binds to the interferon-stimulated response element and activates expression of interferon-induced genes. Proc Natl Acad Sci USA 92:11657–11661
Mossman KL, Macgregor PF, Rozmus JJ, Goryachev AB, Edwards AM, Smiley JR (2001) Herpes simplex virus triggers and then disarms a host antiviral response. J Virol 75:750–758
Nicholl MJ, Robinson LH, Preston CM (2000) Activation of cellular interferon-responsive genes after infection of human cells with herpes simplex virus type 1. J Gen Virol 81:2215–2218
Li J, Peet GW, Balzarano D, Li X, Massa P, Barton RW, Marcu KB (2001) Novel NEMO/IkappaB kinase and NF-kappa B target genes at the pre-B to immature B cell transition. J Biol Chem 276:18579–18590
Gessani S, Belardelli F, Pecorelli A, Puddu P, Baglioni C (1989) Bacterial lipopolysaccharide and gamma interferon induce transcription of beta interferon mRNA and interferon secretion in murine macrophages. J Virol 63:2785–2789
Manthey CL, Wang SW, Kinney SD, Yao Z (1998) SB202190, a selective inhibitor of p38 mitogen-activated protein kinase, is a powerful regulator of LPS-induced mRNAs in monocytes. J Leukoc Biol 64:409–417
Desai SD, Haas AL, Wood LM, Tsai YC, Pestka S, Rubin EH, Saleem A, Nur EKA, Liu LF (2006) Elevated expression of ISG15 in tumor cells interferes with the ubiquitin/26S proteasome pathway. Cancer Res 66:921–928
Dinarello CA (2006) The paradox of pro-inflammatory cytokines in cancer. Cancer Metastasis Rev 25:307–313
Germano G, Allavena P, Mantovani A (2008) Cytokines as a key component of cancer-related inflammation. Cytokine 43:374–379
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25:402–408
Loeb KR, Haas AL (1992) The interferon-inducible 15-kDa ubiquitin homolog conjugates to intracellular proteins. J Biol Chem 267:7806–7813
Wajant H, Pfizenmaier K, Scheurich P (2003) Tumor necrosis factor signaling. Cell Death Differ 10:45–65
Alcami A, Symons JA, Smith GL (2000) The vaccinia virus soluble alpha/beta interferon (IFN) receptor binds to the cell surface and protects cells from the antiviral effects of IFN. J Virol 74:11230–11239
Kundu JK, Surh YJ (2008) Inflammation: gearing the journey to cancer. Mutat Res 659:15–30
Ahrens PB, Besancon F, Memet S, Ankel H (1990) Tumour necrosis factor enhances induction by beta-interferon of a ubiquitin cross-reactive protein. J Gen Virol 71:1675–1682
Reis LF, Ho Lee T, Vilcek J (1989) Tumor necrosis factor acts synergistically with autocrine interferon-beta and increases interferon-beta mRNA levels in human fibroblasts. J Biol Chem 264:16351–16354
Yarilina A, Park-Min KH, Antoniv T, Hu X, Ivashkiv LB (2008) TNF activates an IRF1-dependent autocrine loop leading to sustained expression of chemokines and STAT1-dependent type I interferon-response genes. Nat Immunol 9:378–387
Wu Y, Zhou BP (2010) TNF-alpha/NF-kappaB/Snail pathway in cancer cell migration and invasion. Br J Cancer 102:639–644
Siddoo-Atwal C, Haas AL, Rosin MP (1996) Elevation of interferon beta-inducible proteins in ataxia telangiectasia cells. Cancer Res 56:443–447
Rayet B, Gelinas C (1999) Aberrant rel/nfkb genes and activity in human cancer. Oncogene 18:6938–6947
Sing A, Merlin T, Knopf HP, Nielsen PJ, Loppnow H, Galanos C, Freudenberg MA (2000) Bacterial induction of beta interferon in mice is a function of the lipopolysaccharide component. Infect Immun 68:1600–1607
Toshchakov V, Jones BW, Perera PY, Thomas K, Cody MJ, Zhang S, Williams BR, Major J, Hamilton TA, Fenton MJ, Vogel SN (2002) TLR4, but not TLR2, mediates IFN-beta-induced STAT1alpha/beta-dependent gene expression in macrophages. Nat Immunol 3:392–398
Navarro L, David M (1999) p38-dependent activation of interferon regulatory factor 3 by lipopolysaccharide. J Biol Chem 274:35535–35538
Miscia S, Marchisio M, Grilli A, Di Valerio V, Centurione L, Sabatino G, Garaci F, Zauli G, Bonvini E, Di Baldassarre A (2002) Tumor necrosis factor alpha (TNF-alpha) activates Jak1/Stat3-Stat5B signaling through TNFR-1 in human B cells. Cell Growth Differ 13:13–18
Guo D, Dunbar JD, Yang CH, Pfeffer LM, Donner DB (1998) Induction of Jak/STAT signaling by activation of the type 1 TNF receptor. J Immunol 160:2742–2750
Acknowledgments
This work was supported by grants from the Thailand Research Fund (MRG5180019), Faculty of Dentistry, Mahidol University, and Faculty of Medicine Research Fund, Chiang Mai University, Thailand. The authors would like to thank Professor P. Wilairat, Faculty of Science, Mahidol University for critical reading of the manuscript and Mrs. S. Korsuwannawong for assistance in statistical analysis.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
11010_2012_1360_MOESM1_ESM.tif
Supplementary Fig. Two-dimensional Western blot analysis of TNF-α and IFN-β induced A549 cells. Cells were treated with 15 ng/ml TNF-α or 103 IU/ml IFN-β at 37oC for 24 h. Fifty µg of total proteins was precipitated with 3 volumes of cold acetone. and analyzed in the first dimension by Ettan IPGphor isoelectric focusing system (GE Healthcare, Piscataway, NJ, USA) using premade immobilized pH gradient (IPG) strips with pH range 3-10 (GE Healthcare, Piscataway, NJ, USA). The Strips were further analyzed by SDS-PAGE. ISG15 and ISG15 conjugates cross-reactive spots were detected by immunoblotting as described in legend to Fig. 1. (TIFF 564 kb)
Rights and permissions
About this article
Cite this article
Chairatvit, K., Wongnoppavich, A. & Choonate, S. Up-regulation of interferon-stimulated gene15 and its conjugates by tumor necrosis factor-α via type I interferon-dependent and -independent pathways. Mol Cell Biochem 368, 195–201 (2012). https://doi.org/10.1007/s11010-012-1360-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-012-1360-5