Abstract
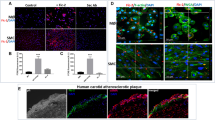
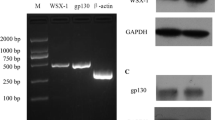
Inflammation and damage promote monocyte adhesion to endothelium and cardiovascular disease (CVD). Elevated inflammation and increased monocyte–endothelial cell interactions represent the initial stages of vascular remodeling associated with a multitude of CVDs. Cathepsins are proteases produced by both cell types that degrade elastin and collagen in arterial walls, and are upregulated in CVD. We hypothesized that the inflammatory cytokine tumor necrosis factor alpha (TNFα) and monocyte binding would stimulate cathepsins K and V expression and activity in endothelial cells that may be responsible for initiating local proteolysis during CVD. Confluent human aortic endothelial cells were stimulated with TNFα or THP-1 monocyte co-cultures, and multiplex cathepsin zymography was used to detect changes in levels of active cathepsins K, L, S, and V. Direct monocyte–endothelial cell co-cultures stimulated with TNFα generated maximally observed cathepsin K and V activities compared to either cell type alone (n = 3, p < 0.05) by a c-Jun N-terminal kinase (JNK)-dependent manner. Inhibition of JNK with SP6000125 blocked upregulation of cathepsin K activity by 49 % and cathepsin V by 81 % in endothelial cells. Together, these data show that inflammatory cues and monocyte–endothelial cell interactions upregulate cathepsin activity via JNK signaling axis and identify a new mechanism to target toward slowing the earliest stages of tissue remodeling in CVD.




Similar content being viewed by others
References
Mestas J, Ley K (2008) Monocyte–endothelial cell interactions in the development of atherosclerosis. Trends Cardiovasc Med 18(6):228–232. doi:10.1016/j.tcm.2008.11.004
Belcher J, Marker P, Weber J, Hebbel R (2000) Activated monocytes in sickle cell disease: potential role in the activation of vascular endothelium. Blood 96:2451–2459
Swirski FK, Libby P, Aikawa E, Alcaide P, Luscinskas FW, Weissleder R, Pittet MJ (2007) Ly-6Chi monocytes dominate hypercholesterolemia-associated monocytosis and give rise to macrophages in atheromata. J Clin Investig 117(1):195–205. doi:10.1172/JCI29950
Li DY, Brooke B, Davis EC, Mecham RP, Sorensen LK, Boak BB, Eichwald E, Keating MT (1998) Elastin is an essential determinant of arterial morphogenesis. Nature 393(6682):276–280. doi:10.1038/30522
Lutgens E, Lutgens SPM, Faber BCG, Heeneman S, Gijbels MMJ, de Winther MPJ, Frederik P, van der Made I, Daugherty A, Sijbers AM, Fisher A, Long CJ, Saftig P, Black D, Daemen MJAP, Cleutjens KBJM (2006) Disruption of the cathepsin K gene reduces atherosclerosis progression and induces plaque fibrosis but accelerates macrophage foam cell formation. Circulation 113(1):98–107. doi:10.1161/CIRCULATIONAHA.105.561449
Platt MO, Ankeny RF, Shi G-P, Weiss D, Vega JD, Taylor WR, Jo H (2007) Expression of cathepsin K is regulated by shear stress in cultured endothelial cells and is increased in endothelium in human atherosclerosis. Am J Physiol Heart Circ Physiol 292(3):H1479–H1486. doi:10.1152/ajpheart.00954.2006
Liu J, Sukhova GK, Sun J-S, Xu W-H, Libby P, Shi G-P (2004) Lysosomal cysteine proteases in atherosclerosis. Arter Thromb Vasc Biol 24(8):1359–1366. doi:10.1161/01.ATV.0000134530.27208.41
Platt MO, Ankeny RF, Jo H (2006) Laminar shear stress inhibits cathepsin L activity in endothelial cells. Arter Thromb Vasc Biol 26(8):1784–1790. doi:10.1161/01.ATV.0000227470.72109.2b
Kafienah W, Brömme D, Buttle DJ, Croucher LJ, Hollander AP (1998) Human cathepsin K cleaves native type I and II collagens at the N-terminal end of the triple helix. Biochem J 331(Pt 3):727–732
Sukhova GKSG, Simon DI, Chapman HA, Libby P (1998) Expression of the elastolytic cathepsins S and K in human atheroma and regulation of their production in smooth muscle cells. J Clin Investig 102:576–583
Yasuda Y, Li Z, Greenbaum D, Bogyo M, Weber E, Brömme D (2004) Cathepsin V, a novel and potent elastolytic activity expressed in activated macrophages. J Biol Chem 279(35):36761–36770. doi:10.1074/jbc.M403986200
Brömme D, Li Z, Barnes M, Mehler E (1999) Human cathepsin V functional expression, tissue distribution, electrostatic surface potential, enzymatic characterization, and chromosomal localization. Biochemistry 38(8):2377–2385. doi:10.1021/bi982175f
Tolosa E, Li W, Yasuda Y, Wienhold W, Denzin LK, Lautwein A, Driessen C, Schnorrer P, Weber E, Stevanovic S, Kurek R, Melms A, Bromme D (2003) Cathepsin V is involved in the degradation of invariant chain in human thymus and is overexpressed in myasthenia gravis. J Clin Investig 112(4):517–526. doi:10.1172/JCI18028
Yang M, Zhang Y, Pan J, Sun J, Liu J, Libby P, Sukhova GK, Doria A, Katunuma N, Peroni OD, Guerre-Millo M, Kahn BB, Clement K, Shi G-P (2007) Cathepsin L activity controls adipogenesis and glucose tolerance. Nat Cell Biol 9(8):970–977. doi:10.1038/ncb1623
Kitamoto S, Sukhova GK, Sun J, Yang M, Libby P, Love V, Duramad P, Sun C, Zhang Y, Yang X, Peters C, Shi G-P (2007) Cathepsin L deficiency reduces diet-induced atherosclerosis in low-density lipoprotein receptor-knockout mice. Circulation 115(15):2065–2075. doi:10.1161/CIRCULATIONAHA.107.688523
Li WA, Barry ZT, Cohen JD, Wilder CL, Deeds RJ, Keegan PM, Platt MO (2010) Detection of femtomole quantities of mature cathepsin K with zymography. Anal Biochem 401(1):91–98. doi:10.1016/j.ab.2010.02.035
Wilder CL, Park K-Y, Keegan PM, Platt MO (2011) Manipulating substrate and pH in zymography protocols selectively distinguishes cathepsins K, L, S, and V activity in cells and tissues. Arch Biochem Biophys 516(1):52–57. doi:10.1016/j.abb.2011.09.009
Lecaille F, Weidauer E, Juliano MA, Brömme D, Lalmanach G (2003) Probing cathepsin K activity with a selective substrate spanning its active site. Biochem J 375(Pt 2):307–312. doi:10.1042/BJ20030468
Ruettger A, Schueler S, Mollenhauer JA, Wiederanders B (2008) Cathepsins B, K, and L are regulated by a defined collagen type II peptide via activation of classical protein kinase C and p38 MAP kinase in articular chondrocytes. J Biol Chem 283(2):1043–1051. doi:10.1074/jbc.M704915200
Gacko MGS (1998) Expression of the elastolytic cathepsins S and K in human atheroma and regulation of their production in smooth muscle cells. Clin Chem 36:449–452
Burns-Kurtis CL, Olzinski AR, Needle S, Fox JH, Capper EA, Kelly FM, McQueney MS, Romanic AM (2004) Cathepsin S expression is up-regulated following balloon angioplasty in the hypercholesterolemic rabbit. Cardiovasc Res 62(3):610–620. doi:10.1016/j.cardiores.2004.02.002
Garcia-Touchard A, Henry TD, Sangiorgi G, Spagnoli LG, Mauriello A, Conover C, Schwartz RS (2005) Extracellular proteases in atherosclerosis and restenosis. Arter Thromb Vasc Biol 25(6):1119–1127. doi:10.1161/01.ATV.0000164311.48592.da
Abdul-Hussien H, Soekhoe RG, Weber E, von der Thusen JH, Kleemann R, Mulder A, van Bockel JH, Hanemaaijer R, Lindeman JH (2007) Collagen degradation in the abdominal aneurysm: a conspiracy of matrix metalloproteinase and cysteine collagenases. Am J Pathol 170(3):809–817. doi:10.2353/ajpath.2007.060522
Ha S-D, Martins A, Khazaie K, Han J, Chan BMC, Kim SO (2008) Cathepsin B is involved in the trafficking of TNF-alpha-containing vesicles to the plasma membrane in macrophages. J Immunol 181(1):690–697
Thoma B, Grell M, Pfizenmaier K, Scheurich P (1990) Identification of a 60-kD tumor necrosis factor (TNF) receptor as the major signal transducing component in TNF responses. J Exp Med 172(4):1019–1023
Chang Y-L, Chen C-L, Kuo C-L, Chen B-C, You J-S (2010) Glycyrrhetinic acid inhibits ICAM-1 expression via blocking JNK and NF-kappaB pathways in TNF-alpha-activated endothelial cells. Acta Pharmacol Sin 31(5):546–553. doi:10.1038/aps.2010.34
Hui C, Like W, Yan F, Tian X, Qiuyan W, Lifeng H (2010) S-allyl-l-cysteine sulfoxide inhibits tumor necrosis factor-alpha induced monocyte adhesion and intercellular cell adhesion molecule-1 expression in human umbilical vein endothelial cells. Anat Rec (Hoboken) 293(3):421–430. doi:10.1002/ar.21070
Pang M, Martinez AF, Fernandez I, Balkan W, Troen BR (2007) AP-1 stimulates the cathepsin K promoter in RAW 264.7 cells. Gene 403(1–2):151–158. doi:10.1016/j.gene.2007.08.007
Acknowledgments
The authors of this study would like to thank Eric Kopfle, Alex Miller, and Sindhuja Surapeneni for assistance with data collection. This work was funded by Georgia Tech startup funds and NIH New Innovator grant #1DP2OD007433-01 (MOP). The project described was supported by Award Number DP2OD007433 from the Office of the Director, National Institutes of Health. The content is solely the responsibility of the authors and does not necessarily represent the official views of the Office of the Director, National Institutes of Health or the National Institutes of Health. PMK was supported by an NSF graduate research fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Keegan, P.M., Wilder, C.L. & Platt, M.O. Tumor necrosis factor alpha stimulates cathepsin K and V activity via juxtacrine monocyte–endothelial cell signaling and JNK activation. Mol Cell Biochem 367, 65–72 (2012). https://doi.org/10.1007/s11010-012-1320-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-012-1320-0