Abstract
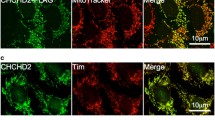
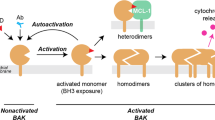
Bax induces mitochondrial-dependent cell apoptosis signals in mammalian cells. However, the mechanism of how Bax is kept inactive is not fully elucidated. Here, we identify FIH1 as a potential interactor of Bax through mass spectrometry analysis. Coimmunoprecipitation and GST pull-down experiments show that FIH1 can directly interact with Bax. Bax-mediated apoptosis is suppressed by FIH1 overexpression, but accelerated by FIH1 deficiency. FIH1 functions as a cytosol retention factor of Bax, blocking Bax translocation from cytosol to mitochondria in response to apoptotic stimuli. Overall, there results unveil a novel role of FIH1 in the regulation of Bax-mediated apoptosis.





Similar content being viewed by others
References
Schofield CJ, Ratcliffe PJ (2008) Oxygen sensing by HIF hydroxylases. Nat Rev Mol Cell Bio 5:343–354
Bracken CP, Fedele AO, Linke S et al (2006) Cell-specific regulation of hypoxia-inducible factor (HIF)-1 and HIF-2 stabilization and transactivation in a graded oxygen environment. J Biol Chem 281:22575–22585
Yan Q, Bartz S, Mao M et al (2007) The hypoxia-inducible factor 2α N-terminal and C-terminal transactivation domains cooperate to promote renal tumorigenesis in vivo. Mol Cell Bio 27:2012–2092
Mahon PC, Hirota K, Semenza GL (2001) FIH-1: a novel protein that interacts with HIF-1 and VHL to mediate repression of HIF-1 transcriptional activity. Gen Dev 15:2675–2686
Ferguson JE III, Wu Y, Smith K et al (2007) ASB4 is a hydroxylation substrate of FIH and promotes vascular differentiation via an oxygen-dependent mechanism. Mol Cell Biol 27:6407–6419
Coleman ML, McDonough MA, Hewitson KS et al (2007) Asparaginyl hydroxylation of the Notch ankyrin repeat domain by factor inhibiting hypoxia-inducible factor. J Biol Chem 282:24027–24038
Zheng X, Linke S, Dias JM et al (2008) Interaction with factor inhibiting HIF-1 defines an additional mode of cross-coupling between the Notch and hypoxia signaling pathways. Proc Natl Acad Sci USA 105:3368–3373
Edmondson RD, Vondriska TM, Biederman KJ et al (2002) Protein kinase Cε signaling complexes include metabolism and transcription/translation-related proteins: complimentary electrophoretic separation techniques with LC/MS/MS. Mol Cell Proteomics 1:421–433
Domon B, Aebersold R (2006) Mass spectrometry and protein analysis. Sci STKE 312:212–217
Oltval ZN, Milliman CL, Korsmeyer SJ (1993) Bcl-2 heterodimerizes in vivo with a conserved homolog, Bax, that accelerates programed cell death. Cell 74:609–619
Simonian PL, Grillot DAM, Merino R et al (1996) Bax can antagonize Bcl-XL during etoposide and cisplatin-induced cell death independently of its heterodimerization with Bcl-XL. J Biol Chem 271:22764–22772
Wei MC, Zong WX, Cheng EHY et al (2001) Proapoptotic BAX and BAK: a requisite gateway to mitochondrial dysfunction and death. Science 292:727–730
Stolze IP, Tian YM, Appelhoff RJ et al (2004) Genetic analysis of the role of the asparaginyl hydroxylase factor inhibiting hypoxia-inducible factor (HIF) in regulating HIF transcriptional target genes. J Biol Chem 279:42719–42725
Deng Y, Wu X (2003) Peg3/Pw1 promotes p53-mediated apoptosis by inducing Bax translocation from cytosol to mitochondria. Proc Natl Acad Sci USA 97:12050–12055
Chittenden T, Harrington EA, O’Connor R et al (1995) Induction of apoptosis by the Bcl-2 homologue. Nature 374:733–736
Weil M, Jacobson MD, Coles HS et al (1996) Constitutive expression of the machinery for programmed cell death. J Cell Biol 133:1053–1059
Nechushtan A, Smith CL, Lamensdorf I et al (2001) Bax and Bak coalesce into novel mitochondria-associated clusters during apoptosis. J Cell Biol 153:1265–1276
Metzen E, Berchner-Pfannschmidt U, Stengel P et al (2003) Intracellular localisation of human HIF-1α hydroxylases: implications for oxygen sensing. J Cell Sci 116:1319–1326
Lancaster DE, McNeill LA, McDonough MA et al (2004) Disruption of dimerization and substrate phosphorylation inhibit factor inhibiting hypoxia-inducible factor (FIH) activity. Biochem J 383:429–437
Hewitson KS, Holmes SL, Ehrismann D et al (2008) Evidence that two enzyme-derived histidine ligands are sufficient for iron binding and catalysis by factor inhibiting HIF (FIH). J Biol Chem 283:25971–25978
Lindsten T, Ross AJ, King A et al (2000) The combined functions of proapoptotic Bcl-2 family members bak and bax are essential for normal development of multiple tissues. Mol Cell 6:1389–1399
Wolter KG, Hsu YT, Smith CL et al (1997) Movement of Bax from the cytosol to mitochondria during apoptosis. J Cell Biol 139:1281–1292
Webby CJ, Wolf A, Gromak N et al (2009) Jmjd6 catalyses lysyl-hydroxylation of U2AF65, a protein associated with RNA splicing. Science 325:90–93
Cory S, Huang DCS, Adams JM (2003) The Bcl-2 family: roles in cell survival and oncogenesis. Oncogene 22:8590–8607
Hsu YT, Wolter KG, Youle RJ (1997) Cytosol-to-membrane redistribution of Bax and Bcl-XL during apoptosis. Proc Natl Acad Sci USA 94:3668–3672
Hsu YT, Youle RJ (1998) Bax in murine thymus is a soluble monomeric protein that displays differential detergent-induced conformations. J Biol Chem 273(17):10777–10783
Nechushtan A, Smith CL, Hsu YT et al (1999) Conformation of the Bax C-terminus regulates subcellular location and cell death. EMBO J 18(9):2330–2341
Desagher S, Osen-Sand A, Nichols A et al (1999) Bid-induced conformational change of Bax is responsible for mitochondrial cytochrome c release during apoptosis. J Cell Biol 144:891–901
Kim H, Rafiuddin-Shah M, Tu HC et al (2006) Hierarchical regulation of mitochondrion-dependent apoptosis by Bcl-2 subfamilies. Nat Cell Biol 8:1348–1358
Cartron PF, Priault M, Oliver L et al (2003) The N-terminal end of Bax contains a mitochondrial-targeting signal. J Biol Chem 278:11633–11641
Chipuk JE, Maurer U, Green DR et al (2003) Pharmacologic activation of p53 elicits Bax-dependent apoptosis in the absence of transcription. Cancer Cell 4:371–381
Perez D, White E (1998) E1B 19K inhibits Fas-mediated apoptosis through FADD-dependent sequestration of FLICE. J Cell Biol 141:1255–1266
Sundararajan R, White E (2001) E1B 19K blocks Bax oligomerization and tumor necrosis factor α-mediated apoptosis. J Virol 75:7506–7516
Guo B, Zhai D, Cabezas E et al (2003) Humanin peptide suppresses apoptosis by interfering with Bax activation. Nature 423:456–461
Sawada M, Sun W, Hayes P et al (2003) Ku70 suppresses the apoptotic translocation of Bax to mitochondria. Nat Cell Biol 5:320–329
Acknowledgments
We thank the Harvard medical school to provide the technical platform, and the joint PhD student Education Program between Harvard University (USA) and Tongji University (China). This study was supported by State Scholarship Fund of China (NO: 2009626129) from China Scholarship Council (CSC). This work was also supported by the ‘973’ Program Fund of China (2007CB512100) and the ‘863’ Program Fund of China (2007AA02Z438).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Yan, B., Kong, M. & Chen, Yh. Prevention of apoptosis by the interaction between FIH1 and Bax. Mol Cell Biochem 348, 1–9 (2011). https://doi.org/10.1007/s11010-010-0631-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11010-010-0631-2