Abstract
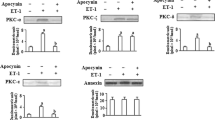
Treatment of bovine pulmonary artery smooth muscle with the O2 •− generating system hypoxanthine plus xanthine oxidase stimulated MMP-2 activity and PKC activity; and inhibited Na+ dependent Ca2+ uptake in the microsomes. Pretreatment of the smooth muscle with SOD (the O2 •− scavenger) and TIMP-2 (MMP-2 inhibitor) prevented the increase in MMP-2 activity and PKC activity, and reversed the inhibition of Na+ dependent Ca2+ uptake in the microsomes. Pretreatment with calphostin C (a general PKC inhibitor) and rottlerin (a PKCδ inhibitor) prevented the increase in PKC activity and reversed O2 •− caused inhibition of Na+ dependent Ca2+ uptake without causing any change in MMP-2 activity in the microsomes of the smooth muscle. Treatment of the smooth muscle with the O2 •− generating system revealed, respectively, 36 kDa RACK-1 and 78 kDa PKCδ immunoreactive protein profile along with an additional 38 kDa immunoreactive fragment in the microsomes. The 38 kDa band appeared to be the proteolytic fragment of the 78 kDa PKCδ since pretreatment with TIMP-2 abolished the increase in the 38 kDa immunoreactive fragment. Co-immunoprecipitation of PKCδ and RACK-1 demonstrated O2 •− dependent increase in PKCδ-RACK-1 interaction in the microsomes. Immunoblot assay elicited an immunoreactive band of 41 kDa G i α in the microsomes. Treatment of the smooth muscle tissue with the O2 •− generating system causes phosphorylation of G i α in the microsomes and pretreatment with TIMP-2 and rottlerin prevented the phosphorylation. Pretreatment of the smooth muscle tissue with pertussis toxin reversed O2 •− caused inhibition of Na+ dependent Ca2+ uptake without affecting the protease activity and PKC activity in the microsomes. We suggest the existence of a pertussis toxin sensitive G protein mediated mechanism for inhibition of Na+ dependent Ca2+ uptake in microsomes of bovine pulmonary artery smooth muscle under O2 •− triggered condition, which is regulated by PKCδ dependent phosphorylation and sensitive to TIMP-2 for its inhibition. (Mol Cell Biochem xxx: 107–117, 2005)
Similar content being viewed by others
Abbreviations
- O2 •− :
-
superoxide
- SOD:
-
superoxide dismutase
- ER:
-
endoplasmic reticulum
- MMP-2:
-
matrix metalloprotease-2
- TIMP-2:
-
tissue inhibitor of metalloprotease-2
- PKC:
-
protein kinase C
- RACK-1:
-
receptor for activated C kinase-1
- E-64d:
-
trans-epoxysuccinyl-L-leucylamido-3-methylbutane ethyl ester
- G i α:
-
α-subunit of the inhibitory G protein
- HBPS:
-
Hank's buffered physiological saline
- IAP:
-
islet activating protein (pertussis toxin)
References
Chakraborti T, Ghosh, SK, Michael JR, Batabyal SK, Chakraborti S: Targets of oxidative stress in the cardiovascular system. Mol Cell Biochem 187: 1–10, 1998
Chakraborti T, Das S, Mandal M, Mandal A, Chakraborti S: Ca2+ dynamics under oxidant stress in the cardiovascular system. In: K.B. Storey, J.M. Storey (eds). Cellular and Molecular Responses to Stress, Vol 2, Elsevier Science, New York, 2002, pp 213–228
Bose R, Li Y, Roberts D: Na+/Ca2+ exchange in activated and non-activated human platelets. Ann. N Y Acad Sci 976: 350–353, 2002
Roychoudhury S, Ghosh SK, Chakraborti T, Chakraborti S: Role of hydroxyl radical in the oxidant H2O2-mediated Ca2+ release from pulmonary smooth muscle mitochondria. Mol Cell Biochem 159: 95–103, 1996
Galis ZS, Sukhova GK, Libby P: Microscopic localization of active proteases by in situ zymography: detection of matrix metalloproteinase activity in vascular tissue. FASEB J 9: 974–980, 1995
Ghosh SK, Chakraborti T, Michael JR, Chakraborti S: Oxidant-mediated proteolytic activation of Ca2+ATPase in microsomes of pulmonary smooth muscle. FEBS Lett 387: 171–174, 1996
Lehotsky J, Kaplan P, Matejovicova M, Murin R, Racay P, Raeymaekers L: Ion transport systems as targets of free radicals during ischemia-reperfusion injury. Gen Physiol Biophys 21: 31–37, 2002
Farrukh IS, Michael JR, Summer WR, Adkinson NF Jr, Gurtner GH: Thromboxane-induced pulmonary vasoconstriction: involvement of calcium. J Appl Physiol 58: 34–44, 1985
Tate RM, Vanbenthuysen KM, Shasby DM, McMurty IF, Repine JE: Oxygen radical-mediated permeability edema and vasoconstriction in isolated perfused rabbit lungs. Am Rev Respir Dis 126: 802–806, 1982
Mandal A, Chakraborti T, Das S, Ghosh B, Ghosh AN, Chakraborti S: Matrix metalloproteinase-2 mediated inhibition of Na+ dependent Ca2+ uptake by superoxide radical (O2 •−) in microsomes of pulmonary smooth muscle. IUBMB Life 56: 267–276, 2004
Shan GX: Selective protein kinase C inhibitors and their applications. Curr Drug Targets Cardiovasc Haematol Disord 3: 301–307, 2003
Eto A, Akita Y, Saido TC, Suzuki K, Kawashima S: The role of the calpain-calpastatin system in thyrotropin-releasing hormone-induced selective down-regulation of a protein kinase C isozyme, nPKC ε, in rat pituitary GH4C1 cells. J Biol Chem 270: 25115–251120, 1995
Balafanova Z, Bolli R, Zhang J, Zheng Y, Pass JM, Bhatnagar A, Tang X-L, Wang O, Cardwell E, Ping P: Nitric oxide (NO) induces nitration of protein kinase Cε (PKCε), facilitating PKCε translocation via enhanced PKCε-RACK2 interactions: a novel mechanism of no-triggered activation of PKCε. J Biol Chem 277: 15021–15027, 2002
Pass JM, Zheng Y, Wead WB, Zhang J, Li RC, Bolli R, Ping P: PKC-ε activation induces dichotomous cardiac phenotypes and modulates PKC-ε-RACK interactions and RACK expression. Am J Physiol Heart Circ Physiol 280: H946–H955, 2001
Mochly-Rosen D, Wu G, Hahn H, Osinska H, Liron T, Lorenz JN, Yatani A, Robbins J, Dorn GW 2nd: Cardiotrophic effects of protein kinase C-ε: analysis by in vivo modulation of PKC-ε translocation. Circ Res 86: 1173–1179, 2000
Ronco, AM, Moraga PF, Llanos MN: Arachidonic acid release from rat Leydig cells: the involvement of G protein, phospholipase A2 and regulation of cAMP production. J Endocrinol 172: 95–104, 2002
Iorio P, Gresele P, Stasi M, Nucciarelli F, Vezza R, Nenci GG, Goracci G: Protein kinase C inhibitors enhance G-protein induced phospholipase A2 activation in intact human platelets. FEBS Lett 381: 244–248, 1996
Katada T, Gilman AG, Watanabe Y, Bauer S, Jakobs KH: Protein kinase C phosphorylates the inhibitory guanine-nucleotide binding regulatory component and apparently suppresses its function in hormonal inhibition of adenylate cyclase. Eur J Biochem 151: 431–437, 1985
Dixon BS, Breckon R, Burke C, Anderson RJ: Phorbol esters inhibit adenylate cyclase activity in cultured collecting tubular cells. Am J Physiol 254: C183–C191, 1988
Choi YJ, Park JW, Woo JH, Kim YH, Lee SH, Lee JM, Kwon TK: Failure to activate caspase 3 in phorbol ester-resistant leukemia cells is associated with resistance to apoptotic cell death. Cancer Lett 182: 183–191, 2002
Emoto Y, Manome Y, Meinhardt G, Kisaki H, Kharbanda S, Robertson M, Ghayur T, Wong WW, Kamen R, Weichselbaum R: Proteolytic activation of protein kinase C-δ by an ICE-like protease in apoptotic cells. EMBO J 14: 6148–6156, 1995
Hashimoto E, Yamamura H: Further studies on the ionic strength-dependent proteolytic activation of protein kinase C in rat liver plasma membrane by endogenous trypsin-like protease. J Biochem (Tokyo) 106: 1041–1048, 1989
Anantharam V, Kitazawa M, Wagner J, Kaul S, Kanthasamy AG: Caspase-3-dependent proteolytic cleavage of protein kinase C-δ is essential for oxidative stress-mediated dopaminergic cell death after exposure to methylcyclopentadienyl manganese tricarbonyl. J Neurosci 22: 1738–1751, 2002
Majumdar S, Rossi MW, Fujiki T, Phillips WA, Disa S, Queen CF, Johnston RB Jr, Rosen OM, Corkey BE, Korchak HM: Protein kinase C isotypes and signaling in neutrophils. Differential substrate specificities of a translocatable calcium- and phospholipid-dependent beta-protein kinase C and a phospholipid-dependent protein kinase which is inhibited by long chain fatty acyl coenzyme A. J Biol Chem 266: 9285–9294, 1991
Chakraborti S, Michael JR: Role of protein kinase C in oxidant–mediated activation of phospholipase A2 in rabbit pulmonary arterial smooth muscle cells. Mol Cell Biochem 122: 9–15, 1993
Exton JH: Mechanisms of action of calcium-mobilizing agonists: some variations on a young theme. FASEB J 2: 2670–2676, 1988
Olson NC, Kruse-Elliott KT, Whorton AR, Dodam JR: Pertussis toxin attenuates platelet-activating factor-induced pulmonary hemodynamic alterations in pigs. Am J Physiol 264: L213–L221, 1993
Chakraborti T, Ghosh SK, Michael JR, Chakraborti S: Role of an aprotinin-sensitive protease in the activation of Ca2+ATPase by superoxide radical (O2 •−) in microsomes of pulmonary smooth muscle. Biochem J 317: 885–890, 1996
Mandal A, Chakraborti T, Choudhury R, Ghosh B, Ghosh AN, Das S, Chakraborti S: Role of MMP-2 in inhibiting Na+ dependent Ca2+ uptake by H2O2 in microsomes isolated from pulmonary smooth muscle. Mol Cell Biochem 270: 79–87, 2005
Masui Y, Takemoto T, Sakakibara S, Hori H, Nagai Y: Synthetic substrates for vertibrate collagenase. Biochem Med 17: 215–221, 1977
Billlings PC, Habres JM, Liao DC, Tuttle SW: Human fibroblasts contain a proteolytic activity which is inhibited by the Bowman-Burk protease inhibitor. Cancer Res 51: 5539–5543, 1991
Dixon IMC, Kaneko M, Hata T, Panagia V, Dhalla NS: Alterations in cardiac membrane calcium transport during oxidative stress. Mol Cell Biochem 99: 125–133, 1990
Kitano T, Go M, Kikkawa U, Nishizuka Y: Assay and purification of protein kinase C. Methods Enzymol 124: 349–352, 1986
Towbin H, Staehlin T, Gordon J: Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets, procedure and some applications. Proc Natl Acad Sci (USA) 76: 4350–4354, 1979
Neyses L, Reinlib L, Carafoli E: Phosphorylation of the Ca2+ pumping ATPase of heart sarcolemma and erythrocyte plasma membrane by cAMP-dependent protein kinase. J Biol Chem 260: 10283–10287, 1985
Crouch MF, Lapetina EG: A role for Gi in control of thrombin receptor-phospholipase C coupling in human platelets. J Biol Chem 263: 3363–3371, 1988
Takehara Y, Kanno T, Yoshioka T, Inoue M, Utsumi K: Oxygen dependent regulation of mitochondrial energy metabolism by nitric oxide. Arch Biochem Biophys 323: 27–32, 1985
Smith PK, Krohn RI, Hermanson GT, Malli A, Gartner FH, Provenzano MD, Fujimoto EK, Goeke NM, Olson BJ, Klenk DC: Measurement of protein using bicinchoninic acid. Anal Biochem 150: 76–85, 1985
Daniel WW: A Foundation for Analysis in the Health Sciences. In: W.W. Daniel (ed). Biostatistics, Estimation. Wiley, New York, 1978, pp 121–157
Rajagopalan S, Meng XP, Ramasamy S, Harrison DG, Galis ZS: Reactive oxygen species produced by macrophage-derived foam cells regulate the activity of the vascular metalloproteinases in vitro. Implications for atherosclerotic plaque stability. J Clin Invest 98: 2572–2579, 1996
Yoshida K, Sorimachi Y, Fujiwara M, Hironaka K: Calpain is implicated in rat myocardial injury after ischemia or reperfusion. Jpn Circ J 59: 40–48, 1995
McGowan EB, Becker E, Detwiler TC: Inhibition of calpain in intact platelets by the thiol protease inhibitor E-64d. Biochem Biophys Res Commun 158: 432–435, 1989
Park HY, Wu H, Killoran CE, Gilchrest BA: The receptor for activated C-kinase-I (RACK-I) anchors activated PKC-β on melanosomes. J Cell Sci 117: 3659–3668, 2004
Schechtman D, Craske ML, Kheifets V, Meyer T, Schechtman J, Mochly-Rosen D: A critical intramolecular interaction for protein kinase C-ε translocation. J Biol Chem 279: 15831–15840, 2004
Inagaki K, Hahn HS, Dorn GW 2nd, Mochly-Rosen D: Additive protection of the ischemic heart ex vivo by combined treatment with δ-protein kinase C inhibitor and ε-protein kinase C activator. Circulation 108: 869–875, 2003
Gruss OJ, Feick P, Frank R, Dobberstein B: Phosphorylation of components of the ER translocation site. Eur J Biochem 260: 785–793, 1999
Alexander SR, Kishimoto TK, Walcheck B: Effects of selective protein kinase C inhibitors on the proteolytic down-regulation of L-selectin from chemoattractant-activated neutrophils. J Leukoc Biol 67: 415–422, 2000
Cressman CM, Mohan PS, Nixon RA, Shea TB: Proteolysis of protein kinase C: mM and microM calcium-requiring calpains have different abilities to generate, and degrade the free catalytic subunit, protein kinase M. FEBS Lett 367: 223–227, 1995
Chakraborti T, Das S, Chakraborti S: Proteolytic activation of protein kinase Cα by peroxynitrite in stimulating cytosolic phospholipase A2 in pulmonary endothelium: involvement of a pertussis toxin sensitive protein. Biochemistry (USA) 44: 5246–5257, 2005
Chakraborti S, Michael JR, Chakraborti T: Role of an aprotinin-sensitive protease in protein kinase C alpha-mediated activation of cytosolic phospholipase A2 by calcium ionophore (A23187) in pulmonary endothelium. Cell Signal 16: 751–762, 2004
Kuroda T, Mikawa K, Mishima H, Kishimoto A: H1 histone stimulates limited proteolysis of protein kinase C subspecies by calpain I. J Biochem (Tokyo) 110: 364–368, 1991
Kikuchi H, Imajoh-Ohmi S: Antibodies specific for proteolyzed forms of protein kinase C alpha. Biochim Biophys Acta 1269: 253–259, 1995
Chakravarthy BR, Whitfield JF, Durkin JP: Inactive membrane protein kinase Cα: a possible target for receptor signaling. Biochem J 304: 809–816, 1994
Sakurai Y, Onishi Y, Tanimoto Y, Kizaki H: Novel protein kinase C-δ isoform insensitive to caspase-3. Biol Pharm Bull 24: 973–977, 2001
Burch RM: Diacylglycerol stimulates phospholipase A2 from Swiss 3T3 fibroblasts. FEBS Lett 234: 283–286, 1988
Yatomi Y, Arata Y, Tada S, Kume S, Ui M: Phosphorylation of the inhibitory guanine-nucleotide-binding protein as a possible mechanism of inhibition by protein kinase C of agonist-induced Ca2+ mobilization in human platelet. Eur J Biochem 205: 1003–1009, 1992
Ginnan R, Singer HA: PKC-δ-dependent pathways contribute to PDGF-stimulated ERK1/2 activation in vascular smooth muscle. Am J Physiol Cell Physiol 288: C1193–1201, 2005
Upmacis RK, Deeb RS, Resnick MJ, Lindenbaum R, Gamss C, Mittar D, Hajjar DP: Involvement of the mitogen-activated protein kinase cascade in peroxynitrite-mediated arachidonic acid release in vascular smooth muscle cells. Am J Physiol Cell Physiol 286: C1271–C1280, 2004
Xing M, Insel PA: Protein kinase C-dependent activation of cytosolic phospholipase A2 and mitogen activated protein kinase by alpha 1-adrenergic receptors in Madin-Darby canine kidney cells. J Clin Invest 97: 1302–1310, 1996
Mueller E, van Breemen C: Role of intracellular Ca2+ sequestration in β-adrenergic relaxation of a smooth muscle. Nature 281: 682–683, 1979
Kaneko M, Matsumoto Y, Hayashi H, Kobayashi A, Yamazaki N: Oxygen free radicals and calcium homeostasis in the heart. Mol Cell Biochem 139: 91–100, 1994
Murtha YM, Allen BM, Orr JA: The role of protein kinase C in thromboxane induced pulmonary artery vasoconstriction. J Biomed Sci 6: 293–295, 1999
Serfilippi G, Ferro TJ, Johnson A: Activation of protein kinase C mediates altered pulmonary vasoreactivity induced by tumor necrosis factor alpha. Am J Physiol 267: L282–L290, 1994
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chakraborti, S., Mandal, A., Das, S. et al. Role of MMP-2 in PKCδ-mediated inhibition of Na+ dependent Ca2+ uptake in microsomes of pulmonary smooth muscle: Involvement of a pertussis toxin sensitive protein. Mol Cell Biochem 280, 107–117 (2005). https://doi.org/10.1007/s11010-005-8237-9
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s11010-005-8237-9