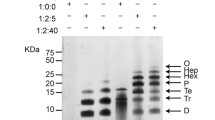
Abstract
Sodium dodecyl sulfate (SDS) was introduced in polyacrylamide gel electrophoresis (PAGE) due to its capacity of helping proteins to become fully denatured and dissociated from each other. Numerous studies which have been undertaken, using electrospray ionization mass spectrometry (ESI–MS), reported on the process of peptide oligomerization. Many of these investigations have included tetraglycine (H2N–Gly–Gly–Gly–Gly–COOH; G4) as model peptide. The aim of this research is to investigate the effect of SDS on G4 oligomerization, and, especially, to emphasize the dismantling of oligomers under micellar conditions. In water, G4 peptide develops dimers and oligomers, which can also be evidenced in high proportion by MS in the gas phase. Although our results show that SDS is able to reduce the proportion of G4 oligomers, the aqueous G4-SDS system may contain G4 dimers, G4-SDS adducts alongside with the expected monomers and some alkaline metal adducts. The mechanism by which SDS disassembled G4 dimers, which includes sodium ion affinity toward negatively charged carboxyl and sulfonyl groups, was also discussed. Amyloid-β peptide1–40 conformation changed considerably and, especially, the proportion of α-helical populations increased upon SDS binding in a concentration-dependent manner. Molecular dynamics studies confirmed the tendency of Aβ molecules to form α-helical conformers, as the CD and FTIR studies showed.









Similar content being viewed by others
References
Ahmed M, Davis J, Aucoin D, Sato T, Ahuja S, Aimoto S, Elliott JI, Van Nostrand WE, Smith SO (2010) Structural conversion of neurotoxic amyloid-β1–42 oligomers to fibrils. Nat Struct Mol Biol 17:561–567. doi:10.1038/nsmb.1799
Bemporad F, Chiti F (2012) Protein misfolded oligomers: experimental approaches, mechanism of formation, and structure-toxicity relationships. Chem Biol 19:315–327. doi:10.1016/j.chembiol.2012.02.003
Bhuyan AK (2010) On the mechanism of SDS-induced protein denaturation. Biopolymers 93:186–199. doi:10.1002/bip.21318
Carvalho do Lago LC, Matias AC, Nomura CS, Cerchiaro G (2011) Radical production by hydrogen peroxide/bicarbonate and copper uptake in mammalian cells: modulation by Cu (II) complexes. J Inorg Biochem 105:189–194. doi:10.1016/j.jinorgbio.2010.10.017
Coles M, Vicknell W, Watson AA, Fairlie D, Craik DJ (1998) Solution structure of amyloid β-peptide (1–40)ox in a water–micelle environment. Is the membrane-spanning domain where we think it is? Biochemistry 37:11064–11077. doi:10.1021/bi972979f
Drochioiu G, Manea M, Dragusanu M, Murariu M, Dragan ES, Petre BA, Mezo G, Przybylski M (2009) Interaction of beta-amyloid(1–40) peptide with pairs of metal ions: An electrospray ion trap mass spectrometric model study. Biophys Chem 144:9–20. doi:10.1016/j.bpc.2009.05.008
Drochioiu G, Tudorachi L, Murariu M (2015) NOSH aspirin may have a protective role in Alzheimer’s disease. Med Hypotheses 84:262–267. doi:10.1016/j.mehy.2015.01.008
Dutta A, Kim TY, Moeller M, Wu J, Alexiev U, Seetharaman JK (2010) Characterization of membrane protein non-native states. 2. The SDS-unfolded states of rhodopsin. Biochemistry 49:6329–6340. doi:10.1021/bi100339x
Fenn JB, Mann M, Meng CK, Wong SF, Whitehouse CM (1989) Electrospray ionization for mass spectrometry of large biomolecules. Science 246:64–71. doi:10.1126/science.2675315
Gao X, Yang S, Zhao C, Ren Y, Wei D (2014) Artificial multienzyme supramolecular device: highly ordered self-assembly of oligomeric enzymes in vitro and in vivo. Angew Chem Int Ed 53:14027–14030. doi:10.1002/anie.201405016
Gitlin IK, Gudiksen L, Whitesides GM (2006) Peracetylated bovine carbonic anhydrase (BCA-Ac18) is kinetically more stable than native BCA to sodium dodecyl sulfate. J Phys Chem B 110:2372–2377. doi:10.1021/jp055699f
Harmeier A, Wozny C, Rost BR, Munter LM, Hua H, Georgiev O, Beyermann M, Hildebrand PW, Weise C, Schaffner W, Schmitz D, Multhaup G (2009) Role of amyloid-β glycine 33 in oligomerization, toxicity, and neuronal plasticity. J Neurosci 29:7582–7590. doi:10.1523/JNEUROSCI.1336-09.2009
Harris RP, Jacobson TA (2006) Omega 3 fatty acids. Novinka Books. doi: 10.1081/E-EDS-120022075
Hubin EN, van Nuland AJ, Broersen K, Pauwels K (2014) Transient dynamics of Aβ contribute to toxicity in Alzheimer’s disease. Cell Mol Life Sci 71:3507–3521. doi:10.1007/s00018-014-1634-z
Karplus M, McCammon JA (2002) Molecular dynamics simulations of biomolecules. Nat Struct Mol Biol 9:646–652. doi:10.1038/nsb0902-646
Khan JM, Qadeer A, Chaturvedi SK, Ahmad E, Rehman SA (2012) SDS can be utilized as an amyloid inducer: a case study on diverse proteins. PLoS ONE 7:e29694. doi:10.1371/journal.pone.0029694
Kong J, Yu S (2007) Fourier transform infrared spectroscopic analysis of protein secondary structures. Acta Biochim Biophys Sin 39:549–559. doi:10.1111/j.1745-7270.2007.00320.x
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685. doi:10.1038/227680a0
Lindberg M, Gräslund A (2001) The position of the cell penetrating peptide penetratin in SDS micelles determined by NMR. FEBS Lett 497:39–44. doi:10.1016/S0014-5793(01)02433-4
Masters CL, Selkoe DJ (2012) Biochemistry of amyloid β-protein and amyloid deposits in Alzheimer disease. Cold Spring Harb Perspect Med 2:a006262. doi:10.1101/cshperspect.a006262
Meier C, Wu Y, Pramanik G, Weil T (2014) Self-assembly of high molecular weight polypeptide copolymers studied via diffusion limited aggregation. Biomacromolecules 15:219–227. doi:10.1021/bm401506a
Michaux C, Pouyez J, Wouters J, Privé GG (2008) Protecting role of cosolvents in protein denaturation by SDS: a structural study. BMC Struct Biol 8:29. doi:10.1186/1472-6807-8-29
Miller Y, Ma B, Nussinov R (2011) The unique Alzheimer’s β-amyloid triangular fibril has a cavity along the fibril axis under physiological conditions. J Am Chem Soc 133:2742–2748. doi:10.1021/ja1100273
Montserret R, McLeish MJ, Böckmann A, Geourjon C, Penin F (2000) Involvement of electrostatic interactions in the mechanism of peptide folding induced by sodium dodecyl sulfate binding. Biochemistry 39:8362–8373. doi:10.1021/bi000208x
Murariu M (2013) ESI-MS study of self-assembly-formed tetraglycine macrocyclic ligand complex of Cu (II). Int J Mass Spectrom 351:12–22. doi:10.1016/j.ijms.2013.05.009
Murariu M, Przybylski M, Stefanescu R, Schlosser G, Hudecz F, Drochioiu G (2008) Copper-induced oligomerization of peptides: a model study. Eur J Mass Spectrom 13:331–337. doi:10.1255/ejms.889
Ngo S, Guo Z (2011) Key residues for the oligomerization of Aβ42 protein in Alzheimer’s disease. Biochem Biophys Res Commun 414:512–516. doi:10.1016/j.bbrc.2011.09.097
Nielsen AD, Arleth L, Westh P (2005) Analysis of protein–surfactant interactions: a titration calorimetric and fluorescence spectroscopic investigation of interactions between Humicola insolens cutinase and an anionic surfactant. BBA-Proteins Proteom 1752:124–132. doi:10.1016/j.bbapap.2005.08.001
Nielsen MM, Andersen KK, Westh P, Otzen DE (2007) Unfolding of β-sheet proteins in SDS. Biophys J 92:3674–3685. doi:10.1529/biophysj.106.101238
Otzen DE (2002) Protein unfolding in detergents: effect of micelle structure, ionic strength, pH, and temperature. Biophys J 83:2219–2230. doi:10.1016/S0006-3495(02)73982-9
Pastoriza-Gallego M, Rabah L, Gibrat G, Thiebot B, Gisou van der Goot F, Auvray L, Betton JM, Pelta J (2011) Dynamics of unfolded protein transport through an aerolysin pore. J Am Chem Soc 133:2923–2931. doi:10.1021/ja1073245
Pountney DL, Henehan CJ, Vasak M (1995) Establishing isostructural metal substitution in metalloproteins using 1H NMR, circular dichroism, and Fourier transform infrared spectroscopy. Protein Sci 4:1571–1576. doi:10.1002/pro.5560040815
Rahimi F, Shanmugam A, Bitan G (2008) Structure–function relationships of pre-fibrillar protein assemblies in Alzheimer’s disease and related disorders. Curr Alzheimer Res 5:319–341. doi:10.2174/156720508784533358
Raz Y, Miller Y (2013) Interactions between Aβ and mutated tau lead to polymorphism and induce aggregation of Aβ-mutated tau oligomeric complexes. PLoS ONE 8:e73303. doi:10.1371/journal.pone.0073303
Song WJ, Tezcan FA (2014) A designed supramolecular protein assembly with in vivo enzymatic activity. Science 346:1525–1528. doi:10.1126/science.1259680
Valencia A, Pazos F (2002) Computational methods for the prediction of protein interactions. Curr Opin Struct Biol 12:368–373. doi:10.1016/S0959-440X(02)00333-0
Watt AD, Perez KA, Rembach A, Sherrat NA, Hung LW, Johanssen T, McLean CA, Kok WM, Hutton CA, Fodero-Tavoletti M, Masters CL, Villemagne VL, Barnham KJ (2013) Oligomers, fact or artefact? SDS-PAGE induces dimerization of β-amyloid in human brain samples. Acta Neuropathol 125:549–564. doi:10.1007/s00401-013-1083-z
Acknowledgments
This work was supported by Romanian Government (UEFISCDI Contract IDEI 313/2011). PhD student Laura Ion gratefully acknowledges the strategic grant POSDRU/159/1.5/S/137750 from EU.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Laura Ion, Catalina Ionica Ciobanu, Manuela Murariu, Vasile-Robert Gradinaru, and Gabi Drochioiu declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Rights and permissions
About this article
Cite this article
Ion, L., Ciobanu, C.I., Murariu, M. et al. SDS-induced Peptide Conformational Changes: From Triglycyl-glycine to Amyloid-β Oligomers Associated with Alzheimer’s Disease. Int J Pept Res Ther 22, 45–55 (2016). https://doi.org/10.1007/s10989-015-9483-7
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10989-015-9483-7