Abstract
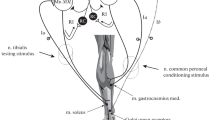
We investigated the extent to which activity induced by chronic electrical stimulation could restore the mass and contractile function of rabbit tibialis anterior (TA) muscles that had undergone atrophy as a result of prolonged denervation. Denervation was carried out by selectively interrupting the motor nerve branches to the ankle dorsiflexors in one hind limb. Stimulators were implanted, with electrodes on the superficial and deep surfaces of the denervated TA muscle. Ten weeks later, the mass and mid-belly cross-sectional area (CSA) of TA muscles subjected to denervation alone had fallen to approximately 40% of normal. At this stage, stimulators in the other rabbits were activated for 1 h/day to deliver 20-ms rectangular bipolar constant-current pulses of 4 mA amplitude at 20 Hz with a duty cycle of 1s ON/2s OFF, a total of 24,000 impulses/day. The animals were examined after a further 2, 6 or 10 weeks. Stimulation restored the wet weight of the denervated muscles to values not significantly different to those of normal, innervated controls. It increased CSA from 39% to 66% of normal, and there was a commensurate increase in maximum isometric tetanic force from 27% to 50% of normal. Light and electron microscopic examination revealed a marked improvement in the size, packing, and internal organization of the stimulated-denervated muscle fibres, suggestive of an ongoing process of restoration. Excitability, contractile speed, power, and fatigue resistance had not, however, been restored to normal levels after 10 weeks of stimulation. Similar results were found for muscles that had been denervated for 39 weeks and then stimulated for 12 weeks. The study demonstrates worthwhile benefits of long-term electrical stimulation in the treatment of established denervation atrophy.











Similar content being viewed by others
References
al-Amood WS, Lewis DM, Schmalbruch H (1991) Effects of chronic electrical stimulation on contractile properties of long-term denervated rat skeletal muscle. J Physiol 441:243–256
Ashley Z, Sutherland H, Lanmuller H, Russold MF, Unger E, Bijak M, Mayr W, Boncompagni S, Protasi F, Salmons S, Jarvis JC (2007) Atrophy, but not necrosis, in rabbit skeletal muscle denervated for periods up to one year. Am J Physiol 292:C440–C451
Aydin MA, Mackinnon SE, Gu XM, Kobayashi J, Kuzon WM Jr. (2004) Force deficits in skeletal muscle after delayed reinnervation. Plast Reconstr Surg 113:1712–1718
Borisov AB, Dedkov EI, Carlson BM (2001) Interrelations of myogenic response, progressive atrophy of muscle fibers, and cell death in denervated skeletal muscle. Anat Rec 264:203–218
Carlson BM, Borisov AB, Dedkov EI, Dow DE, Kostrominova TY (2002) The biology and restorative capacity of long-term denervated skeletal muscle. Basic Appl Myol 12:247–254
Dedkov EI, Kostrominova TY, Borisov AB, Carlson BM (2001) Reparative myogenesis in long-term denervated skeletal muscles of adult rats results in a reduction of the satellite cell population. Anat Rec 263:139–154
Dennis RG, Dow DE, Faulkner JA (2003) An implantable device for stimulation of denervated muscles in rats. Med Eng Phys 25:239–253
Dow DE, Cederna PS, Hassett CA, Kostrominova TY, Faulkner JA, Dennis RG (2004) Number of contractions to maintain mass and force of a denervated rat muscle. Muscle Nerve 30:77–86
Eisenberg BR, Salmons S (1981) The reorganisation of subcellular structure in muscle undergoing fast-to-slow type transformation: a stereological study. Cell Tiss Res 220:449–471
Flucher BE, Takekura H, Franzini-Armstrong C (1993) Development of the excitation-contraction coupling apparatus in skeletal muscle: association of sarcoplasmic reticulum and transverse tubules with myofibrils. Dev Biol 160:135–147
Forbes MS, Plantholt BA, Sperelakis N (1977) Cytochemical staining procedures selective for sarcotubular systems of muscle: modifications and applications. J Ultrastruct Res 60:306–327
Franzini-Armstrong C (1991) Simultaneous maturation of transverse tubules and sarcoplasmic reticulum during muscle differentiation in the mouse. Dev Biol 146:353–363
Kelly AM (1980) T tubules in neonatal rat soleus and extensor digitorum longus muscles. Dev Biol 80:501–505
Kern H, Hofer C, Strohhofer M, Mayr W, Richter W, Stöhr H (1999) Standing up with denervated muscles in humans using functional electrical stimulation. Artif Org 23:447–452
Kern H, Salmons S, Mayr W, Rossini K, Carraro U (2005) Recovery of long-term denervated human muscles induced by electrical stimulation. Muscle Nerve 31:98–101
Kostrominova TY, Dow DE, Dennis RG, Miller RA, Faulkner JA (2005) Comparison of gene expression of two-month denervated, two-month stimulated-denervated and control rat skeletal muscles. Physiol Genomics 22:227–243
Lanmüller H, Ashley Z, Unger E, Sutherland H, Reichel M, Russold M, Jarvis J, Mayr W, Salmons S (2005) Implantable device for long-term electrical stimulation of denervated muscles in rabbits. Med Biol Eng Comput 43:535–540
Lewis DM, al-Amood WS, Schmalbruch H (1997) Effects of long-term phasic electrical stimulation on denervated soleus muscle: guinea-pig contrasted with rat. J Musc Res Cell Motil 18:573–586
Lu DX, Huang SK, Carlson BM (1997) Electron microscopic study of long-term denervated rat skeletal muscle. Anat Rec 248:355–365
Misawa A, Shimada Y, Matsunaga T, Sato K (2001) The effects of therapeutic electric stimulation on acute muscle atrophy in rats after spinal cord injury. Arch Phys Med Rehabil 82:1596–1603
Salmons S, Sréter FA (1976) Significance of impulse activity in the transformation of skeletal muscle type. Nature 263:30–34
Salmons S, Gale DR, Sréter FA (1978) Ultrastructural aspects of the transformation of muscle fibre type by long term stimulation: changes in Z-discs and mitochondria. J Anat 127:17–31
Schmalbruch H, al-Amood WS, Lewis DM (1991) Morphology of long-term denervated rat soleus muscle and the effect of chronic electrical stimulation. J Physiol 441:233–241
Sommer JR, Waugh RA (1976) The ultrastructure of the mammalian cardiac muscle cell–with special emphasis on the tubular membrane systems. A review. Am J Pathol 82:192–232
Viguie CA, Lu DX, Huang SK, Rengen H, Carlson BM (1997) Quantitative study of the effects of long-term denervation on the extensor digitorum longus muscle of the rat. Anat Rec 248:346–354
Wyndaele M, Wyndaele J-J (2006) Incidence, prevalence and epidemiology of spinal cord injury: what learns a worldwide literature survey? Spinal Cord 44:523–529
Acknowledgements
This work was supported by European Commission Project “RISE”, Contract Nr. QLG5-CT-2001-02191. The authors are grateful for many valuable discussions with their colleagues in this network and particularly for the clinical leadership provided by Dr Helmut Kern.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ashley, Z., Salmons, S., Boncompagni, S. et al. Effects of chronic electrical stimulation on long-term denervated muscles of the rabbit hind limb. J Muscle Res Cell Motil 28, 203–217 (2007). https://doi.org/10.1007/s10974-007-9119-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10974-007-9119-4