Abstract
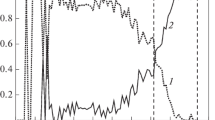
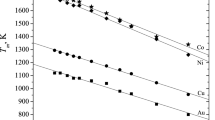
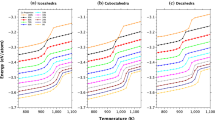
An approach combining atomistic molecular dynamics (MD) and thermodynamic simulations has been applied to predict the distribution of components in binary Ni–Cu and Au–Ag nanoparticles consisting of 2000 atoms (of about 4 nm in size). The term ‘thermodynamic simulation’ has referred to solving, in some approximations, the Butler equation for a core–shell particle model. Both atomistic and thermodynamic approaches predict the surface segregation of Cu atoms in Ni–Cu nanoparticles and segregation of Ag to the surface of Au–Ag nanoalloys. Then, contrary to the Ni–Cu systems, some Au–Ag nanoparticles demonstrated an onion-like structure with the outer Ag monolayer. The results of MD and thermodynamic simulations agree with each other and with some available direct and indirect experimental data.








Similar content being viewed by others
References
Buffat Ph, Borel J-P. Size effect on the melting temperature of gold particles. Phys Rev A. 1976;13:2287–98.
Hori H, Teranishi T, Taki M, Yamada S, Miyake M, Yamamoto Y. Magnetic properties of nano-particles of Au, Pd and Pd/Ni alloys. J Magn Magn Mater. 2001;226–230:1910–1.
Heurlin M, Magnusson MH, Lindgren D, Ek M, Wallenberg LR, Deppert K, Samuelson L. Continuous gas-phase synthesis of nanowires with tunable properties. Nature. 2012;492:90–4.
Zare M, Ketabchi M. Effect of chromium element on transformation, mechanical and corrosion behavior of thermomechanically induced Cu–Al–Ni shape-memory alloys. J Therm Anal Calorim. 2017;127:2113–23.
Chowdhury ND, Ghosh KS. Calorimetric studies of Ag–Sn–Cu dental amalgam alloy powders and their amalgams. J Therm Anal Calorim. 2017;130:623–37.
Hamana D, Hamana M. Precipitation and dissolution-grains growth effects and kinetics during non-isothermal heating of deformed Cu–7 mass% Ag alloy. J Therm Anal Calorim. 2016;123:1063–71.
Alexeev OS, Gates BC. Supported bimetallic cluster catalysts. Ind Eng Chem Res. 2003;42:1571–87.
Guisbiers G, Abudukelimu G, Hourlier D. Size-dependent catalytic and melting properties of platinum-palladium nanoparticles. Nanoscale Res Lett. 2011;6:396.
Paz-Borbón LO. Computational studies of transition metal nanoalloys. Berlin: Springer; 2011.
Li X, Chen Q, McCue I, Snyder J, Crozier P, Erlebacher J, Sieradzki K. Dealloying of noble-metal alloy nanoparticles. Nano Lett. 2014;14:2569–77.
Calagua A, Alarcon H, Paraguay F, Rodriguez J. Synthesis and characterization of bimetallic gold-silver core-shell nanoparticles: a green approach. Adv Nanoparticles. 2015;4:116–21.
Cleri F, Rosato V. Tight-binding potentials for transition metals and alloys. Phys Rev B. 1993;40:22–33.
Han SW, Kim Y, Kim K. Dodecanethiol-derivatized Au/Ag bimetallic nanoparticles: TEM, UV/VIS, XPS, and FTIR analysis. J Colloid Interface Sci. 1998;208:272–8.
Butler JAV. Thermodynamics of the surface of solutions. Proc R Soc. 1932;A135:348–63.
Rusanov AI. The essence of the new approach to the equation of the monolayer state. Colloid J. 2007;69:131–43.
Kaptay G. Modelling equilibrium grain boundary segregation, grain boundary energy and grain boundary segregation transition by the extended Butler equation. J Mater Sci. 2016;51:1738–55.
Pellicer E, Varea A, Sivaraman KM, Pane S, Surinash S, Baro MD, Nogues J, Nelson BJ, Sort J. Grain boundary segregation and interdiffusion effects in the nickel-copper alloys: an effective means to improve the thermal stability of nanocrystalline nickel. ACS Appl Mater Interfaces. 2011;3:2265–74.
Kaptay G. On the partial surface tension of components of a solution. Langmuir. 2015;31:5796–804.
Korozs J, Kaptay G. Derivation of the Butler equation from the requirement of the minimum Gibbs energy of a solution phase, taking into account its surface area. Coll Surf A. 2017;533:296–301.
Kaptay G. On the negative surface tension of solution and on spontaneous emulsification. Langmuir. 2017;33:10550–60.
Foiles SM. Calculation of the surface segregation of Ni-Cu alloys with the use of the embedded-atom method. Phys Rev B. 1985;32:7685–93.
Zhou XW, Johnson RA, Wadley HNG. Misfit-energy-increasing dislocations in vapor-deposited CoFe/NiFe multilayers. Phys Rev B. 2004;69:144113.
Grochola G, Russo SP, Snook IK. On fitting a gold embedded atom method potential using the force matching method. J Chem Phys. 2005;123:204719.
Williams PL, Mishin Y, Hamilton JC. An embedded-atom potential for the Cu-Ag system. Model Simul Mater Sci Eng. 2006;14:817–33.
Lewis LJ, Jensen P, Barrat J-L. Melting, freezing and coalescence of gold nanonoclusters. Phys Rev B. 1997;56:2248–57.
Qi Y, Cagin T, Johnson WL, Goddard WA. Melting and crystallization in Ni nanoclusters: The mesoscale regime. J Chem Phys. 2001;114:385–94.
Samsonov VM, Bembel AG, Shakulo OV, Vasilyev SA. Comparative molecular dynamics study of melting and crystallization of Ni and Au nanoclusters. Crystallogr Rep. 2014;59:580–5.
Samsonov VM, Vasilyev SA, Talyzin IV, Ryzhkov YuA. On reasons for the hysteresis of melting and crystallization of nanoparticles. J Exp Theor Phys. 2016;103:94–9.
Dick K, Dhanasekaran T, Xhang Z, Meisel D. Size-dependent melting of silica-encapsulated gold nanoparticles. J Am Chem Soc. 2002;124:2312–7.
Castro T, Reifenberger R, Choi E, Andres RP. Size-dependent melting temperature of individual nanometer-sized metallic clusters. Phys Rev B. 1990;42:8548–56.
Wang L, Zhang Y, Bian X, Chen Y. Melting of Cu nanoclusters by molecular dynamics simulation. Phys Lett A. 2003;310:197–202.
Gafner SL, Kosterin SV, Gafner JJ. Formation of structural modifications in copper nanoclusters. Phys Solid State. 2007;49:1558–63.
Peters KF, Cohen JB, Chung Y-W. Melting of Pb nanocrystals. Phys Rev B. 1998;21:13430–8.
Samsonov VM, Vasilyev SA, Bembel AG. Size dependence of the melting temperature of metallic nanoclusters from the viewpoint of the thermodynamic theory of similarity. Phys Met Metallogr. 2016;117:749–55.
Handbook of physical quantities, ed. by IS Grigorev, EZ Meilikhov. Boca Raton: CRC Press LLC; 1997.
Samsonov VM. Conditions for the applicability of a thermodynamic description of highly disperse and microheterogeneous systems. Russ J Phys Chem. 2002;76:1863–7.
Samsonov VM, Bazulev AN, Sdobnyakov N. On applicability of Gibbs thermodynamics to nanoparticles. Centr Eur J Phys. 2003;1:474–84.
Guggenheim MA. Modern thermodynamics by the method of Willard Gibbs. London: Methuen and Co Ltd; 1933.
Alchagirov AB, Alchairov BB, Taova TM, Khokonov KhB. Surface energy and surface tension of solid and liquid metals Recommended values. Trans Join Weld Res Inst Osaka Univ. 2001;30:287–91.
Wilson NT, Johnston RL. A theoretical study of atom ordering in coper-gold nanoalloy clusters. J Mater Chem. 2002;12:2913–22.
Acknowledgements
The work was supported by the Ministry of Education and Science of the Russian Federation in the framework of the State Program in the Field of the Research Activity (No. 3.5506.2017/BP) and by Russian Foundation for Basic Research (Projects No. 16-33-60171 and No. 18-03-00132).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Samsonov, V.M., Bembel, A.G., Kartoshkin, A.Y. et al. Molecular dynamics and thermodynamic simulations of segregation phenomena in binary metal nanoparticles. J Therm Anal Calorim 133, 1207–1217 (2018). https://doi.org/10.1007/s10973-018-7245-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-018-7245-4