Abstract
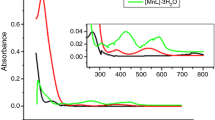
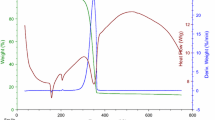
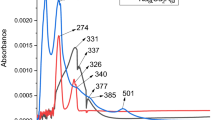
Coordination compounds of transition metals with norfloxacin (Nor) have potential to improve its effectiveness, as already discussed for some compounds found in the literature. The complexes in the solid state were prepared by precipitation of the respective metal cation (M = Mn(II), Co(II), Ni(II), Cu(II) and Zn(II)) with a norfloxacin sodium salt solution. The thermal behavior under oxidative and pyrolysis conditions was investigated employing thermogravimetry and differential thermal analysis (TG/DTG–DTA) and evolved gas analysis (EGA/TG-FTIR), and the complexes were characterized by elemental analysis (EA), EDTA complexometric titration, infrared spectroscopy (FTIR) and X-ray powder diffraction (XRD). By these results, the minimum formula was established as [M(Nor)2(H2O)2]·nH2O, where n = 3 (Ni), 2.5 (Mn Co, Zn), or 2 (Cu). The evolved gases identified during thermal decomposition of Ni and Cu complexes were ethylene, CO2 and CO for Ni; CO2 and ethane for Cu. Also, antimicrobial activity of the complexes was evaluated by in vitro susceptibility test using the agar diffusion method, and the results were compared with the uncomplexed molecule. It was found that norfloxacin complexation modifies its antibacterial activity. The activity depends on the type of metal ion and microorganism; Mn(II), Co(II) and Zn(II) complexes significantly increased activity against the tested gram-negative bacteria.






Similar content being viewed by others
Change history
21 February 2018
In the original publication, the fifth author’s given name was misspelled as “Gabriel”. The corrected name is given in this erratum. The original article has been corrected.
References
Mazuel C. Norfloxacin. Anal Profiles Drug Subst. 1991; 557–600. http://linkinghub.elsevier.com/retrieve/pii/S0099542808605407.
Shaikh AR, Giridhar R, Yadav MR. Bismuth-norfloxacin complex: synthesis, physicochemical and antimicrobial evaluation. Int J Pharm. 2007;332:24–30.
Takahata M, Nishino T. DNA gyrase of Staphylococcus aureus and inhibitory effect of quinolones on its activity. Antimicrob Agents Chemother. 1988;32:1192–5.
Garnayak S, Patel S. Oxidative degradation of norfloxacin by a lipophilic oxidant, cetyltrimethylammonium permanganate in water-acetonitrile medium: a kinetic and mechanistic study. J Mol Liq. 2015;209:327–35. https://doi.org/10.1016/j.molliq.2015.06.003.
Zhu Y, Shu G, Yang Y, Dong Q, Zou L. Synthesis and electrochemiluminescence properties of a new ternary terbium complex and its application for the determination of norfloxacin. J Electroanal Chem. 2014;727:113–9. https://doi.org/10.1016/j.jelechem.2014.05.006.
Kamble R, Sharma S, Mehta P. Norfloxacin mixed solvency based solid dispersions: An in vitro and in vivo investigation. J Taibah Univ Sci. 2017;11:512–22. http://linkinghub.elsevier.com/retrieve/pii/S1658365517300122.
Efthimiadou EK, Psomas G, Sanakis Y, Katsaros N, Karaliota A. Metal complexes with the quinolone antibacterial agent N-propyl-norfloxacin: synthesis, structure and bioactivity. J Inorg Biochem. 2007;101:525–35.
Živec P, Perdih F, Turel I, Giester G, Psomas G. Different types of copper complexes with the quinolone antimicrobial drugs ofloxacin and norfloxacin: structure, DNA- and albumin-binding. J Inorg Biochem. 2012;117:35–47.
Sadeek SA. Synthesis, thermogravimetric analysis, infrared, electronic and mass spectra of Mn(II), Co(II) and Fe(III) norfloxacin complexes. J Mol Struct. 2005;753:1–12.
Sadeek SA, Refat MS, Hashem HA. Complexation and thermogravimetric investigation on tin(II) and tin(IV) with norfloxacin as antibacterial agent. J Coord Chem. 2006;59:759–75.
Ahmadi F, Saberkari M, Abiri R, Motlagh HM, Saberkari H. In vitro evaluation of Zn–Norfloxacin complex as a potent cytotoxic and antibacterial agent, proposed model for DNA binding. Appl Biochem Biotechnol. 2013;170:988–1009. https://doi.org/10.1007/s12010-013-0255-6.
Badea M, Olar R, Silvestro L, Maurer M, Uivarosi V. Synthesis, spectral and thermal studies of the sodium salts of some Ru(III) complexes with quinolone antibiotics. J Therm Anal Calorim. 2017;127:721–9.
Mehta JV, Gajera SB, Patel DD, Patel MN. Synthesis, spectral investigation and development of tetrahedral copper(II) complexes as artificial metallonucleases and antimalarial agents. Appl Organomet Chem. 2015;29:357–67. https://doi.org/10.1002/aoc.3299.
Mehta JV, Gajera SB, Patel MN. Antimalarial, antimicrobial, cytotoxic, DNA interaction and SOD like activities of tetrahedral copper(II) complexes. Spectrochim Acta Part A Mol Biomol Spectrosc. 2015;136:1881–92. http://linkinghub.elsevier.com/retrieve/pii/S138614251401600X.
Sarkar K, Sen K. Some drugs in action: metal ions do influence the activity! Int J Pharm Sci Res. 2015;6:1–13.
Refat MS. Synthesis and characterization of norfloxacin-transition metal complexes (group 11, IB): spectroscopic, thermal, kinetic measurements and biological activity. Spectrochim Acta Part A Mol Biomol Spectrosc. 2007;68:1393–405.
Refat MS, Mohamed GG. Ti(IV), Cr(III), Mn(II), and Ni (II) complexes of the norfloxacin antibiotic drug: spectroscopic and thermal characterizations. J Chem Eng Data. 2010;55:3239–46. https://doi.org/10.1021/je100064h.
Wang Y-J, Lin Q-Y, Feng J, Wang N. Diaquabis (norfloxacinato) manganese(II) 2,2′-bipyridine solvate tetrahydrate. Acta Crystallogr Sect E Struct Reports Online. International Union of Crystallography; 2009;65:m806–m806. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2969423/.
Teixeira JA, Nunes WDG, Fernandes RP, do Nascimento ALCS, Caires FJ, Ionashiro M. Thermal behavior in oxidative and pyrolysis conditions and characterization of some metal p-aminobenzoate compounds using TG–DTA, EGA and DSC-photovisual system. J Anal Appl Pyrolysis, 2017. http://linkinghub.elsevier.com/retrieve/pii/S0165237017306526.
Suzuki Y, Muraishi K, Ito H. Thermal decomposition of manganese(II) dicarboxylate anhydrides in various atmospheres. Thermochim Acta. 1995;258:231–41.
Xu ZP, Zeng HC. Thermal evolution of cobalt hydroxides: a comparative study of their various structural phases. J Mater Chem. 1998;8:2499–506. http://xlink.rsc.org/?DOI=a804767g.
Stein SE. “Infrared spectra” by NIST mass spec data center. In: Linstrom PJ, Mallard WG, editors. NIST Chem. WebBook, NIST Stand. Ref. Database Number 69. Gaithersburg, MD: National Institute of Standards and Technology; [cited 2017 Sep 12]. http://webbook.nist.gov/chemistry/.
Sahoo S, Chakraborti CK, Mishra SC, Nanda UN, Naik S. FTIR and XRD investigations of some fluoroquinolones. Int J Pharm Pharm Sci. 2011;3:165–70.
Huang X-F, Zhang Z-H, Zhang Q-Q, Wang L-Z, He M-Y, Chen Q, et al. Norfloxacin salts with benzenedicarboxylic acids: charge-assisted hydrogen-bonding recognition and solubility regulation. CrystEngComm. 2013;15:6090–100.
Gouvea LR, Martins DA, Batista DDGJ, de Maria Nazaré CS, Louro SR, Barbeira PJ, et al. Norfloxacin Zn(II)-based complexes: acid base ionization constant determination, DNA and albumin binding properties and the biological effect against Trypanosoma cruzi. Biometals. 2013;26:813–25.
Martins DA, Gouvea LR, Muniz GSV, Louro SRW, Batista D da GJ, Soeiro MDNC, et al. Norfloxacin and N-Donor mixed-ligand Copper(II) complexes: synthesis, albumin interaction, and anti-Trypanosoma cruzi activity. Bioinorg Chem Appl 2016;2016:1–11. http://www.hindawi.com/journals/bca/2016/5027404/.
Refat MS, Mohamed GG, De Farias RF, Powell AK, El-Garib MS, El-Korashy SA, et al. Spectroscopic, thermal and kinetic studies of coordination compounds of Zn(II), Cd(II) and Hg(II) with norfloxacin. J Therm Anal Calorim. 2010;102:225–32.
Deacon G, Phillips RJ. Relationships between the carbon–oxygen stretching frequencies of carboxylato complexes and the type of carboxylate coordination. Coord Chem Rev. 1980 [cited 2014 Apr 28];33:227–50. http://www.sciencedirect.com/science/article/pii/S0010854500804555.
Sha J-Q, Li X, Qiu H-B, Zhang Y-H, Yan H. Nickel complexes of the different quinolone antibacterial drugs: synthesis, structure and interaction with DNA. Inorg Chim Acta. 2012;383:178–84. http://linkinghub.elsevier.com/retrieve/pii/S0020169311009108.
Huber PC, Reis GP, Amstalden MCK, Lancellotti M, Almeida WP. Synthesis, spectroscopic characterizations and antimicrobial activity of copper and zinc complexes of levofloxacin, ciprofloxacin and 3-carboxy-4-quinolone. Polyhedron. Elsevier Ltd. 2013;57:14–9.
Acknowledgements
The authors thank FAPESP (Proc. 2017/14936-9), CNPq (Proc. 421469/2016-1) and CAPES Foundation (Brazil) for financial support and PhD Patricia Neves Mendes (UNIFAL-MG) for the aid in the statistical tests.
Author information
Authors and Affiliations
Corresponding author
Additional information
The original version of this article was revised: The fifth author’s given name was misspelled as “Gabriel”. The author name is corrected in the article.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nunes, W.D.G., do Nascimento, A.L.C.S., Moura, A. et al. Thermal, spectroscopic and antimicrobial activity characterization of some norfloxacin complexes. J Therm Anal Calorim 132, 1077–1088 (2018). https://doi.org/10.1007/s10973-018-7019-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-018-7019-z