Abstract
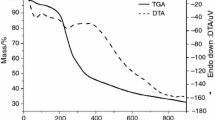
Urtica dioica L. has been used by Brazilian population as an herbal medicine to treat several diseases, such as respiratory allergies. Thus, this study aimed to characterize the U. dioica plant drug samples in different particle sizes and its extract by thermogravimetry (TG), differential thermal analysis (DTA) and pyrolysis coupled to gas chromatography and mass spectrometry (Pyr–GC/MS). The plant drug samples were named UD42, UD100, UD200 and UD400 according to mesh. The extracts were dried by spray dryer at 160 °C and oven-drying at 42 °C. The all samples were submitted to dynamic TG in two different atmospheres (nitrogen and air), TG isothermal in the air and DTA in the nitrogen. The kinetic parameters were determined by Ozawa model and Arrhenius equation. The molecules degradable at 250, 350 and 450 °C were identified by Pyr–GC/MS. The dynamic TG curves showed five decomposition steps for all samples and six for extracts in the nitrogen. In the air showed six decomposition steps for all samples. The sample with small-sized particles (UD400) had greater loss of the total mass, take less time for degradation of 5% of the mass at 30 °C, showed lower enthalpy and activation energy than the others plant drug samples. The dry extract through spray dryer showed higher thermal stability than extract dried in oven-drying. Were identified 54 different molecules in all plant drug samples and 42 in extracts. TG, DTA and Pyr–GC/MS are useful auxiliary tools to characterize vegetable raw material for the production of phytotherapeutic drugs.






Similar content being viewed by others
Abbreviations
- A :
-
Frequency factor
- DTA:
-
Differential thermal analysis
- E a :
-
Activation energy
- EI:
-
Electron impact
- K :
-
Rate constant
- m/z :
-
Mass-to-charge ratio
- N :
-
Reaction order
- Pyr–GC/MS:
-
Pyrolysis coupled to gas chromatography interfaced with mass spectrometry
- R :
-
Gas constant (8.314 J K−1 mol−1)
- RT:
-
Retention time
- T :
-
Temperature
- TG:
-
Thermogravimetry
- UD:
-
Urtica dioca
References
Araújo KRM, Kerntopf MR, Oliveira DR, Menezes IRA, Brito Júnior FE. Medicinal plants in the treatment of respiratoty diseases in childhood: a view from popular knowledge. Rev Rene. 2012;13:659–66.
Silveira PF, Bandeira MAM, Arrais PSD. Farmacovigilância e reações adversas às plantas medicinais e fitoterápicos: uma realidade. Braz J Pharmacogn. 2008;18:618–26.
Pedrosa RC, Yunes RA, Cechinel Filho V. Fármacos e fitoterápicos: a necessidade do desenvolvimento da indústria de fitoterápicos e fitofármacos no Brasil. Quim Nova. 2001;24:147–52.
Bisht S, Snehlata B, Bisht NS. Urtica dioica (L.): an undervalued, economically important plant. Agric Sci Res J. 2002;2:250–2.
Anderson BE, Miller CJ, Adams DR. Stinging nettle dermatitis. Am J Contact Dermat. 2003;14:44–6.
Safari VZ, Ngugi MP, Orinda G, Njagi EM. Anti-pyretic, Anti-inflammatory and analgesic activities of aqueous leaf extract of Urtica Dioica (L.) in Albino Mice. Med Aromat Plants. 2016;5:237.
Shailajan S, Hande H, Singh D, Tiwari B. Estimation of ursolic acid from Urtica dioica L. using validated HPTLC method. J Appl Pharm Sci. 2014;4:92–5.
Cummings AJ, Olsen M. Mechanism of action of stinging nettles. Wilderness Environ Med. 2011;22:136–9.
Safari VZ, Ngugi MP, Orinda G, Njagi EM. Anti-pyretic, Anti-inflammatory and analgesic activities of aqueous leaf extract of Urtica Dioica (L.) in Albino Mice. Med Aromat Plants. 2016;5:237.
Farahpour MR, Khoshgozaran L. Antinociceptive and anti-Inflammatory activities of hydroethanolic extract of Urtica dioica. Int J Biol Pharm. 2015;4:160–70.
Ayers S, Roschek B Jr, Williams JM, Alberte RS. Pharmacokinetic analysis of anti-allergy and anti-inflammation bioactives in a nettle (Urtica dioica) extract. Online J Pharmacol Pharmacokin. 2008;5:6–21.
Akbay P, Basaran AA, Undeger U, Basaran N. In vitro immunomodulatory activity of flavonoid glycosides from Urtica dioica L. Phytother Res. 2003;17:34–7.
Bnouham M, Merhfour FZ, Ziyyat A, Mekhfi H, Aziz M, Legssyer A. Antihyperglycemic activity of the aqueous extract of Urtica dioica. Fitoterapia. 2003;74:677–81.
Al-Tameme HJ, Hadi MY, Hameed IH. Phytochemical analysis of Urtica dioica leaves by fourier-transform infrared spectroscopy and gas chromatography-mass spectrometry. J Pharmacogn Phytother. 2015;7:238–52.
Said AAH, Otmani IS, Derfoufi S, Benmoussa A. Highlights on nutritional and therapeutic value of stinging nettle (Urtica dioica). Int J Pharm Pharm Sci. 2015;7:8–14.
Farhan SA, Faraj M, Al-Shemari HH, Jassim AKMN. Study of some Urtica dioica L. Leaves components and effect of their extracts on growth of pathogenic bacteria and identify of some flavonoids by HPLC. Al-Mustansiriya J. Sci. 2012;23:79–86.
Otles S, Yalcin B. Phenolic compounds analysis of root, stalk, and leaves of nettle. Sci World J. 2012;3:1–12.
Pinelli P, Ieri F, Vignolini P, Bacci L, Baronti S, Romani A. Extraction and HPLC Analysis of phenolic compounds in leaves, stalks, and textile fibers of Urtica dioica L. J Agric Food Chem. 2008;56:9127–32.
Brasil. Agencia Nacional de Vigilância Sanitária. Resolução-RDC no 26, de 13 de maio de 2014. Dispõe sobre o registro de medicamentos fitoterápicos e o registro e a notificação de produtos tradicionais fitoterápicos. Diário Oficial da União. Brasília, DF; 2014.
Correia LP, Santana CP, Medeiros ACD, Macêdo RO. Sideroxylon obtusifolium herbal medicine characterization using pyrolysis–GC/MS, SEM, and different thermoanalytical techniques. J Therm Anal Calorim. 2016;123:993–1001.
Wesołowski M, Konieczynski P. Thermoanalytical, chemical and principal component analysis of plant drugs. Int J Pharm. 2003;262:29–37.
Machado RD. Desenvolvimento tecnológico e caracterização de extratos vegetais obtidos a partir das raízes de Piper umbellatum L. (Piperaceae). Universidade Federal de Goiás, Goiânia. 2014.
List PH, Schimidt PC. Phytopharmaceutical technology. Boca Raton: CRC; 1989.
Silva RMF, Gomes TCBL, Albuquerque MM, Silva Junior JOC, Barbosa WLR, Rolim Neto PJ. Approach on different drying processes employed for obtaining dry extracts from medicinal plants. Rev Bras Plantas Med. 2012;14:103–9.
Oliveira PC, Conceição EC, Oliveira PA, Nascimento MV, Costa EA, De Paula JR, Bara MTF. Obtaining a dry extract of Pterodon emarginatus (Fabaceae) fruits by spray-drying. J Pharm Res. 2012;5:641–5.
Souza KCB, Petrovick PR, Bassani VL, Ortega GG. The adjuvants Aerosil 200 and Gelita-Sol-P influence on the technological characteristics of spray-dried powders from Passiflora edulis var. flavicarpa. Drug Dev Ind Pharm. 2000;26:331–6.
Oliveira MA. Análise Térmica Aplicada A Fármacos E Formulações Farmacêuticas Na Indústria Farmacêutica. Quim Nova. 2011;34:1224–30.
Corrigan OI. Thermal analysis of spray dried products. Thermochim Acta Irlanda. 1993;248:245–58.
Oliveira AH, Moura EA, Pinto MF, Procópio JVV, Souza VG, Souza FS, Macêdo RO. Thermal characterization of raw material pentoxifylline using thermoanalytical techniques and Pyr–GC/MS. J Therm Anal Calorim. 2011;106:763–6.
Procópio JVV, Souza VG, Costa RA, Correia LP, Souza FS, Macêdo RO. Application of thermal analysis and pyrolysis coupled to GC/MS in the qualification of simvastatin pharmaceutical raw material. J Therm Anal Calorim. 2011;106:665–70.
Correia LP, Procópio JVV, Santana CP, Santos AFO, Cavalcante HMM, Macêdo RO. Characterization of herbal medicine with different particle sizes using pyrolysis GC/MS, SEM and thermal techniques. J Therm Anal Calorim. 2013;111:1691–8.
Wang L, Wang C, Pan Z, Sun Y, Zhu X. Application of pyrolysis–gas chromatography and hierarchical cluster analysis to the discrimination of the Chinese traditional medicine Dendrobium candidum Wall. ex Lindl. J Anal Appl Pyrol. 2011;90:13–7.
Oliveira EJ, Alvarez EDA, Lima NGPB, Macêdo RO. Usefulness of pyrolysis coupled to gas chromatography/mass spectrometry for evaluating the reproducibility of commercial samples of Cymbopogon citratus Stapf., Poaceae. Braz J Pharmacogn. 2010;20:93–9.
Guimarães GP, Santos RL, Brandão DO, et al. Thermoanalytical characterization of herbal drugs from Poincianella pyramidalis in different particle sizes. J Therm Anal Calorim. 2017. https://doi.org/10.1007/s10973-016-6076-4.
Acknowledgements
The authors are grateful to Programa de Estudantes-Convênio de Pós-Graduação (PEC-PG) of the CAPES/CNPq, Brazil, for financial support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Cuinica, L.G., Macêdo, R.O. Thermoanalytical characterization of plant drug and extract of Urtica dioica L. and kinetic parameters analysis. J Therm Anal Calorim 133, 591–602 (2018). https://doi.org/10.1007/s10973-018-6986-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-018-6986-4