Abstract
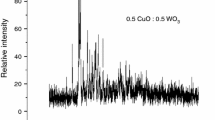
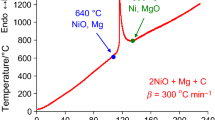
The mechanism and kinetics of tungsten and copper oxides joint reduction by Mg + C combined reducer was studied at high heating rates by thermal analysis method utilizing high-speed temperature scanner. The effective values of activation energy for magnesiothermic reduction stage for the binary (WO3–Mg, CuO–Mg), ternary (WO3–Mg–C, CuO–Mg–C, WO3–CuO–Mg) and quaternary (WO3–CuO–Mg–C) systems were determined in a new and wide range of heating rates (Vh = 100–5200 °C min−1). It was shown that for all the systems under study the increasing of heating rate shifts T* values toward to high-temperature area and unlike the low heating rates (Vh = 5–20 °C min−1, DTA/DTG studies) at high heating rates Mg always participates in molten state. In addition, by varying heating rates of reagents it was possible to separate the main stages and analyze intermediate compounds, making useful tool for the exploration of interaction mechanism in the complex systems. On the other hand, the tendency of merging of metals reduction stages at higher heating rates has an essential practical interest. That is, simultaneous reduction in metals is very prominent for obtaining metal composites with more homogeneous microstructure.







Similar content being viewed by others
References
Habashi F. Alloys: preparation, properties, applications. New York: Wiley; 2008.
Selvakumar N, Vettivel SC. Thermal, electrical and wear behavior of sintered Cu–W nanocomposite. Mater Des. 2013;46:16–25.
Minakova RV, Lesnik ND, Kresanova AP, Flis AA, Khomenko EV. Contact interaction, structure, and properties of W (Mo, Cr)–Cu composites with additives. Powder Metall Met Cer. 1996;35(7):363–71.
Bukhanovs’kyi VV, Rudnyts’kyi MP, Kharchenko VV, Minakova RV, Grechanyuk MI, Mamuzic I. Relationship between composition, structure, and mechanical properties of a condensed composite of copper–tungsten system. Strength Mater. 2011;43(4):426–37.
Bukhanovskii VV, Minakova RV, Grechanyuk IN, Mamuziæ I, Rudnitskii NP. Effect of composition and heat treatment on the structure and properties of condensed composites of the Cu–W system. Met Sci Heat Treat. 2011;53(1-2):14–23.
Kaczmar JW, Pietrzak K, Włosiński W. The production and application of metal matrix composite materials. J Mater Process Technol. 2000;106(1):58–67.
Dorfman LP, Scheithauer MJ, Paliwal M, Houck DL, Spencer JR. Alloy for electrical contacts and electrodes and method of making. U.S. Patent No. 6,375,708; 2002.
Rani AM, Mahamat AZ, Ab Adzis AH. Novel nano copper-tungsten-based EDM electrode. In: Viswanatha Sharma K, Hisham B Hamid N, editors. Engineering applications of nanotechnology. Berlin: Springer; 2017. p. 193–224.
He L, Yu J, Duan W, Liu Z, Yin S, Luo H. Copper–tungsten electrode wear process and carbon layer characterization in electrical discharge machining. Int J Adv Manuf Technol. 2016;85(5-8):1759–68.
Guo TB, Zhao JY, Ding YT. Microstructure and properties of tungsten copper composite material. In: Materials science forum, vol 788. Trans Tech Publications; 2014, pp 646–51.
Chen CF, Pokharel R, Brand MJ, Tegtmeier EL, Clausen B, Dombrowski DE, Ickes TL, Lebensohn RA. Processing and consolidation of copper/tungsten. J Mater Sci. 2017;52(2):1172–82.
Daoush WM, Yao J, Shamma M, Morsi K. Ultra-rapid processing of high-hardness tungsten–copper nanocomposites. Scr Mater. 2016;113:246–9.
Xu L, Srinivasakannan C, Zhang L, Yan M, Peng J, Xia H, Guo S. Fabrication of tungsten–copper alloys by microwave hot pressing sintering. J Alloys Compd. 2016;658:23–8.
Lin D, Han JS, Kwon YS, Ha S, Bollina R, Park SJ. High-temperature compression behavior of W–10 wt% Cu composite. Int J Refract Met Hard Mater. 2015;53:87–91.
Duan L, Lin W, Wang J, Yang G. Thermal properties of W–Cu composites manufactured by copper infiltration into tungsten fiber matrix. Int J Refract Met Hard Mater. 2014;46:96–100.
Xi X, Xu X, Nie Z, He S, Wang W, Yi J, Tieyong Z. Preparation of W–Cu nano-composite powder using a freeze-drying technique. Int J Refract Met Hard Mater. 2010;28(2):301–4.
Basu AK, Sale FR. Copper-tungsten composite powders by the hydrogen reduction of copper tungstate. J Mater Sci. 1978;13(12):2703–11.
Raghu T, Sundaresan R, Ramakrishnan P, Mohan TR. Synthesis of nanocrystalline copper–tungsten alloys by mechanical alloying. Mater Sci Eng A. 2001;304:438–41.
Jech DE, Juan LS, Anthony BT. Process for making improved copper/tungsten composites. U.S. Patent No. 5,686,676; 1997.
Kirakosyan HV, Aydinyan SV, Kharatyan SL. W–Cu composite powders obtained by joint reduction of oxides in combustion mode. Int J SHS. 2016;25(4):215–23.
Aydinyan SV, Kirakosyan HV, Zakaryan MK, Kharatyan SL. Combustion synthesis of W–Cu composite powders from oxide precursors with various proportions of metals. Int J Refract Met Hard Mater. 2017;64:176–83.
Aydinyan SV, Kirakosyan HV, Kharatyan SL. Cu–Mo composite powders obtained by combustion-coreduction process. Int J Refract Met Hard Mater. 2016;54:455–63.
Nepapushev AA, Kirakosyan KG, Moskovskikh DO, Kharatyan SL, Rogachev AS, Mukasyan AS. Influence of high-energy ball milling on reaction kinetics in the Ni–Al system: an electrothermorgaphic study. Int J SHS. 2015;24(1):21–8.
Hobosyan MA, Kirakosyan KG, Kharatyan SL, Martirosyan KS. PTFE–Al2O3 reactive interaction at high heating rates. J Therm Anal Calorim. 2015;119(1):245–51.
Hobosyan MA, Kirakosyan KG, Kharatyan SL, Martirosyan KS. Study of dynamic features of highly energetic reactions by DSC and high-speed temperature scanner (HSTS). In: MRS proceedings. Cambridge University Press. 2013; p 1521.
Baghdasaryan AM, Niazyan OM, Khachatryan HL, Kharatyan SL. DTA/TG study of tungsten oxide and ammonium tungstate reduction by (Mg + C) combined reducers at non-isothermal conditions. Int J Refract Met Hard Mater. 2014;43:216–21.
Baghdasaryan AM, Niazyan OM, Khachatryan HL, Kharatyan SL. DTA/TG study of molybdenum oxide reduction by Mg/Zn & Mg/C combined reducers at non-isothermal conditions. Int J Refract Met Hard Mater. 2015;51:315–23.
Kirakosyan HV, Minasyan TT, Niazyan OM, Aydinyan SV, Kharatyan SL. DTA/TGA study of CuO and MoO3 co-reduction by combined Mg/C reducers. J Therm Anal Calorim. 2016;123:35–41.
Niazyan OM, Aydinyan SV, Kharatyan SL. DTA/TG study of reduction mechanism of WO3 + CuO mixture by combined Mg/C reducer. Chem J Armenia. 2016;69(4):399–406.
Aydinyan S, Kirakosyan H, Niazyan O, Tumanyan M, Nazaretyan Kh, Kharatyan S. Reaction pathway in the WO3–CuO–Mg–C system at nonisothermal conditions. Armen J Phys. 2016;9(1):83–8.
Starink MJ. The determination of activation energy from linear heating rate experiments: a comparison of the accuracy of isoconversion methods. Thermochim Acta. 2003;404(1-2):163–76.
Wang LL, Munir ZA, Maximov YM. Review, Thermite reactions: their utilization synthesis and processing of materials. J Mater Sci. 1993;28:3693–708 (Primary source: G.D. MILLER, Thermochim. Acta 34 (1979) 357).
Hosseini SG, Sheikhpour A, Keshavarz MH, Tavangar S. The effect of metal oxide particle size on the thermal behavior and ignition kinetic of Mg–CuO thermite mixture. Thermochim Acta. 2016. https://doi.org/10.1016/j.tca.2016.01.005.
Schoenitz M, Umbrajkar S, Dreizin EL. Kinetic analysis of thermite reactions in Al–MoO3 nanocomposites. J Propuls Power. 2007. https://doi.org/10.2514/1.24853.
Wang Y, et al. Energy release characteristics of impact-initiated energetic aluminum–magnesium mechanical alloy particles with nanometer-scale structure. Thermochim Acta. 2011;512(1):233–9.
Sheikhpour A, Hosseini SG, Tavangar S, Keshavarz MH. The influence of magnesium powder on the thermal behavior of Al–CuO thermite mixture. J Therm Anal Calorim. 2017;129(3):1847–54. https://doi.org/10.1007/s10973-017-6343-z.
Acknowledgements
This work was financially supported by the International Science and Technology Center (Project No. A-2123).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aydinyan, S.V., Nazaretyan, K.T., Zargaryan, A.G. et al. Reduction mechanism of WO3 + CuO mixture by combined Mg/C reducer. J Therm Anal Calorim 133, 261–269 (2018). https://doi.org/10.1007/s10973-018-6985-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-018-6985-5