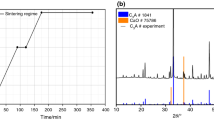
Abstract
The present study reports the results of investigation on the role of metakaolin in the formation of ettringite in a model relevant to Portland cement. The model consists of ternary system (Trio) metakaolin–lime–gypsum. Five samples of defined ternary system were cured at different temperatures 20, 30, 40, 50 and 60 °C. Conduction calorimeter TAM AIR was mainly used to capture heat evolution at different temperatures. Thermoanalytical (simultaneous TGA/DSC) and X-ray diffraction methods were used to identify different products after curing. It results that ettringite is the main hydration product supplemented by calcium silicate and calcium aluminosilicate hydrates according to sample composition. The mechanism and kinetics of hydration, as displayed by calorimetric curves, depend on composition of samples and curing temperatures. Two main types of processes have been elucidated: reaction of aluminum ions with sulfate ones in the presence of calcium ions in aqueous solution to form ettringite supplemented by pozzolanic activity leading to the formation of calcium silicate and calcium aluminosilicate hydrates. Concomitant condensation of alumina and silica species and carbonation have influenced the course of hydration. Activation energy E a depends slightly on composition of ternary system.











Similar content being viewed by others
References
Li C, Sun H, Li L. A review: the comparison between alkali-activated slag (Si+ Ca) and metakaolin (Si+ Al) cements. Cem Concr Res. 2010;40:1341–9.
Sha W, Pereira GB. Differential scanning calorimetry study of ordinary Portland cement paste containing metakaolin and theoretical approach of metakaolin activity. Cem Concr Compos. 2001;25:455–61.
Kuliffayová M, Krajči Ľ, Janotka I, Šmatko V. Thermal behaviour and characterization of cement composites with burnt kaolin sand. J Therm Anal Calorim. 2012;108:425–32.
Krajčí Ľ, Janotka I, Jamnický P. Burnt kaolin sand as pozzolanic material for cement hydration. Ceram Silik. 2007;51:217–24.
Snelson DG, Wild S, O’Farrell M. Heat of hydration of Portland cement–metakaolin–fly ash (PC–MK–PFA) blends. Cem Concr Res. 2008;38:832–40.
Ptáček P, Opravil T, Šoukal F, Wasserbauer J, Masilko J, Baracek J. The influence of structure order on the kinetics of dehydroxylation of kaolinite. J Eur Ceram Soc. 2013;33:2793–9. https://doi.org/10.1016/j.jeurceramsoc.2013.04.033.
Ptáček P, Kubatová D, Havlica J, Brandštetr J, Šoukal F, Opravil T. The non-isothermal kinetic analysis of the thermal decomposition of kaolinite by thermogravimetric analysis. Powder Technol. 2010;204:222–7.
Li Z, Ding Z. Property improvement of Portland cement by incorporating with metakaolin and slag. Cem Concr Res. 2003;33:579–84.
Kostuch JA, Walters GV, Jones TR. High performance concretes incorporating metakaolin—a review. In: Dhir RK, Jones MR, editors. Concrete 2000. London: E&FN Spon; 1993. p. 1799–811.
Nadeem A, Memon SA, Lo TY. Mechanical performance, durability, qualitative and quantitative analysis of microstructure of fly ash and metakaolin mortar at elevated temperatures. Constr Build Mater. 2013;38:338–47.
Minárik L, Kopecskó K. Impact of metakaolin—a new supplementary material—on the hydration mechanism of cement, Acta Tech. Napocensis Civ Eng Arch. 2013;56(2):100–110. https://constructii.utcluj.ro/ActaCivilEng/download/atn/ATN2013(2)_9.pdf.
Morsy MS, Al-Salloum Y, Almusallam T, Abbas H. Effect of nano-metakaolin addition on the hydration characteristics of fly ash blended cement mortar. J Therm Anal Calorim. 2014;116:845–52.
Gallucci E, Mathur P, Scrivener KL. Microstructural development of early age hydration shells around cement grains. Cem Concr Res. 2010;40:4–13.
Jansen D, Goety-Neunhoeffer F, Lothenbach B, Neubauer J. The early hydration of Ordinary Portland Cement (OPC): An approach comparing measured heat flow with calculated heat flow from QXRD. Cem Concr Res. 2012;42:134–8.
Copeland LE, Kantro DL, Verbeck GJ. Chemistry of hydration of Portland cement. Proc Symp Cem Chemistry Cem Concr Assoc Wash. 1960;1:429–68.
El-Didamony H, Tm El-Sokkari, Kha Khalil, Mohamed H, Ahmed IA. Hydration mechanisms of calcium sulphoaluminate C4A3S̄, C4AS̄ phase and active belite β-C2S. Ceram Silik. 2012;56:389–95.
Juenger MCG, Winnefeld F, Provis JL, Ideker JH. Advances in alternative cementitious binders. Cem Concr Res. 2011;41:1232–43.
Palou MT, Majling J. Effects of sulfate, calcium and aluminium ions upon the hydration of sulphoaluminate belite cement. J Therm Anal. 1996;46:549–56.
Pelletier L, Winnefeld F, Lothenbach B. The ternary system Portland cement–calcium sulphoaluminate clinker–anhydrite: hydration mechanism and mortar properties. Cem Concr Comp. 2010;32:497–507.
Winnefeld F, Lothenbach B. Hydration of calcium sulfoaluminate cements—experimental findings and thermodynamic modelling. Cem Concr Res. 2010;40:1239–47.
Palou MT, Kuzielová E, Novotny RF, Žemlička M. Blended cements consisting of Portland cement–slag–silica fume–metakaolin system. J Therm Anal Calorim. 2016;125:998–1004.
Boháč M, Palou MT, Novotný R, Másilko J, Všianský D, Staněk T. Investigation on early hydration of Portland cement-blast-furnace slag-metakaolin ternary blends. Constr Build Mater. 2014;64:333–41.
Boháč M, Palou M, Novotný R, Másilko JF, Opravil T. Influence of temperature on early hydration of Portland cement-metakaolin-slag system. J Therm Anal Calorim. 2017;127:309–18.
Žemlička M, Kuzielová E, Kuliffayová M, Tkacz J, Palou MT. Study of hydration products in the model systems metakaolin–lime and metakaolin–lime–gypsum. Ceram Silik. 2015;59:283–91.
Frías M, Sánchez de Rojas MI, Cabrera J. The effect that the pozzolanic reaction of metakaolin has on the heat evolution in metakaolin-cement mortars. Cem Concr Res. 2000;30:209–16.
Rahhal V, Cabrera O, Talero R, Delgado A. Calorimetry of Portland cement with silica fume and gypsum additions. J Therm Anal Calorim. 2007;87:331–6.
Gruyaert E, Robeyst N, De Belie N. Study of the hydration of Portland cement blended with blast-furnace slag by calorimetry and thermogravimetry. J Therm Anal Calorim. 2010;102:941–51.
Morsy MS. Effect of temperature on hydration kinetics and stability of hydration phases of metakaolin–lime sludge–silica fume system. Ceramics. 2005;49:225–9.
Siler P, Kratky J, De Belie N. Isothermal and solution calorimetry to assess the effect of superplasticizers and mineral admixtures on cement hydration. J Therm Anal Calorim. 2012;107:313–20.
Pacewska B, Wilinska I, Bukowska M. Calorimetric investigations of the influence of waste aluminosilicate on the hydration of different cements. J Therm Anal Calorim. 2009;97:61–6.
Zielenkiewicz W, Kaminski M. A conduction calorimeter for measuring the heat of cement hydration in the initial hydration period. J Therm Anal Calorim. 2001;65:335–40.
Copeland LE, Kantro DL, Verbeck GJ. Chemistry of hydration of Portland cement. Proc Symp Cem Chem Cem Concr Assoc Wash. 1960;1:429–68.
Thomas JJ. The instantaneous apparent activation energy of cement hydration measured using a novel calorimetry-based method. J Am Ceram Soc. 2012;95–10:3291–6.
Ježo Ľ, Ifka T, Cvopa B, Škundová J, Kovár V, Palou M. Effect of temperature upon the strength development rate and upon the hydravion kinetice of cements. Ceram Silik. 2010;54:269–76.
Feldman RF, Ramachandran VS, Sereda PJ. Influence of CaCO3 on the hydration of 3CaO. Al2O3. J Am Ceram Soc. 1965;48:25–30.
Bensted J. Some hydration investigations involving Portland cement effect of calcium carbonate substitution of gypsum. World Cem Technol. 1980;11:395–406.
Weng L, Sagoe-Crentsil K. Dissolution processes, hydrolysis and condensation reactions during geopolymer synthesis: part I—Low Si/Al ratio systems. J Mater Sci. 2007;42:2997–3006.
Weng L, Sagoe-Crentsil K, Brown T, Song S. Mater Sci Eng B Solid-State Mater Adv Technol. 2005;117(2):163.
Acknowledgements
The authors are grateful for Grant APVV-15-0631 and for the support of the project Sustainability and Development REG LO1211 addressed to the Materials Research Center at FCH VUT.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Palou, M., Kuzielová, E., Žemlička, M. et al. The effect of metakaolin upon the formation of ettringite in metakaolin–lime–gypsum ternary systems. J Therm Anal Calorim 133, 77–86 (2018). https://doi.org/10.1007/s10973-017-6885-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-017-6885-0