Abstract
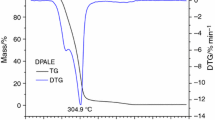
To rich flavor additive species of pyrazines, two new compounds of 3,6-dimethyl-2,5-pyrazinedicarboxylic acid 1-octen-3-yl ester (DMPOE) and 3,5,6-trimethyl-2-pyrazinecarboxylic acid 1-octen-3-yl ester (TMPOE) were synthesized by KMnO4 oxidation, acylating chlorination and esterification reaction, in which tetramethylpyrazine and 1-octen-3-ol were used as initial materials. Thermogravimetry (TG), differential scanning calorimeter (DSC) and pyrolysis–gas chromatography/mass spectrometry (Py–GC/MS) were conducted to investigate the thermal degradation behaviors of DMPOE and TMPOE. TG–DTG results indicated that the T p of DMPOE and TMPOE with the largest mass loss rate was at 310 and 250 °C, respectively. The T peak of DMPOE and TMPOE showed by DSC curves was 301.8 and 260.0 °C, respectively. Py–GC/MS was performed to benefit the simulation of cigarette burning conditions, and the results indicated that DMPOE and TMPOE could release specific flavors of 1-octen-3-ol and diversified alkylpyrazines. Furthermore, the thermal degradation mechanisms of the flavor precursors of DMPOE and TMPOE were discussed. The study on the thermal behavior of these two methylpyrazinecarboxylates would provide theoretical basis for their application in tobacco.







Similar content being viewed by others
References
Pagoria PF, Lee GS, Mitchell AR, Schmidt RD. A review of energetic materials synthesis. Thermochim Acta. 2002;384:187–204.
Hawksworth G, Scheline RR. Metabolism in the rat of some pyrazine derivatives having flavour importance in foods. Xenobiotica. 1975;5:389–99.
Ramli N, Hassan O, Said M, Samsudin W, Idris NA. Influence of roasting conditions on volatile flavor of roasted Malaysian cocoa beans. J Food Process Preserv. 2006;30:280–98.
Ruan ED, Aalhus JL, Juárez M, Sabik H. Analysis of volatile and flavor compounds in grilled lean beef by stir bar sorptive extraction and thermal desorption–gas chromatography mass spectrometry. Food Anal Methods. 2015;8:363–70.
Lee SE, Chung H, Kim YS. Effects of enzymatic modification of wheat protein on the formation of pyrazines and other volatile components in the Maillard reaction. Food Chem. 2012;131:1248–54.
Fan W, Xu Y, Zhang Y. Characterization of pyrazines in some Chinese liquors and their approximate concentrations. J Agric Food Chem. 2007;55:9956–62.
Lancker FV, Adams A, Kimpe ND. Impact of the N-terminal amino acid on the formation of pyrazines from peptides in maillard model systems. J Agric Food Chem. 2012;60:4697–708.
Yu AN, Tan ZW, Wang FS. Mechanistic studies on the formation of pyrazines by Maillard reaction between l-ascorbic acid and l-glutamic acid. LWT-Food Sci Technol. 2013;50:64–71.
Nicolau LP, Revel GD, Bertrand A, Maujean A. Formation of flavor components by the reaction of amino acid and carbonyl compounds in mild conditions. J Agric Food Chem. 2000;48:3761–6.
Hwang HI, Hartman TG, Rosen RT, Chi TH. Formation of pyrazines from the Maillard reaction of glucose and glutamine-amide-15N. J Agric Food Chem. 1993;41:2112–5.
Adams A, Polizzi V, Boekel MV, Kimpe ND. Formation of pyrazines and a novel pyrrole in Maillard model systems of 1,3-dihydroxyacetone and 2-oxopropanal. J Agric Food Chem. 2008;56:2147–53.
Maga JA, Sizer CE, Myhre DV. Pyrazines in foods. Crit Rev Food Sci. 1973;4:39–115.
Niebler J, Buettner A. Pyrolysis–GC–MS–olfactometry: a new approach to identify thermally generated odorants in frankincense. J Anal Appl Pyrolysis. 2015;113:690–700.
Takken W, Kline DL. Carbon dioxide and 1-octen-3-ol as mosquito attractants. J Am Mosq Control. 1989;5:311–6.
Leduc F, Tournayre P, Kondjoyan N, Mercier F, Malle P, Kol O, Berdagué JL, Duflos G. Evolution of volatile odorous compounds during the storage of European seabass (Dicentrarchus labrax). Food Chem. 2012;131:1304–11.
Song S, Zhang X, Hayat K, Liu P, Jia C, Xia S, Xiao Z, Tian H, Niu Y. Formation of the beef flavour precursors and their correlation with chemical parameters during the controlled thermal oxidation of tallow. Food Chem. 2011;124:203–9.
Baker RR. A review of pyrolysis studies to unravel reaction steps in burning tobacco. J Anal Appl Pyrolysis. 1987;11:555–73.
Xie WC, Gu XH, Tan ZC, Tang J, Wang GY, Luo CR, Sun LX. Thermal decomposition of two synthetic glycosides by TG, DSC and simultaneous Py–GC–MS analysis. J Therm Anal Calorim. 2006;87:505–10.
Xie W, Tang J, Gu X, Luo C, Wang G. Thermal decomposition study of menthyl-glycoside by TGA/SDTA, DSC and simultaneous Py–GC–MS analysis. J Anal Appl Pyrolysis. 2007;78:180–4.
Zeng S, Sun S, Liu S, Hu J, He B. Synthesis of α-ionyl-β-d-glucoside and its property of flavor release. J Therm Anal Calorim. 2014;115:1049–56.
Zhu P, Sui S, Wang B, Sun K, Sun G. A study of pyrolysis and pyrolysis products of flame-retardant cotton fabrics by DSC, TGA, and PY–GC–MS. J Anal Appl Pyrolysis. 2004;71:645–55.
Werner K, Pommer L, Broström M. Thermal decomposition of hemicelluloses. J Anal Appl Pyrolysis. 2014;110:130–7.
Bikiaris DN, Chrissafis K, Paraskevopoulos KM, Triantafyllidis KS, Antonakou EV. Investigation of thermal degradation mechanism of an aliphatic polyester using pyrolysis–gas chromatography–mass spectrometry and a kinetic study of the effect of the amount of polymerisation catalyst. Polym Degrad Stab. 2007;92:525–36.
Price D, Horrocks AR, Akalin M, Faroq AA. Influence of flame retardants on the mechanism of pyrolysis of cotton (cellulose) fabrics in air. J Anal Appl Pyrolysis. 1997;40:511–24.
Chen T, Oakley DM. Thermal analysis of proteins of pharmaceutical interest. Thermochim Acta. 1995;248:229–44.
Stotesbury S, Digard H, Willoughby L, Couch A. The pyrolysis of tobacco additives as a means of predicting their behaviour in a burning cigarette. Beitr Tab Int. 1999;18:147–63.
Baker RR, Bishop LJ. The pyrolysis of non-volatile tobacco ingredients using a system that simulates cigarette combustion conditions. J Anal Appl Pyrolysis. 2005;74:145–70.
Baker RR, Bishop LJ. The pyrolysis of tobacco ingredients. J Anal Appl Pyrolysis. 2004;71:223–311.
Lai M, Zhao B, Bao X, Zhao M, Ji X, Fu P, Zhang Y. Pyrolysates of novel latent fragrant compound 3,6-dimethyl-2,5-pyrazinedicarboxylic acid menthol ester. Chin J Chromatogr. 2015;33:46–51.
Chen H, Li G, Zhan P, Li H, Wang S, Liu X. Design, synthesis and biological evaluation of novel trimethylpyrazine-2-carbonyloxy-cinnamic acids as potent cardiovascular agents. MedChemComm. 2014;5:711–8.
Adams TB, Doull J, Feron VJ, Goodman JI, Marnet LJ, Munro IC, Newberne PM, Portoghese PS, Smith RL, Waddell WJ, Wagner BM. The FEMA GRAS assessment of pyrazine derivatives used as flavor ingredients. Food Chem Toxicol. 2002;40:429–51.
Holmelid B, Kleinert M, Barth T. Reactivity and reaction pathways in thermochemical treatment of selected lignin-like model compounds under hydrogen rich conditions. J Anal Appl Pyrolysis. 2012;98:37–44.
Lai M, Zhao B, Ji X, Fu P, Wang P, Bao X, Zhao M. Thermal behavior of two synthesized flavor precursors of pyrazine esters. J Therm Anal Calorim. 2016;123:479–87.
Paine JB, Pithawalla YB, Naworal JD. Carbohydrate pyrolysis mechanisms from isotopic labeling: part 4. The pyrolysis of d-glucose: the formation of furans. J Anal Appl Pyrolysis. 2008;83:37–63.
Wang S, Liu B, Su Q. Pyrolysis–gas chromatography/mass spectrometry as a useful technique to evaluate the pyrolysis pathways of phenylalanine. J Anal Appl Pyrolysis. 2004;71:393–403.
Acknowledgements
This work was supported by Key Science and Technology Program of Science and Technology Department of Henan Province (Nos. 132102210042, 152102210058).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lai, M., Ji, X., Tao, T. et al. Synthesis and pyrolysis of two flavor precursors of oct-1-en-3-yl methylpyrazinecarboxylates. J Therm Anal Calorim 128, 1627–1638 (2017). https://doi.org/10.1007/s10973-016-6083-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-016-6083-5