Abstract
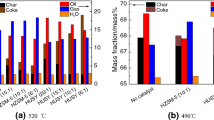
The thermal mass conversion of crude oil from the Carmópolis field (19.53 oAPI) was studied by thermal analysis and with an off-line micropyrolysis system. The products of the micropyrolysis experiments were characterized by gas chromatography/mass spectrometry (GC/MS). The effects of the zeolite catalysts ferrierite and type Y on the TG curves for the crude oil were investigated applying different percentages of catalyst [10, 30 and 50 % (w/w)]. The influence of the heating rate (5, 10 and 20 °C min−1) on the decomposition profile was also evaluated. The thermal analysis showed that, in general, lower heating rates lead to a better mass conversion. Based on the TG curves obtained at 400 °C, the presence of the catalysts resulted in a reduction in the initial pyrolysis temperature and a gain of 10 % in the mass conversion. A detailed analysis of the hydrocarbons showed that the catalysts are not selective, acting similarly on the decomposition over the entire range of hydrocarbons. Also, the n-alkanes fingerprint shows a bimodal distribution, indicating that this onshore oil is associated with terrigenous higher-plant waxes. From the analysis of the isoprenoids, phytane was present in a higher percentage than pristane which, according to the literature, indicates that the oil was generated in an anoxic environment.










Similar content being viewed by others
References
Owenn NA, Inderwildi OR, King DA. The status of conventional world oil reserves—hype or cause for concern? Energy Policy. 2010;38:4743–9.
Kök MV. Clay concentration and heating rate effect on crude oil combustion by thermogravimetry. Fuel Process Technol. 2012;96:134–9.
Kök MV, Gul KG. Thermal characteristics and kinetics of crude oils and SARA fractions. Thermochim Acta. 2013;569:66–70.
Li Y, Zhao J, Pu W, Jia H, Peng H, Zhong D, Wang S. Catalytic effect analysis of metallic additives on light crude oil by TG and DSC tests. J Therm Anal Calorim. 2013;113:579–87.
Mothé MG, Mothé CG, Carvalho CHM, Oliveira MCK. Thermal evaluation of heavy crude oil by simultaneous TG-DSC-FTIR: part 2. J Therm Anal Calorim. 2014;117:1357–63.
Brown M, Gallagher P. Handbook of thermal analysis and calorimetry: applications to inorganic and miscellaneous materials, vol. 2. Amsterdam: Elsevier; 2003.
Medeiros RI, Leles MIG, Dias FC, Kunert R, Antonio Filho NR. Thermogravimetric isolation and chromatographic characterization of volatile fractions of petroleum. J Therm Anal Calorim. 2008;91:225–9.
Mothé MG, Mothé CG, Carvalho CHM, Oliveira MCK. Thermal investigation of heavy crude oil by simultaneous TG-DSC-FTIR and EDXRF: part 1. J Therm Anal Calorim. 2013;113:525–31.
Gonçalves MLA, Barreto JRC, Cerqueira WV, Teixeira AMRF. Effect of zeolite, kaolin and alumina during cracking of heavy petroleum residue evaluated by thermogravimetry. J Therm Anal Calorim. 2009;97:515–9.
Komatsu T. Catalytic cracking of paraffins on zeolite catalysts for the production of light olefins. 20th Annual Saudi-Japan Symposium, catalysts in petroleum refining & petrochemicals, Dhahran, Saudi Arabia; 2010
Abrevaya H. Cracking of naphtha range alkanes and naphthenes over zeolites. Stud Surf Sci Catal. 2007;170:1244–51.
Bastiani R, Lam YL, Henriques CA, Silva VT. Application of ferrierite zeolite in high-olefin catalytic cracking. Fuel. 2013;107:680–7.
Meng X, Xu C, Gao J, Li L. Catalytic pyrolysis of heavy oils: 8-lump kinetic model. Appl Catal A Gen. 2006;301:32–8.
Meng X, Xu C, Gao J, Li L. Studies on catalytic pyrolysis of heavy oils: reaction behaviors and mechanistic pathways. Appl Catal A Gen. 2005;294:168–76.
Fabbri D, Vassura I. Evaluating emission levels of polycyclic aromatic hydrocarbons from organic materials by analytical pyrolysis. J Anal Appl Pyrolysis. 2006;75:150–8.
Love GD, Snape CE, Carr AD, Houghton RC. Release of covalently-bound alkane biomarkers in high yields from kerogen via catalytic hydropyrolysis. Org Geochem. 1995;23(10):981–6.
Meredith W, Sun C, Snape CE, Sephton MA, Love GD. The use of model compounds to investigate the release of covalently bound biomarkers via hydropyrolysis. Org Geochem. 2006;37:1705–14.
Meredith W, Russell CA, Cooper M, Snape CE, Love GD, Fabbri D, Vane CH. Trapping hydropyrolysates on silica and their subsequent thermal desorption to facilitate rapid fingerprinting by GC–MS. Org Geochem. 2004;35:73–89.
Lima DI, Lima SF, Silva VV, Sant’ana AEG, Rebouças LMC. Biomarcadores saturados em óleos da Bacia Sergipe-Alagoas Brasil: Distribuição e Concentração. Geochim Bras. 2007;21:99–110.
Fogler HS. Elements of chemical reaction engineering. 4th ed. Rio de Janeiro: LTC; 2009. p. 522–3.
Peters KE, Walters CC, Moldowan JM. The biomarker guide. Cambridge: Cambridge University Press; 2005.
Peters KE, Moldowan JM. The biomarker guide: a interpreting molecular fossil in petroleum and ancient sediments. New Jersey: Prentice-Hall Inc.; 1993.
Acknowledgements
The authors are grateful to PETROBRAS/UO-SEAL/ENGP/LABF for the crude oil sample, to National Council for Scientific and Technological Development—CNPq for fellowships, to LABCAT—Laboratory of Catalysis/UFS for the catalysts, to the Postgraduate Program in Chemistry/PPGQ-UFS for the use of their analytical infrastructure and sincerely thank the two anonymous reviewers.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maciel, S.T.A., Wisniewski, A. & de Souza, M.J.B. Use of micropyrolysis and TG to study the thermal catalytic conversion of onshore crude oil using the zeolite catalysts type Y and ferrierite. J Therm Anal Calorim 122, 369–377 (2015). https://doi.org/10.1007/s10973-015-4679-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-015-4679-9