Abstract
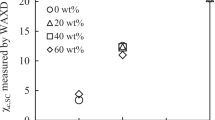
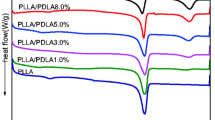
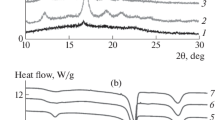
Stereocomplex-poly(l- and d-lactide) (sc-PLA) and poly(methyl methacrylate) (PMMA) blends were prepared by solution blending at PMMA loadings from 20 to 80 mass%. The miscibility and crystallization behaviors of the blends have been studied in detail by differential scanning calorimeter. The single-glass transition temperatures (T g) of the blends demonstrated that the obtained system was miscible in the amorphous state. It was observed that the crystallization peak temperature of sc-PLA/PMMA blends was marginally lower than that of neat sc-PLA at various cooling rates, indicating the dilution effect of PMMA on the sc-PLA component to restrain the overall crystallization process. In the study of isothermal crystallization kinetics, the reciprocal value of crystallization peak time (\( t_{\text{p}}^{ - 1} \)) decreased with increasing PMMA content, indicating that the addition of non-crystalline PMMA inhibited the isothermal crystallization of sc-PLA at an identical crystallization temperature (T c). Moreover, the negative value of Flory–Huggins interaction parameter (χ 12 = −0.16) of the blend further indicated that sc-PLA and PMMA formed miscible blends.








Similar content being viewed by others
References
Drumright RE, Gruber PR, Henton DE. Polylactic acid technology. Adv Mater. 2000;12:1841–6.
Zuk PA, Zhu M, Ashjian P, Ugarte DAD, Huang JI, Mizuno H, Alfonso ZC, Fraser JK, Benhaim P, Hedrick MH. Human adipose tissue is a source of multipotential stem cells. Mol Biol Cell. 2002;13:4279–95.
Gottschalk C, Frey H. Hyperbranched polylactide copolymers. Macromolecules. 2006;39:1719–23.
Anderson JM, Shive MS. Biodegradation and biocompatibility of PLA and PLGA microspheres. Adv Drug Deliv Rev. 1997;28:5–24.
Auras R, Harte B, Selke S. An overview of polylactides as packaging materials. Macromol Biosci. 2004;4:835–64.
Hollinger JO. Preliminary report on the osteogenic potential of a biodegradable copolymer of polylactide (PLA) and polyglycolide (PGA). J Biomed Mater Res. 1983;17:71–82.
Samuel C, Cayuela J, Barakat I, Müller AJ, Raquez JM, Dubois P. Stereocomplexation of polylactide enhanced by poly(methyl methacrylate): improved processability and thermomechanical properties of stereocomplexable polylactide-based materials. ACS Appl Mater Interfaces. 2013;5:11797–807.
Saeidlou S, Huneault MA, Li H, Park CB. Poly(lactic acid) crystallization. Prog Polym Sci. 2012;37:1657–77.
Blanco I, Siracusa V. Kinetic study of the thermal and thermo-oxidative degradations of polylactide-modified films for food packaging. J Therm Anal Calorim. 2013;112:1171–7.
Murariu M, Dechief AL, Bonnaud L, Paint Y, Gallos A, Fontaine G, Bourbigot S, Dubois P. The production and properties of polylactide composites filled with expanded graphite. Polym Degrad Stab. 2010;95:889–900.
Kim KW, Lee BH, Kim HJ, Sriroth K, Dorgan JR. Thermal and mechanical properties of cassava and pineapple flours-filled PLA bio-composites. J Therm Anal Calorim. 2012;108:1131–9.
Blanco I. End-life prediction of commercial PLA used for food packaging through short term TGA experiments: real chance or low reliability? Chin J Polym Sci. 2014;32:681–9.
Dong QL, Li Y, Han CY, Zhang Z, Xu K, Zhang HL, Dong LS. Poly(l-lactide)/poly(d-lactide)/multiwalled carbon nanotubes nanocomposites: enhanced dispersion, crystallization, mechanical properties, and hydrolytic degradation. J Appl Polym Sci. 2013;130:3919–29.
Ikada Y, Jamshidi K, Tsuji H, Hyon S. Stereocomplex formation between enantiomeric poly(lactides). Macromolecules. 1987;20:906–8.
Tsuji H. Poly(lactide) stereocomplexes: formation, structure, properties, degradation, and applications. Macromol Biosci. 2005;5:569–97.
Gallos A, Fontaine G, Bourbigot S. Reactive extrusion of intumescent stereocomplexed poly-l, d-lactide: characterization and reaction to fire. Polym Adv Technol. 2013;24:130–3.
Tan BH, Hussain H, Lin TT, Chua CY, Leong YW, Tjiu WW, Wong PK, He CB. Stable dispersions of hybrid nanoparticles induced by stereocomplexation between enantiomeric poly(lactide) star polymers. Langmuir. 2011;27:10538–47.
Ahmed J, Varshney SK, Janvier F. Rheological and thermal properties of stereocomplexed polylactide films. J Therm Anal Calorim. 2014;115:2035–61.
Maglio G, Malinconico M, Migliozzi A, Groeninckx G. Immiscible poly(l-lactide)/poly(ε-caprolactone) blends: influence of the addition of a poly(l-lactide)–poly(oxyethylene) block copolymer on thermal behavior and morphology. Macromol Chem Phys. 2004;205:947–50.
Takayama T, Todo M. Improvement of impact fracture properties of PLA/PCL polymer blend due to LTI addition. J Mater Sci. 2006;41:4989–92.
Han LJ, Han CY, Dong LS. Morphology and properties of the biosourced poly(lactic acid)/poly(ethylene oxide-b-amide-12) blends. Polym Compos. 2013;34:122–30.
Yokohara T, Yamaguchi M. Structure and properties for biomass-based polyester blends of PLA and PBS. Eur Polym J. 2008;44:677–85.
Chen GX, Kim HS, Kim ES, Yoon JS. Compatibilization-like effect of reactive organoclay on the poly(l-lactide)/poly(butylene succinate) blends. Polymer. 2005;46:11829–36.
Coltelli MB, Bronco S, Chine C. The effect of free radical reactions on structure and properties of poly(lactic acid) (PLA) based blends. Polym Degrad Stab. 2010;95:332–41.
Li YJ, Shimizu H. Toughening of polylactide by melt blending with a biodegradable poly(ether)urethane elastomer. Macromol Biosci. 2007;7:921–8.
Chena H, Pydab M, Cebea P. Non-isothermal crystallization of PET/PLA blends. Thermochim Acta. 2009;492:61–6.
Han LJ, Han CY, Zhang HL, Chen S, Dong LS. Morphology and properties of biodegradable and biosourced polylactide blends with poly(3-hydroxybutyrate-co-4-hydroxybutyrate). Polym Compos. 2012;33:850–9.
Park JW, Im SS. Miscibility and morphology in blends of poly(l-lactic acid) and poly(vinyl acetate-co-vinyl alcohol). Polymer. 2003;44:4341–54.
You Y, Youk JH, Lee SW, Min BM, Lee SJ, Park WH. Preparation of porous ultrafine PGA fibers via selective dissolution of electrospun PGA/PLA blend fibers. Mater Lett. 2006;60:757–60.
Lin Y, Zhang KY, Dong ZM, Dong LS, Li YS. Study of hydrogen-bonded blend of polylactide with biodegradable hyperbranched poly(ester amide). Macromolecules. 2007;40:6257–67.
Ishida S, Nagasaki R, Chino K, Dong T, Inoue Y. Toughening of poly(l-lactide) by melt blending with rubbers. J Appl Polym Sci. 2009;113:558–66.
Jaratrotkamjorn R, Khaokong C, Tanrattanakul V. Toughness enhancement of poly(lactic acid) by melt blending with natural rubber. J Appl Polym Sci. 2012;124:5027–36.
Ash BJ, Siegel RW, Schadler LS. Glass-transition temperature behavior of alumina/PMMA nanocomposites. J Polym Sci B. 2004;42:4371–83.
Galka P, Kowalonek J, Kaczmarek H. Thermogravimetric analysis of thermal stability of poly(methyl methacrylate) films modified with photoinitiators. J Therm Anal Calorim. 2014;115:1387–94.
Pope EJA, Asami M, Mackenzie JD. Transparent silica gel–PMMA composites. J Mater Res. 1989;4:1018–26.
Clayton LM, Sikder AK, Kumar A, Cinke M, Meyyappan M, Gerasimov TG, Harmon JP. Transparent poly(methyl methacrylate)/single-walled carbon nanotube (PMMA/SWNT) composite films with increased dielectric constants. Adv Funct Mater. 2005;15:101–6.
Blanco I, Abate L, Antonelli ML. The regression of isothermal thermogravimetric data to evaluate degradation Ea values of polymers: a comparison with literature methods and an evaluation of lifetime prediction reliability. Polym Degrad Stab. 2011;96:1947–54.
Blanco I, Abate L, Antonelli ML, Bottino FA. The regression of isothermal thermogravimetric data to evaluate degradation Ea values of polymers: a comparison with literature methods and an evaluation of lifetime predictions reliability. Part II. Polym Degrad Stab. 2013;98:2291–6.
Zhang G, Zhang J, Wang S, Shen D. Miscibility and phase structure of binary blends of polylactide and poly(methyl methacrylate). J Polym Sci B. 2003;41:23–30.
Li SH, Woo EM. Immiscibility–miscibility phase transitions in blends of poly(l-lactide) with poly(methyl methacrylate). Polym Int. 2008;57:1242–51.
Samuel C, Raquez JM, Dubois P. PLLA/PMMA blends: a shear-induced miscibility with tunable morphologies and properties? Polymer. 2013;54:3931–9.
Shirahase T, Komatsu Y, Tominaga Y, Asai S, Sumita M. Miscibility and hydrolytic degradation in alkaline solution of poly(l-lactide) and poly(methyl methacrylate) blends. Polymer. 2006;47:4839–44.
Eguiburua JL, Iruina JJ, Fernandez-Berridia MJ, Roman JS. Blends of amorphous and crystalline polylactides with poly(methyl methacrylate) and poly(methyl acrylate): a miscibility study. Polymer. 1998;39:6891–7.
Hirota S, Sato T, Tominaga Y, Asai S, Sumita M. The effect of high-pressure carbon dioxide treatment on the crystallization behavior and mechanical properties of poly(l-lactic acid)/poly(methyl methacrylate) blends. Polymer. 2006;47:3954–60.
Xing PX, Dong LS, An YX, Feng ZL. Miscibility and crystallization of poly(β-hydroxybutyrate) and poly(p-vinylphenol) blends. Macromolecules. 1997;30:2726–33.
Lima LT, Aurasb R, Rubinob M. Processing technologies for poly(lactic acid). Prog Polym Sci. 2008;33:820–52.
Tsuji H. Crystallization from the melt of poly(lactide)s with different optical purities and their blends. Macromol Chem Phys. 1996;197:3483–99.
Fox TG. Influence of diluent and of copolymer composition on the glass temperature of a polymer system. Bull Am Phys Soc. 1956;1:123–5.
Gordon M, Taylor JS. Ideal copolymers and the second-order transitions of synthetic rubbers. I. Non-crystalline copolymers. J Appl Chem. 1952;2:493–500.
Bélorgey G, Aubin M, Prud’homme RE. Studies of polyester/chlorinated poly(vinyl chloride) blends. Polymer. 1982;23:1051–6.
Chiu FC, Min K. Miscibility, morphology and tensile properties of vinyl chloride polymer and poly(ε-caprolactone) blends. Polym Int. 2000;49:223–34.
Xing PX, Ai X, Dong LS, Feng ZL. Miscibility and crystallization of poly(β-hydroxybutyrate)/poly(vinyl acetate-co-vinyl alcohol) blends. Macromolecules. 1998;31:6898–907.
Pan PJ, Liang ZC, Zhu B, Dong T, Inoue Y. Blending effects on polymorphic crystallization of poly(l-lactide). Macromolecules. 2009;42:3374–80.
Chiu FC, Li MT. Miscibility, thermal properties and polymorphism of syndiotactic polystyrene/poly(styrene-co-α-methyl styrene) blends. Polymer. 2003;44:8013–23.
López LC, Wilkes GL. Non-isothermal crystallization kinetics of poly(p-phenylene sulphide). Polymer. 1989;30:882–7.
Avrami M. Kinetics of phase change. I. General theory. J Chem Phys. 1939;7:1103–12.
Avrami M. Kinetics of phase change. II. Transformation–time relations for random distribution of nuclei. J Chem Phys. 1940;8:212–24.
Nishil T, Wang TT. Melting point depression and kinetic effects of cooling on crystallization in poly(vinylidene fluoride)–poly (methyl methacrylate) mixtures. Macromolecules. 1975;8:909–15.
Hoffman JD, Weeks JJ. Melting process and the equilibrium melting temperature of polychlorotrifluoroethylene. J Res Natl Bur Stand A. 1962;66:13–28.
Sawai D, Tsugane Y, Tamada M, Kanamoto T, Sungil M, Hyon S. Crystal density and heat of fusion for a stereo-complex of poly(l-lactic acid) and poly(d-lactic acid). J Polym Sci B. 2007;45:2632–9.
Acknowledgements
This work is supported by the National Science Foundation of China (51021003, 50703042).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dong, Q., Bian, Y., Li, Y. et al. Miscibility and crystallization behaviors of stereocomplex-type poly(l- and d-lactide)/poly(methyl methacrylate) blends. J Therm Anal Calorim 118, 359–367 (2014). https://doi.org/10.1007/s10973-014-3966-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-014-3966-1