Abstract
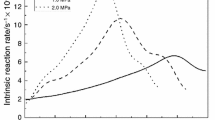
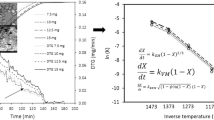
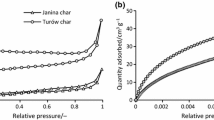
The char gasification characteristics and the composition of evolved gases in a CO2 environment have been studied using a thermogravimetric analyzer (TG) coupled with a mass spectrometer. Three types of coal char were studied: lignite (TXL), sub-bituminous (PRB), and bituminous (KYB). TG results showed that the reactivities of TXL and PRB were higher than that of KYB, and the reactivity of TXL was higher than that of PRB. The characterization of the chars implied that the mineral content in the char plays an important role in the reactivity and that the surface area and pore volume may accelerate the reactivity of chars. The evolved gases from three chars were mainly CO and SO2. SO2 was slightly delayed by CO during gasification of TXL and PRB chars, but for KYB, SO2 and CO formed in the same temperature range, but at higher temperatures compared with TXL and PRB. The CO production of KYB was the best, 0.98 mg mg−1; and SO2 was the least, 0.031 mg mg−1. PRB and TXL chars had similar CO production, but SO2 in TXL was higher.



Similar content being viewed by others
References
Kajitani S, Suzuki N, Ashizawa M, Hara S. CO2 gasification rate analysis of coal char in entrained flow coal gasifier. Fuel. 2006;85(2):163–9.
Liu H, Luo CH, Kato SG, Uemiya SYK, et al. Kinetics of CO2/Char gasification at elevated temperatures, Part I: experimental results. Fuel Process Technol. 2006;87:775–81.
Liu TF, Fang YT, Wang Y. An experimental investigation into the gasification reactivity of chars prepared at high temperatures. Fuel. 2008;87(4–5):460–6.
Sun QL, Li W, Chen HK, Li BQ. The CO2-gasification and kinetics of Shenmu maceral chars with and without catalyst. Fuel. 2004;83:1787–93.
Tomaszewicz M, Labojko G, Tomaszewicz G, Kotyczka M. The kinetics of CO2 gasification chars. J Therm Anal Claorim. 2013;113:1327–35.
Salatino P, Senneca O, Masi S. Assessment of thermodeactivation during gasification of a bituminous coal char. Energy Fuels. 1999;13:1154–9.
Shao J, Yan R, Chen H, Wang B, Lee D, Liang D. Pyrolysis characteristics and kinetics of sewage sludge by thermogravimetry Fourier transform infrared analysis. Energy Fuels. 2008;22:38–45.
Lu R, Purushothama S, Yang X, Hyatt J, Pan W, Riley J, Lloyd W. TG/FTIR/MS study of organic compounds evolved during the co-firing of coal and refuse-derived fuels. Fuel Process Technol. 1999;59:35–50.
Fushimi C, Araki K, Yamaguchi Y, Tsutsumi A. Effect of heating rates on steam gasification of biomass, thermogravimetric-mass spectrometric (TG-MS) analysis off gas evolution. Ind Eng Chem Res. 2003;42:3929–36.
Zhang LX, Huang JJ, Fang YT, Wang Y. Gasification reactivity and kinetics of typical Chinese anthracite chars with steam and CO2. Energy Fuels. 2006;20:1201–10.
Zhou ZJ, Lin M, Kuang JP, Liu JZ, et al. Effect of char making temperature and coke-forming time on char gasification specific activity. Coal Convers. 2006;29(3):21–4.
Hurt RH, Sarofim AF, Longwell JP. The role of microporous surface area in the gasification of chars from a sub-bituminous coal. Fuel. 1991;70(9):1079–82.
Sakawa M, Sakurai Y, Hara Y. Influence of coal characteristics on CO2 gasification. Fuel. 1982;61(8):717–20.
Morales IF, Garzon FJL, Peinado AL, et al. Study of heat-treated Spanish lignites: characteristics and behavior in CO2 and O2 gasification reactions. Fuel. 1985;64(5):664–73.
Zhong H, Pu WX, Si HA. The influence of included minerals on the intrinsic reactivity of chars prepared at 900 °C in a drop tube furnace and a muffle furnace. Fuel. 2009;88(11):2303–10.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, H., Cao, Y., Orndorff, W. et al. Gasification characteristics of coal char under CO2 atmosphere. J Therm Anal Calorim 116, 1267–1272 (2014). https://doi.org/10.1007/s10973-013-3627-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-013-3627-9