Abstract
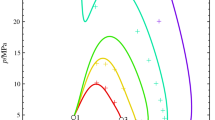
The relations between the shapes of the critical loci of binary fluid mixtures and the positions of the critical points of their component fluids on the p–T plane were considered theoretically. The consideration was carried out for vapor–liquid phase equilibria in the model systems without liquid–liquid equilibria. We focused only the relations between the shapes and the tangent of the straight lines connecting the critical points of the component fluids on the p–T plane, and so the effects of the distance between the critical points were out of the scope of this work. The changes in the shapes of the critical loci were classified logically into three patterns. Literature survey has shown that the two patterns exist in the experimental results for several types of mixtures. The shapes of the critical loci of the binary fluid mixtures on the p–T plane are closely related to the relative positions between the critical points of their component fluids.





Similar content being viewed by others
References
Hicks CP, Young CL. The gas–liquid critical properties of binary mixtures. Chem Rev. 1975;75:119–75.
Dohrn R, Brunner G. High-pressure fluid-phase equilibria: experimental methods and systems investigated (1988–1993). Fluid Phase Equilib. 1995;106:213–82.
Polishuk I, Wisniak J, Segura H. Simultaneous prediction of the critical and sub-critical phase behavior in mixtures using equation of state I. Carbon dioxide–alkanols. Chem Eng Sci. 2001;56:6485–510.
Polishuk I, Wisniak J, Segura H. Simultaneous prediction of the critical and sub-critical phase behavior in mixtures using equations of state II. Carbon dioxide–heavy n-alkanes. Chem Eng Sci. 2003;58:2529–50.
Polishuk I, Wisniak J, Segura H. Simultaneous prediction of the critical and sub-critical phase behavior in mixtures using equations of state III. Methane–n-alkanes. Chem Eng Sci. 2003;58:4363–76.
Van Konynenburg PH, Scott RL. Critical lines and phase equilibria in binary van der Waals mixtures. Phil Trans R Soc Lond A. 1980;298:495–540.
Rowlinson JS, Swinton FL. Liquids and liquid mixtures. 3rd ed. London: Butterworths Scientific; 1982. p. 191–4.
Kay WB. The critical locus curve and the phase behavior of mixtures. Acc Chem Res. 1968;1:344–51.
Kay WB. P–T–x diagrams in the critical region. Acetone–n-alkane systems. J Phys Chem. 1964;68:827–31.
Poettmann FH, Katz DL. Phase behavior of binary carbon dioxide: paraffin systems. Ind Eng Chem. 1945;37:847–53.
Ohgaki K, Katayama T. Isothermal vapor–liquid equilibrium data for the ethane–carbon dioxide system at high pressures. Fluid Phase Equilib. 1977;1:27–32.
Kay WB, Albert RE. Liquid–vapor equilibrium relations in the ethane–cyclohexane system. Ind Eng Chem. 1956;48:422–6.
Pak SC, Kay WB. The critical properties of binary hydrocarbon systems. Ind Eng Chem Fundam. 1972;11:255–67.
Sage BH, Lacey WN, Schaafsma JG. Phase equilibria in hydrocarbon systems II. Methane–propane system. Ind Eng Chem. 1934;26:214–7.
Akers WW, Burns JF, Fairchild WR. Low-temperature phase equilibria. Methane–propane system. Ind Eng Chem. 1954;46:2531–4.
Wichterle I, Kobayashi R. Vapor–liquid equilibrium of methane–propane system at low temperatures and high pressures. J Chem Eng Data. 1972;17:4–9.
McCurdy JL, Katz DL. Phase equilibria in the system. Ethane–ethylene–acetylene. Ind Eng Chem. 1944;36:674–80.
Diefenbacher A, Türk M. Critical (p, ρ, T) properties of CH2F2, {xCO2 + (1 − x)SF6}, {xSF6 + (1 − x)CH2F2}, and {xCHF3 + (1 − x)CH2F2}. J Chem Thermodyn. 1999;31:905–19.
Acknowledgments
This work was partly supported by the fund for Agriomics project from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tsukamoto, K., Fang, G., Maebayashi, M. et al. The shapes of critical loci of binary fluid mixtures on the p–T plane. J Therm Anal Calorim 113, 1551–1554 (2013). https://doi.org/10.1007/s10973-013-3073-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10973-013-3073-8